Psychiatric Comorbidities in Restless Legs Syndrome
Abstract
Restless legs syndrome (RLS) is a neurological sleep disorder with frequent (39%) coexisting psychiatric comorbidities. Patients with any psychiatric comorbidity had fewer periodic leg movements in sleep. Psychiatric disorders should be taken into account in patients with RLS.
Restless legs syndrome (RLS) is one of the most common neurological disorders, with prevalence estimates between 2% and 10% in the general population.1 An uncomfortable sensation mainly located in the legs, which worsens or appears in the evening or night during rest and improves or disappears with movement, is the core symptom required for the clinical diagnosis of RLS.2 Periodic limb movements (PLMs) during wakefulness or sleep occur in more than 80% of patients with RLS.3 RLS is typically associated with insomnia and sometimes with daytime sleepiness.4 Several studies, which were often retrospective and mainly used questionnaires but not a detailed psychiatric assessment, have shown an increased frequency of anxiety, depression, and somatoform disorders among patients with RLS.5,6 Psychiatric comorbidities in RLS present two challenging issues in clinical practice: first, a psychiatric comorbidity may complicate the diagnosis of RLS, which is based on subjective reports and requires careful recognition of the so-called “RLS mimics” conditions; and secondly, most antidepressant and neuroleptic drugs worsen RLS symptoms, which may pose a challenge for treatment.7,8 However, dopamine agonists, which represent the first-line treatment for RLS, may have antidepressant properties. Limited data from recent trials with dopamine agonists indicate an improvement in (mild) depressive symptoms among patients with RLS who also suffered from depression or anxiety disorders.9
This study aimed to determine the type and frequency of psychiatric comorbidities in patients with RLS and to characterize patients with RLS with and without psychiatric disorders.
Methods
Outpatients aged 18–65 years who fulfilled all essential diagnostic criteria for RLS2 were consecutively enrolled in a prospective observational study. An announcement was placed in the local newspaper to help with recruitment. This study included only drug-naïve patients who were affected by an idiopathic form of RLS and had not had any previous RLS therapy or other pharmacological treatments. Individuals who had symptomatic forms of RLS or any other neurological, medical, or other sleep disorder were excluded from this study. The neurological examination had to be unremarkable in order for a patient to be included in the study. Routine blood tests (including thyroid-stimulating hormone, ferritin, and vitamin B12) and PLM analysis were performed for all patients. The diagnosis of RLS was assessed by trained neurologists who are experts in sleep disorders (U.K. and C.L.A.B.). A detailed psychiatric assessment was based on a computer-supported personal interview using the Composite International Interview10 (CIDI/DIA-X) for DSM-IV and was performed by trained psychologists (A.S. and S.T.). Further assessment included questionnaires on the severity of RLS (International Restless Legs Syndrome Study Group Rating Scale [IRLS], overall score 0–40),11 quality of life (12-Item Short Form Health Survey [SF-12]), and daytime sleepiness (Epworth Sleepiness Scale [ESS]). We used the Actiwatch monitor system (Cambridge Neuro-Technology Ltd., Cambridge, UK) to measure PLMs.12 The Actiwatch has high sensitivity and specificity to detect PLMs. It represents a convenient and cost-effective alternative to polysomnography. Actigraph measurements were obtained from both legs over three consecutive nights at the patient’s home. The analysis only considered the periods when the patient was in bed with the lights turned off. PLMs were analyzed automatically and checked visually. Periods with clear artifacts were excluded from the analysis. A PLM index was calculated as the number of PLMs per hour of night by using the standard scoring criteria.13 The PLM index from all three nights and from both legs was then averaged.
This study was conducted in accordance with good clinical practice and was approved by the local ethics committee. All patients provided written informed consent.
Results
The inclusion process is shown in Figure 1. Forty-nine patients were included in this study (34 women [69.4%] and 15 men). The mean age was 49.9±11.3 years (range=24–65 years) and the mean IRLS score was 17±8. The mean PLM index was 23.6±21.6 (N=48); PLM data from one patient could not be included for analysis because of technical problems. There were no statistically significant differences in age, gender, IRLS score, or laboratory data (Table 1).
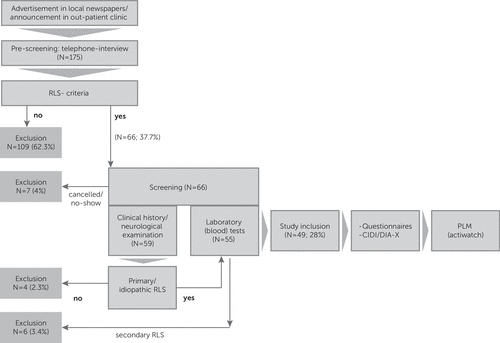
FIGURE 1. Study Inclusion Processa
aCIDI/DIA-X, Composite International Interview; PLM, periodic limb movement; RLS, restless legs syndrome.
| Parameter | RLS (N=30) | RLS Plus Comorbid Psychiatric Disorder (N=19) | p Value |
|---|---|---|---|
| Demographic data | |||
| Age (years) | 49.4±11.1 | 50.6±11.9 | 0.726 |
| Gender (male/female) | 12/18 | 3/16 | 0.068 |
| Smoking status (nonsmoker/smoker) | 28/2 | 17/2 | 0.507 |
| Laboratory values | |||
| Ferritin (ng/ml) | 99.0±107.3 | 81.1±65.3 | 0.524 |
| Vitamine B12 (ng/l) | 324.5±93.4 | 296.2±75.2 | 0.312 |
| Folate (nmol/l) | 9.1±7.2 | 10.3±5.0 | 0.404 |
| TSH basal (µIU/ml) | 2.1±1.7 | 1.8±1.0 | 0.275 |
| RLS-PLM data (N) | |||
| Family history of RLS | |||
| Yes | 12 | 13 | |
| No | 15 | 6 | 0.097 |
| Unknown | 3 | 0 | Chi-square=4.6 |
| IRLS score | 16±6.6 | 18±10 | 0.311 |
| PLM index (N=29/19) | 26.9±18.0 | 18.5±21.0 | 0.149 |
| Questionnaires | |||
| Epworth Sleepiness Scale | 7.0±3.1 | 6.5±3.0 | 0.618 |
| Fatigue Severity Scale | 3.4±2.1 | 4.1±1.4 | 0.145 |
TABLE 1. RLS/PLM Data for Patients With RLS With and Without Comorbid Psychiatric Disordersa
A psychiatric diagnosis was identified in 19 of 49 patients (39%) according to the CIDI/DIAX. Fourteen patients had a single psychiatric diagnosis; five patients had more than one. Nine patients (18.3%) had a somatoform disorder, eight patients (16.3%) had a depressive disorder, and six patients (12.2%) had an anxiety disorder. Two patients (4.1%) fulfilled criteria for a posttraumatic stress disorder; one patient had an eating disorder. According to the patients’ medical history, RLS preceded depression in 75% of patients, whereas anxiety and/or panic disturbances appeared before RLS symptoms in 83%. Patients with higher disease severity (IRLS scores ≥20) more often had a psychiatric comorbidity (10 of 17 versus 9 of 32; p=0.037). Patients with any psychiatric comorbidity had fewer PLMs. In patients with RLS without a psychiatric comorbidity, PLMs (index >5) occurred among 28 of 29 (97%) and 18 of 29 (62%) when the PLM index was >15 per hour, respectively. In contrast, among patients with RLS suffering from a comorbid psychiatric disorder, only 10 of 18 (55.6%) had a PLM index >5 and only six of 18 (33.3%) had a PLM index >15. The comparison of groups (RLS versus RLS plus psychiatric comorbidity) with a PLM index >15 was statistically significant (p=0.052) (Figure 2).
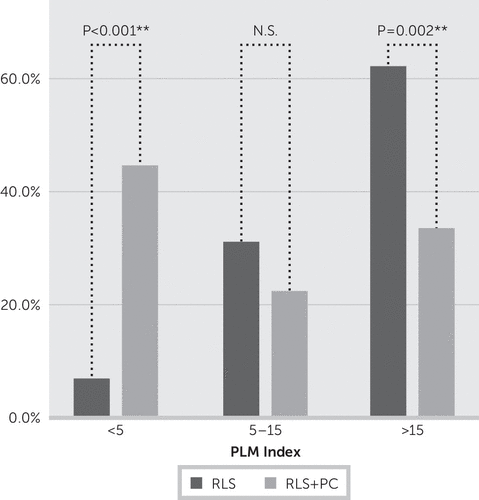
FIGURE 2. PLM Index in Patients With RLS Versus Patients With RLS Plus Psychiatric Comorbidity, According to PLM Index Categoriesa
a PC, psychiatric comorbidity; PLM, periodic limb movement; RLS, restless legs syndrome.
Quality of life was worse in patients with psychiatric codisorders (17 versus 30 points on the SF-12, p=0.001).
No differences were found in blood analysis results for patients with and without a psychiatric comorbidity (Table 1).
Discussion
This prospective study using a detailed psychiatric assessment confirms previous reports indicating the frequent occurrence of psychiatric codisorders, particularly affective disorders, in patients with RLS.5,6 Interestingly, RLS usually preceded depression and thus might be considered as a risk factor for depression. Recent epidemiologic studies support our data14 and point to a bidirectional relationship between RLS and depression.
PLM is an important associated feature for the diagnosis of RLS, occurring in more than 80% of patients. There are diverging results on the relationship between IRLS score and the PLM index.15 In our series, 78% of patients with RLS (38 of 49) had PLMs. The majority of patients without PLMs (10 of 11, 91%) had a psychiatric comorbidity. Taking into account that these patients described a more severe disease severity (IRLS score), this might be a confounding factor in RLS studies explaining the missing correlation between IRLS and the PLM index.
One possible explanation for the lower number of PLMs in patients with RLS with a psychiatric comorbidity is different sleep architecture. In RLS, PLMs mainly occur in non-REM (NREM) sleep and in the first part of the night (first sleep cycle). Depression is associated with short REM sleep latency. In patients with RLS with a psychiatric comorbidity, patients rapidly enter into REM sleep and thus spend less time in NREM sleep and consequently have fewer PLMs.
A detailed psychiatric examination and identification of psychiatric comorbidity is essentially needed to better differentiate and treat patients with RLS. The PLM index seems to be useful to differentiate RLS from RLS with psychiatric comorbidity. Our study supports the need to perform PLM analysis in patients with RLS and strengthens the concept of an “RLS-like” phenotype that is different from RLS-PLMs and is characterized by comorbid psychiatric disorders without or with few PLMs but with subjective description of a severe disease burden.
Limitations of this study include its small sample size and consequent inadequate statistical power. Larger studies are needed to confirm our study results and to perform additional (subgroup) analysis on age and gender relationships and on the clinical course of both illnesses.
1 : Epidemiology of restless legs syndrome: a synthesis of the literature. Sleep Med Rev 2012; 16:283–295Crossref, Medline, Google Scholar
2 : Restless legs syndrome/Willis-Ekbom disease diagnostic criteria: updated International Restless Legs Syndrome Study Group (IRLSSG) consensus criteria--history, rationale, description, and significance. Sleep Med 2014; 15:860–873Crossref, Medline, Google Scholar
3 : Is daytime sleepiness a neglected problem in patients with restless legs syndrome? Mov Disord 2007; 22(Suppl 18):S409–S413Crossref, Medline, Google Scholar
4 : Dopaminergic treatment in idiopathic restless legs syndrome: effects on subjective sleepiness. Clin Neuropharmacol 2010; 33:276–278Crossref, Medline, Google Scholar
5 : “Anxietas tibiarum”. Depression and anxiety disorders in patients with restless legs syndrome. J Neurol 2005; 252:67–71Crossref, Medline, Google Scholar
6 : Restless legs syndrome is associated with DSM-IV major depressive disorder and panic disorder in the community. J Neuropsychiatry Clin Neurosci 2008; 20:101–105Link, Google Scholar
7 : Mirtazapine-induced worsening of restless legs syndrome (RLS) and ropinirole-induced psychosis: challenges in management of depression in RLS. Psychosomatics 2011; 52:92–94Crossref, Medline, Google Scholar
8 : Restless legs syndrome induced by escitalopram: case report and review of the literature. Pharmacotherapy 2008; 28:271–280Crossref, Medline, Google Scholar
9 : Ropinirole improves depressive symptoms and restless legs syndrome severity in RLS patients: a multicentre, randomized, placebo-controlled study. J Neurol 2011; 258:1046–1054Crossref, Medline, Google Scholar
10 : The World Mental Health (WMH) Survey Initiative Version of the World Health Organization (WHO) Composite International Diagnostic Interview (CIDI). Int J Methods Psychiatr Res 2004; 13:93–121Crossref, Medline, Google Scholar
11 : Validation of the International Restless Legs Syndrome Study Group rating scale for restless legs syndrome. Sleep Med 2003; 4:121–132Crossref, Medline, Google Scholar
12 : The validation of a new actigraphy system for the measurement of periodic leg movements in sleep. Sleep Med 2005; 6:507–513Crossref, Medline, Google Scholar
13 : The official World Association of Sleep Medicine (WASM) standards for recording and scoring periodic leg movements in sleep (PLMS) and wakefulness (PLMW) developed in collaboration with a task force from the International Restless Legs Syndrome Study Group (IRLSSG). Sleep Med 2006; 7:175–183Crossref, Medline, Google Scholar
14 : The relationship between depressive symptoms and restless legs syndrome in two prospective cohort studies. Psychosom Med 2013; 75:359–365Crossref, Medline, Google Scholar
15 : Relationship of periodic leg movements and severity of restless legs syndrome: a study in unmedicated and medicated patients. Clin Neurophysiol 2007; 118:1532–1537Crossref, Medline, Google Scholar



