The Influence of Neuropsychological Functioning on Neuropsychiatric Problems in Dementia
Several cross-sectional studies have reported associations between neuropsychological impairments and psychosis, but less is known about other neuropsychiatric problems. Studies of neuropsychological correlates of psychosis, and specifically delusions, have provided conflicting results. Though some authors found associations with frontal-temporal-related deficits, 3 – 5 others did not find more neuropsychological deficits in patients with delusions. 6 , 7 Furthermore, specific associations have been found between agitation and executive dysfunction; 8 apathy and lower scores on tests of verbal memory, naming, set shifting, and verbal fluency; 9 and depression and impairments of recent memory and attention. 10 In contrast, other authors found no association between cognitive dysfunction and depression, 9 anxiety, 10 or apathy and irritability. 11
Several methodological limitations, such as small samples of patients, the use of different instruments for assessing neuropsychological impairments and neuropsychiatric problems, and a cross-sectional study design, may add to these discrepancies. The aim of our study was to determine the influence of specific neuropsychological impairments on the development of a broad range of neuropsychiatric problems in patients with dementia. To our knowledge, this is the first large prospective study of this topic. We hypothesized that specific types of cognitive impairment, and in particular, language impairments, are related to more severe neuropsychiatric problems. Patients with dementia who have difficulties with expressing their thoughts and feelings may express this by means of neuropsychiatric symptoms.
METHOD
Patients
The present study was part of the Maastricht Study of Behavior in Dementia (MAASBED), a study which focuses on the course and risk factors of behavioral and psychological symptoms in dementia. MAASBED is a 2-year prospective study of 199 community-based patients with dementia who are seen at 6-month intervals. Patients were subsequently referred to the Maastricht Memory Clinic of the University Hospital or the geriatric division of the Regional Institute for Community Mental Health (RIAGG). We assessed 126 patients for whom complete 2-year follow-up data were available.
Patients were included if they met DSM-IV criteria for dementia 12 and if there was a reliable informant. One hundred patients met the NINCDS-ADRDA criteria 13 for (possible, probable) Alzheimer’s type dementia; 15 patients met the NINCDS-AIREN criteria for vascular dementia; 14 and 11 patients met criteria for dementia due to multiple etiologies. Patients were excluded if they were living in a nursing home at the start of the study. Written consent was given by the caregiver, and, when possible, by the patient. The Medical Ethics Committee of the University Hospital Maastricht approved this study.
Neuropsychiatric Problems
Neuropsychiatric problems were assessed with the Neuropsychiatric Inventory (NPI), 15 an informant-based rating scale developed to assess psychopathology in patients with dementia. The current version 16 evaluates 12 neuropsychiatric symptoms commonly observed in dementia. The severity and frequency of each symptom are scored on the basis of structured questions administered to the patient’s caregiver. The score for each symptom is obtained by multiplying severity (1 to 3) by frequency (1 to 4). The summed symptom scores give the total NPI score. The validity and reliability of the NPI 17 and its Dutch version 18 have been established.
In a previous study, principal component analysis of the NPI identified three neuropsychiatric subsyndromes: 1) a “mood/apathy” factor, including depression, apathy, night-time behavior disturbances, and eating abnormalities (four items, Cronbach’s alpha=0.63); 2) a “hyperactivity factor,” including symptoms of agitation, euphoria, irritability, disinhibition, and aberrant motor behavior (five items, Cronbach’s alpha=0.73); and 3) a “psychosis factor,” including hallucinations and delusions (two items, Cronbach’s alpha=0.72). 19 The total score of each subsyndrome was calculated by summing the NPI item scores for each factor.
Neuropsychological Assessment
In addition to general information on demographics and medical history gathered from the primary caregivers, trained research assistants administered cognitive tests to the patients.
The neuropsychological assessment covered the domains of language, memory, executive functioning, attention, orientation, perception, and praxis. Tests included:
| • | A 10-item word list memory task 20 based on the Auditory Verbal Learning Test. It is an episodic memory test in which words are orally presented, one after another. Then the subject is asked to recall as many words as possible. This procedure is repeated five times, and after 20 minutes, delayed recall and recognition are tested. | ||||
| • | Naming subtests of the Aachen Aphasia test 21 were used to determine impairments in naming abilities | ||||
| • | The Verbal Fluency Test (categorical version) 22 measures the ability to produce as many words as possible in a fixed time span. It can be regarded as a measure for the adequate, strategy-driven retrieval of information from semantic memory. We asked the subjects to name as many animals and professions as possible within 90 seconds. | ||||
| • | The Cambridge Cognitive Examination (CAMCOG), which is part of the Cambridge Examination for Mental Disorder of the Elderly (CAMDEX). 23 , 24 The cognitive functions assessed with the CAMCOG included language (comprehension and expression), memory (recent, remote, and new learning), attention/calculation, praxis, abstract reasoning, perception, and orientation. It consists of 64 items with a total range of 0 (severe cognitive impairment) to 105 (no cognitive impairment). The CAMCOG is often used for the diagnosis and gradation of dementia, and one of its major advantages is the ability to detect mild forms of cognitive impairment. The Mini-Mental State Examination (MMSE) 25 score was derived from the CAMCOG. | ||||
| • | The Global Deterioration Scale (GDS) 26 was used to rate the severity of dementia. The GDS is subdivided into seven stages, ranging from no cognitive deterioration at all (Stage 1) to severe cognitive and functional deterioration (Stage 7). It is one of the most widely used clinician-rated instruments to stage the course and severity of dementia. In the present study, the GDS score was included as a covariate, instead of the often-used MMSE score, because of the strong association between MMSE score and neuropsychological impairments 27 – 29 and specifically the CAMCOG. 30 In addition to cognition, the GDS covers daily life functioning and behavioral aspects. The GDS was included as a covariate to remove the effect of general cognitive decline and ensure the specificity of the findings. | ||||
Statistical Analyses
We performed statistical analyses with the Statistical Package for Social Sciences (SPSS), version 10. When a patient had only one missing assessment during the study, missing data of the NPI were imputed by either “mean substitution” of the previous and the next assessment, or “last case carried forward.” Thus, the present study involved 126 patients for whom complete 2-year follow-up data (five assessments of the NPI) were available. To determine whether cognitive performance at baseline was a predictor of neuropsychiatric problems over the 2-year period, repeated measures analyses of covariance (ANCOVAs) were performed for each of 13 different cognitive domains. These included the 10-Item Word List Task, Aachen Aphasia naming task, Verbal Fluency Test, CAMCOG total score, language (expression, comprehension), memory, attention, praxis, abstract reasoning, perception, and orientation. Patients were assigned to “low performance” and “high performance” groups according to a median-split on each cognitive neuropsychological domain as measured at baseline. “Group” (high versus low performance at baseline) was entered into the ANCOVAs as the between-subjects factor; and “time” (baseline, 6 months, 1 year, 18 months, and 2-year follow-up) was entered as the within-subject factor, after correction for global dementia severity. The following eight NPI variables were entered as dependent variables for the repeated measures analyses: the three subsyndromes, NPI total score and the separate symptoms, delusions, depression, apathy, and aberrant motor behavior. These latter symptoms were selected for separate analyses because of their high prevalence among dementia patients. 1
For only 122 and 113 patients were complete data for the CAMCOG and the Word List Memory Task, respectively, available; thus, analyses with these variables had lower sample sizes. All other analyses were performed on data for 126 patients. All significance tests were performed at a two-tailed alpha level of 0.05. Because of the explorative nature of the study, Bonferroni-corrections were not made.
RESULTS
Patient Characteristics
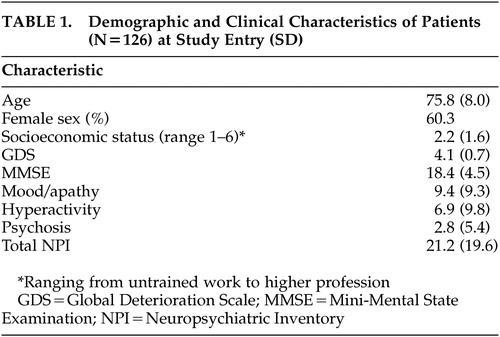
Table 1 shows the demographic and clinical characteristics of patients. At baseline, 50 men (39.7%) and 76 women (60.3%) were included in the study. The mean age was 75.8 (SD=8.0) years (range=53 to 92 years). Most patients had a relatively low socioeconomic status. Patients had an average MMSE score of 18.4 (SD=4.5) (mild to moderate severity) at baseline, which decreased to 14.7 (SD=5.8) (moderate to severe dementia) after 2 years. GDS scores increased during follow-up, indicating that the severity of dementia increased with time. We did not distinguish between different types of dementia because too few patients had a diagnosis other than Alzheimer’s disease. Separate analysis using the data of the group of Alzheimer’s patients alone yielded similar results.
 |
A number of patients dropped out during the study (refusal, n=37; death, n=36), so that complete 2-year follow-up data were available for 126 patients (63.3%). At study entry, neuropsychiatric problems (NPI total score and scores for sub-syndromes), severity of dementia (MMSE and GDS), and most major demographic characteristics did not differ between the patients who did or did not complete the study. However, the patients who died during the study had significant higher GDS scores than the patients who refused (GDS score of 4.5 versus 4, respectively; p=0.004), and significant higher GDS scores (GDS score of 4.5 versus 4.1, respectively; p=0.001), and age (79.2 years versus 75.8 years; p=0.027) compared with the patients who completed the study.
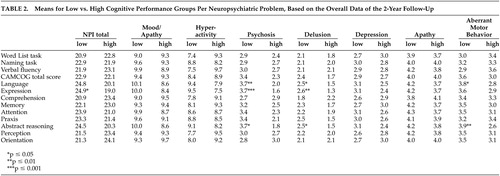
Cognitive Functioning as a Predictor of Neuropsychiatric Problems
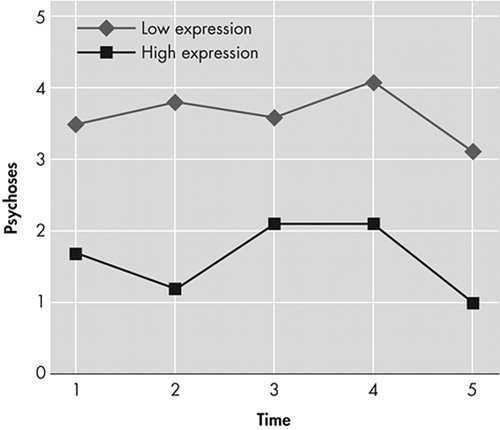
There were no main effects of time and no interaction effects between time and cognitive variables for any of the neuropsychiatric problems. There were no significant findings demonstrating a relationship between any of the cognitive function variables and the NPI subsyndromes of mood/apathy, hyperactivity, and the separate symptoms of depression and apathy ( Table 2 ). There were significant main effects of language impairment on the subsyndrome psychosis (F=7.8 (1, 119) , p=0.006) and on the symptoms of delusions (F=6.2 (1, 119) , p=0.014) and aberrant motor behavior (F=4.5 (1, 119) , p=0.037). Separate analyses revealed specific significant main effects of language expression on psychosis (F=10.8 (1, 119) , p=0.001) ( Figure 1 ), delusions (F=9.5 (1, 119) , p=0.003), and total NPI score (F=5.7 (1, 119) , p=0.019). There was not a significant relationship between language comprehension and any neuropsychiatric problem. In addition, there were significant main effects of abstract reasoning on psychosis (F=9.4 (1, 119) , p=0.003), delusions (F=6.3 (1, 119) , p=0.013), and aberrant motor behavior (F=7.2 (1, 119) , p=0.008).
 |
DISCUSSION
We investigated how neuropsychological functioning affected the development of neuropsychiatric problems over a 2-year period. An impairment of language expression was found to be related to overall neuropsychiatric problems, and to psychosis in particular. Aberrant motor behavior was associated with overall language impairment. These findings are consistent with those of Potkins et al., 31 who reported an association between expressive language impairment and the presence of delusions in their cross-sectional study. In contrast to our study, they found aberrant motor behavior to be specifically associated with receptive language difficulties. We found language comprehension to be unrelated to any neuropsychiatric problem, which may be because our subjects suffered from less severe dementia than the subjects of the study of Potkins et al., which included patients living in residential homes. In addition, we measured language comprehension just with the CAMCOG, which may not have been sensitive enough for detecting comprehension impairments. Ballard et al. 32 also found an association between psychosis and expressive language, as measured with the CAMCOG. It is hypothesized that an impaired ability to communicate verbally may trigger frustration, resulting in neuropsychiatric problems, and especially psychosis.
However, further research is needed to confirm this hypothesis. As mentioned by Potkins et al., aberrant motor behavior may be the consequence of increased social isolation and diminished participation in activities as a result of poor language skills. 31 The cognitive domains for naming ability and verbal fluency had no influence on neuropsychiatric problems, suggesting that the appearance of symptoms is more related to impairments of language expression than to a dysfunction of semantic memory. The influence of verbal expression on psychosis and other neuropsychiatric problems emphasizes the role of temporal lobe lesions, with the disruption of limbic connections, in the development of these disorders. This is consistent with the results of Hirono et al., 33 , 34 who found dysfunction of temporal brain areas in Alzheimer’s patients with delusions and aggression. Language functions in the temporal lobe might involve the posterior and superior dorsal regions, while the temporo-limbic connections linked with psychosis might involve medial temporal areas.
We also found that both psychosis and aberrant motor behavior were influenced by impairments of abstract reasoning, a frontal lobe function. There is substantial evidence that patients with psychosis have frontal lobe and executive function deficits, 3 – 5 , 35 and neuroimaging studies have shown dysfunction of the frontal lobes 36 – 38 and temporal areas 39 – 42 in dementia patients with psychosis. This pattern of results lends support to the theory that frontal-temporal (limbic) dysfunction may underlie the presence of psychosis, and specifically delusions, in dementia. Lopez et al. 42 suggested that dysfunction of both the frontal lobe and the temporal lobe is needed for the development of psychotic symptoms.
Less is known about neuropsychological correlates of aberrant motor behavior. Previous research has focused on neuropsychological correlates of wandering, a symptom included in the NPI symptom “aberrant motor behavior.” A recent review of wandering behavior reported controversies about the relationship with specific neuropsychological correlates. 43 Our data suggest that neuropsychiatric problems are related to impairments of language and abstract reasoning. It remains to be determined whether aberrant motor behavior, other neuropsychiatric problems, and psychosis can be explained by a common underlying pathophysiological alteration or whether they can be explained by psychological mechanisms. Several studies have mentioned the possibility of a shared neuropathology. 6 , 42 , 44 Therefore, it must be kept in mind that it may be possible that the neuropsychological correlates and the neuropsychiatric problems are the consequence of the same organic defect in the brain.
Our study had several limitations. A first limitation of our study is that it was based on only 126 patients (63.3%, i.e., only 63% of the original sample had complete data). However, patients lost to follow-up did not differ in terms of severity of neuropsychiatric problems or severity of dementia at baseline from the patients who completed this study, so selective attrition seems unlikely. A second important limitation is that our conclusions must be regarded as preliminary, needing further research. Because of the explorative nature of the study, Bonferroni corrections were not made, increasing the chance of type I errors (i.e., potential for false positives) given the large number of comparisons. In addition, by dichotomizing the cognitive scores, additional information may be lost. Lastly, the lack of time effect should be mentioned. This suggests that specific forms of cognitive dysfunction at baseline did not predict the course or progression of various neuropsychiatric problems. But the main effects did suggest associations between specific cognitive deficits and neuropsychiatric problems that held constant across all time points.
In conclusion, our findings indicate that impairments of language expression and abstract reasoning are related to the emergence of psychosis, aberrant motor behavior, and total level of neuropsychiatric problems. These results imply that clinicians should be more alert to communication and executive deficits in patients with dementia, as they point to patients who are more prone to develop these types of noncognitive symptoms. In doing so, they can help patients to compensate for these problems and thereby prevent the development of neuropsychiatric problems.
1. Aalten P, de Vugt ME, Jaspers N, et al: The course of neuropsychiatric symptoms in dementia: part I: findings from the two-year longitudinal MAASBED study. Int J Geriatr Psychiatry 2005; 20:523–530Google Scholar
2. Aalten P, de Vugt ME, Jaspers N, et al: The course of neuropsychiatric symptoms in dementia: part II: relationships among behavioural sub-syndromes and the influence of clinical variables. Int J Geriatr Psychiatry 2005; 20:531–536Google Scholar
3. Lopez OL, Becker JT, Brenner RP, et al: Alzheimer’s disease with delusions and hallucinations: neuropsychological and electroencephalographic correlates. Neurology 1991; 41:906–912Google Scholar
4. Jeste DV, Wragg RE, Salmon DP, et al: Cognitive deficits of patients with Alzheimer’s disease with and without delusions. Am J Psychiatry 1992; 149:184–189Google Scholar
5. Paulsen JS, Salmon DP, Thal LJ, et al: Incidence of and risk factors for hallucinations and delusions in patients with probable AD. Neurology 2000; 54:1965–1971Google Scholar
6. Flynn FG, Cummings JL, Gornbein J: Delusions in dementia syndromes: investigation of behavioral and neuropsychological correlates. J Neuropsychiatry Clin Neurosci 1991; 3:364–370Google Scholar
7. Migliorelli R, Petracca G, Teson A, et al: Neuropsychiatric and neuropsychological correlates of delusions in Alzheimer’s disease. Psychol Med 1995; 25:505–513Google Scholar
8. Chen ST, Sultzer DL, Hinkin CH, et al: Executive dysfunction in Alzheimer’s disease: association with neuropsychiatric symptoms and functional impairment. J Neuropsychiatry Clin Neurosci 1998; 10:426–432Google Scholar
9. Kuzis G, Sabe L, Tiberti C, et al: Neuropsychological correlates of apathy and depression in patients with dementia. Neurology 1999; 52:1403–1407Google Scholar
10. Ballard C, Neill D, O’Brien J, et al: Anxiety, depression and psychosis in vascular dementia: prevalence and associations. J Affect Disord 2000; 59:97–106Google Scholar
11. Starkstein SE, Migliorelli R, Manes F, et al: The prevalence and clinical correlates of apathy and irritability in Alzheimer’s disease. Eur J Neurol 1995; 2:540–546Google Scholar
12. American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders, 4th ed. Washington, DC, American Psychiatric Association, 1994Google Scholar
13. McKhann G, Drachman D, Folstein M, et al: Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 1984; 34:939–944Google Scholar
14. Roman GC, Tatemichi TK, Erkinjuntti T, et al: Vascular dementia: diagnostic criteria for research studies: report of the NINDS-AIREN International Workshop. Neurology 1993; 43:250–260Google Scholar
15. Cummings JL, Mega M, Gray K, et al: The Neuropsychiatric Inventory: comprehensive assessment of psychopathology in dementia. Neurology 1994; 44:2308–2314Google Scholar
16. Cummings JL: The Neuropsychiatric Inventory: assessing psychopathology in dementia patients. Neurology 1997; 48:10–16Google Scholar
17. Cummings JL, McPherson S: Neuropsychiatric assessment of Alzheimer’s disease and related dementias. Aging 2001; 13:240–246Google Scholar
18. Kat MG, de Jonghe JF, Aalten P, et al: Neuropsychiatric symptoms of dementia: psychometric aspects of the Dutch Neuropsychiatric Inventory (NPI). Tijdschr Gerontol Geriatr 2002; 33:150–155Google Scholar
19. Aalten P, De Vugt ME, Lousberg R, et al: Behavioral problems in dementia: a factor analysis of the Neuropsychiatric Inventory. Dement Geriatr Cogn Disord 2003; 15:99–105Google Scholar
20. Brand N, Jolles J: Learning and retrieval rate of words presented auditorily and visually. J Gen Psychol 1985; 112:201–210Google Scholar
21. Huber WK, Poeck D, Weniger K, et al: Der Aachener Aphasia Test. Gottingen, Hogrefe, 1983Google Scholar
22. Lezak: Neuropsychological Assessment, 3rd ed. New York, Oxford University Press, 1995Google Scholar
23. Roth M, Tym E, Mountjoy CQ, et al: CAMDEX: a standardised instrument for the diagnosis of mental disorder in the elderly with special reference to the early detection of dementia. Br J Psychiatry 1986; 149:698–709Google Scholar
24. Verhey FR, Huppert FA, Korten EC, et al: Cross-national comparisons of the Cambridge Cognitive Examination-Revised: the CAMCOG-R: results from the European Harmonization Project for instruments in dementia. Age Ageing 2003; 32:534–540Google Scholar
25. Folstein MF, Folstein SE, McHugh PR: “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975; 12:189–198Google Scholar
26. Reisberg B, Ferris SH, de Leon MJ, et al: The Global Deterioration Scale for assessment of primary degenerative dementia. Am J Psychiatry 1982; 139:1136–1139Google Scholar
27. Jefferson AL, Cosentino SA, Ball SK, et al: Errors produced on the Mini-Mental State Examination and neuropsychological test performance in Alzheimer’s disease, ischemic vascular dementia, and Parkinson’s disease. J Neuropsychiatry Clin Neurosci 2002; 14:311–320Google Scholar
28. Tierney MC, Szalai JP, Snow WG, et al: Domain specificity of the subtests of the Mini-Mental State Examination. Arch Neurol 1997; 54:713–716Google Scholar
29. Tombaugh TN, McIntyre NJ: The Mini-Mental State Examination: a comprehensive review. J Am Geriatr Soc 1992; 40:922–935Google Scholar
30. Blessed G, Black SE, Butler T, et al: The diagnosis of dementia in the elderly: a comparison of CAMCOG (the cognitive section of CAMDEX), the AGECAT program, DSM-III, the Mini-Mental State Examination and some short rating scales. Br J Psychiatry 1991; 159:193–198Google Scholar
31. Potkins D, Myint P, Bannister C, et al: Language impairment in dementia: impact on symptoms and care needs in residential homes. Int J Geriatr Psychiatry 2003; 18:1002–1006Google Scholar
32. Ballard CG, Bannister CL, Patel A, et al: Classification of psychotic symptoms in dementia sufferers. Acta Psychiatr Scand 1995; 92:63–68Google Scholar
33. Hirono N, Mori E, Ishii K, et al: Alteration of regional cerebral glucose utilization with delusions in Alzheimer’s disease. J Neuropsychiatry Clin Neurosci 1998; 10:433–439Google Scholar
34. Hirono N, Mega MS, Dinov ID, et al: Left frontotemporal hypoperfusion is associated with aggression in patients with dementia. Arch Neurol 2000; 57:861–866Google Scholar
35. Schneider LS, Dagerman KS: Psychosis of Alzheimer’s disease: clinical characteristics and history. J Psychiatr Res 2004; 38:105–111Google Scholar
36. Sultzer DL, Mahler ME, Mandelkern MA, et al: The relationship between psychiatric symptoms and regional cortical metabolism in Alzheimer’s disease. J Neuropsychiatry Clin Neurosci 1995; 7:476–484Google Scholar
37. Mega MS, Lee L, Dinov ID, et al: Cerebral correlates of psychotic symptoms in Alzheimer’s disease. J Neurol Neurosurg Psychiatry 2000; 69:167–171Google Scholar
38. Sultzer DL, Brown CV, Mandelkern MA, et al: Delusional thoughts and regional frontal/temporal cortex metabolism in Alzheimer’s disease. Am J Psychiatry 2003; 160:341–349Google Scholar
39. Cummings JL: Psychosis in neurologic disease: neurobiology and pathogenesis. Neuropsychiatry Neuropsychol Behav Neurol 1992; 5:144–150Google Scholar
40. Starkstein SE, Vazquez S, Petracca G, et al: A SPECT study of delusions in Alzheimer’s disease. Neurology 1994; 44:2055–2059Google Scholar
41. Kotrla KJ, Chacko RC, Harper RG, et al: SPECT findings on psychosis in Alzheimer’s disease. Am J Psychiatry 1995; 152:1470–1475Google Scholar
42. Lopez OL, Smith G, Becker JT, et al: The psychotic phenomenon in probable Alzheimer’s disease: a positron emission tomography study. J Neuropsychiatry Clin Neurosci 2001; 13:50–55Google Scholar
43. Lay CKY, Arthur DG: Wandering behaviour in people with dementia. J Advanced Nursing 2003; 44:173–182Google Scholar
44. Lopez OL, Becker JT, Sweet RA, et al: Psychiatric symptoms vary with the severity of dementia in probable Alzheimer’s disease. J Neuropsychiatry Clin Neurosci 2003; 15:346–353Google Scholar



