Soft Neurological Signs in Drug-Free People With Schizophrenia With and Without Obsessive-Compulsive Symptoms
Abstract
Obsessive-compulsive (OC) symptoms are clinically important phenomena in people with schizophrenia. Schizophrenia and obsessive-compulsive disorder (OCD) are considered as neurodevelopmental disorders with dysfunctional frontal subcortical circuitry. In this cross-sectional study, the authors tested the hypothesis that people with schizophrenia with OC symptoms form a subtype with a distinct neurological substrate. Using the Neurological Evaluation Scale (NES), the authors assessed 65 people with ICD-10 criteria for schizophrenia, 21 of whom had OC symptoms, and who were drug-naive or had not received any antipsychotic or anti-obsessive drugs for the previous 3 months. People with schizophrenia and OC symptoms were better educated than those without OC symptoms and differed significantly in proportion to premorbid OC symptoms, but did not differ on measures of psychopathology. Total NES scores and subgroup scores did not differ significantly between people with schizophrenia with or without OC symptoms, but the groups did differ in motor coordination test scores for the left side. Total NES scores correlated significantly with Positive and Negative Symptom Scales negative symptoms scores in those with OC symptoms but not in those without OC symptoms; this correlation was not observed with Yale-Brown Obsessive-Compulsive Scale scores. These findings, if replicated, suggest that OC symptoms in people with schizophrenia are due to differences in frontocerebellar neurological circuitry from those without OC symptoms, with possible neuro-developmental origins.
Schizophrenia and obsessive compulsive disorder (OCD) are major mental disorders that often have their onset in adolescence, follow an episodic or chronic course, and result in considerable distress and disability.1 Studies conducted over the past three decades estimate that 7.8% to 46% of people with schizophrenia have concomitant obsessive-compulsive (OC) symptoms2–5; considerably higher than the rates of 1.2% to 2.4% reported in the general population.6 This could suggest a pathophysiologic link between the two disorders.
OC symptoms may precede (predate) the onset of schizophrenia, may occur in the prodromal phase, or during the course of the illness. People with schizophrenia and comorbid obsessive compulsive symptoms are generally characterized by lower levels of social functioning, longer duration of hospitalization, more neurocognitive impairment, greater severity of negative symptoms, and greater propensity to develop extrapyramidal effects and treatment-resistance than those without OC symptoms.2–5,7–11 Hence, OC symptoms may constitute a distinct cluster separate from psychosis in schizophrenia, thus raising the possibility of a distinct subtype of schizophrenia.
Many symptoms of schizophrenia can be explained by deficits of working memory that are, in turn, caused by dysfunction of dorsolateral prefrontal cortex (PFC).12 The orbitofrontal cortex is a major neuroanatomical area associated with OCD;13 hence, abnormalities in both dorsolateral PFC and orbitofrontal cortex may be involved in people with schizophrenia with comorbid OC symptoms.
Neurological soft signs and negative symptoms in schizophrenia patients may be considered as predisposing factors, as postulated by neurodevelopmental theory. It is speculated that schizophrenic patients with OC symptoms may have a greater propensity to basal ganglia dysfunction than those with schizophrenia-alone, which could result in increased parkinsonian symptoms.14,15 Neuroimaging studies have revealed many common as well as dissimilar areas of structural changes in the brains of people with schizophrenia with and without OCD.16–20 These common abnormalities, detected even early in the course of the disorder, and the overlap in the clinical presentations of these conditions, suggest a common neurodevelopmental origin for these conditions. They provide the basis for the suggestion that people with schizophrenia and comorbid OCD may be a distinct subtype of schizophrenia that some have called “schizo-obsessive disorder”17,21 or “obsessive-compulsive schizophrenia”,8 with further subgroups defined by time of onset of OCD in the course of the disorder and by other clinical or biological parameters.22
This study aimed to compare the pattern of soft neurological signs in drug-free patients with schizophrenia with and without OC symptoms, with a view to elucidating differences in the two subgroups that would point to a neurological substrate for the differing clinical presentations.
METHOD
This study was conducted over 18 months (July 1, 2005 to December 30, 2006) in the outpatient services of the Department of Psychiatry, Christian Medical College, a secondary and tertiary care, teaching, multispecialty medical institution in Vellore, Tamil Nadu, India. The protocol of this study was approved by the institutional review board (research and ethics committees).
Participants
Consecutively-registered outpatients over 18 years of age and diagnosed with schizophrenia (ICD-10) by clinicians were screened for inclusion in the study after we obtained informed consent.
Included were those with a diagnosis of schizophrenia, confirmed by using the International Classification of Diseases (ICD-10) Research Diagnostic Criteria,23 and who were drug-naive or had not used antipsychotic or antidepressant drugs over the previous 3 months. Excluded were with those with a history of neurological illness, treatment with antipsychotics/antidepressants within the previous 3 months, seizure disorder, head injury, mental retardation, and substance abuse (except tobacco use).
Forty-four men and 21 women, with a mean age of 32.4 years (standard deviation [SD]: 9.8) met these criteria. We assessed each participant for the presence of neurological soft signs using the Neurological Evaluation Scale (NES), a structured scale presenting scores of subscales of motor coordination, sensory integration, sequencing of complex motor acts, and “other” signs.14,15,24 The NES has 26 items rated on a scale of 0 to 2 (0: normal; 1: some disruption; 2: major disruption), according to standardized instructions. The Motor Coordination subscale combines the scores for tandem walk, rapid alternation movements, finger/thumb opposition, and the finger-to-nose test. This subscale assesses fronto-cerebellar functions. The Sensory Integration subscale includes audiovisual integration, stereognosis, graphesthesia, extinction, and right/left confusion. These tests evaluate parietal lobe functions. Sequencing of Motor Acts includes the first-ring test, the first-edge-palm test, the Ozeretski test, and rhythm-taping test. This subscale assesses prefrontal functions. The other subscales are the Primitive Reflex subscale that assesses frontal lobe functions and the Hard Neurological Signs subscale that evaluates the CNS, including cranial nerves.15
Classification of participants as Cases (those with OC symptoms) and Controls (those without OC symptoms) was done using the checklist of the Yale-Brown Obsessive Compulsive Scale (Y-BOCS)25 after NES assessments were completed. Participants were also rated for severity on the Y-BOCS. Sociodemographic details, family history, and clinical data were also obtained from interviews with participants and their relatives and from case records. Each participant was also rated using the Positive and Negative Symptom Scale (PANSS),26 and the Clinical Global Impression—Severity scale (CGI).27
Data Analysis
We compared categorical data between Cases and Controls with the chi-square test or Fisher's exact test, and continuous variables with the t-test or Mann-Whitney U test (if data were skewed). Pearson's correlations between scale scores on NES and the PANSS scores as well as NES total subscale scores and Y-BOCS total and subscale scores were computed after testing the data for normality and linearity of distributions. All tests were two-tailed, with a significance level of 0.05. All comparative analyses were conducted using the Statistical Package for Social Sciences, Version 10. (SPSS, Inc.).
RESULTS
Demographic and clinical characteristics of the study participants are shown in Table 1. There was no significant difference in the proportion of men and women or the mean age of the study participants in the two groups. The participants with schizophrenia and OC symptoms had significantly more years of education than those without and were also more likely to be single. They also differed in proportions with premorbid OC symptoms, but were similar in other demographic and clinical characteristics. Scores on the PANSS and for Clinical Global Impression Severity scale did not differ between those with and without OC symptoms.
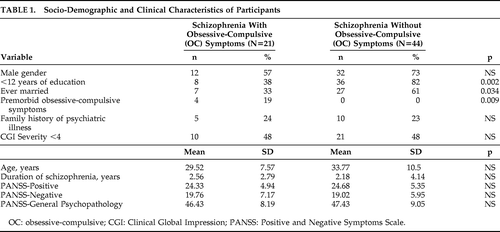 |
Participants without OC symptoms did not score on the Y-BOCS, whereas the mean scores for those with OC symptoms were 19.4 (SD=5.6) for the Total score, 9.0 (SD=4.2) on the Obsessions subscale and 10.4 (SD=3.2) on the Compulsion subscale. Of these 21 people, 5 were rated mild for severity on the Y-BOCS, 15 as moderate, and 1 rated as severe; 14 had multiple symptoms, and the others had single symptoms related to contamination, washing, symmetry, hoarding, or compulsions to utter or ask.
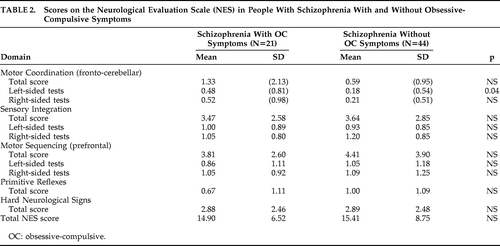
People with schizophrenia with and without OC symptoms did not differ significantly in total scores on the NES or in any of the individual tests (Table 2). However, the two groups differed significantly in left-sided tests on the Motor Coordination subscale of the NES, which assesses fronto-cerebellar functions, although this was not observed in right-sided tests (Table 2). All people with OC symptoms were assessed to have left cerebral dominance as evaluated by NES tests for handedness and footedness, whereas 40 of those without OC symptoms (91%) showed left cerebral dominance on these tests.
 |
There was a significant positive correlation found between the PANSS-Negative symptoms scores and the NES total scores in the subgroup with OC symptoms, but not those without OC symptoms (Table 3).
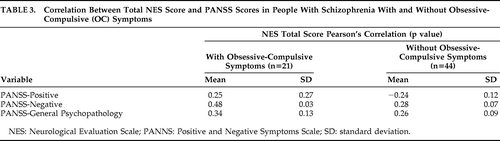 |
Scores on the NES (total or subscales) did not correlate significantly with Y-BOCS total score or subscale scores for obsessions or compulsions, nor were significant correlations observed between Y-BOCS total or subscale scores and PANSS scores for Positive, Negative or General psychopathology scales (data available on request).
DISCUSSION
This cross-sectional study revealed many similarities and some differences in people with schizophrenia with and without OC symptoms. The two groups did not differ at baseline in clinical measures of schizophrenia psychopathology. Those with OC symptoms were better educated and more likely to have been ever married, although there were no differences in their socioeconomic status or duration or severity of the symptoms of schizophrenia. In the group with OC symptoms, 19% reported that OC symptoms antedated the development of the symptoms of schizophrenia, and, since all were antipsychotic drug-naive, the role of drugs in the development of OC symptoms can be ruled out in this sample.
The two groups did not differ significantly in total NES scores, but did differ significantly in performance in the aggregation of tests that form the motor coordination subscale of the NES that assesses fronto-cerebellar functions. This difference was seen only for the tests assessing left-sided fronto-cerebellar functions. This differential performance suggests that people with schizophrenia and OC symptoms differ from those without OC symptoms in fronto-cerebellar circuitry involving the left side.
We had originally hoped to recruit a larger number of people with schizophrenia and OC symptoms, and it is possible that the absence of significant difference in NES total scores or for subtest scores may be due to the relatively small sample (Type II error). However, even a trend toward significant differences was not seen in these scores, so this does not seem the most likely explanation for the findings.
The other possibility that needs consideration in explaining the difference in NES scores of motor coordination of left-sided tests is a type I error or a false-positive finding due to improper methods of assessment. Studies assessing neurological soft signs in people with schizophrenia should take cognizance of the direct and indirect effects of antipsychotic drugs on such evaluations. Motor symptoms due to antipsychotic drugs such as tremor, adventitious overflow, rigidity, and poor balance may be erroneously rated as neurological signs. Higher antipsychotic doses can result in poorer performance for prefrontal signs and better performance in parietal and non-localizing signs.28 Antipsychotic-treated patients are reported to evidence a higher rate of soft neurological signs than antipsychotic-naive people in structured assessments.29 In this study, we included participants who were drug-naive or free of antipsychotic or antidepressant drugs for 3 months in order to eliminate the effects these drugs may have on clinical symptoms or on test performance.
It is possible, however, that the significant difference in motor coordination scores for left-sided tests observed between those with and without OC symptoms is a chance association due to the multiplicity of statistical tests performed, and correcting for this would render the observed statistical difference nonsignificant. Hence, these results need replication in larger samples, although it would be imprudent to ignore the observed differences altogether.
Although there is no published research investigating the unique neurotransmitter involvement in people with schizophrenia and obsessive-compulsive symptoms, serotonin and dopamine have most consistently emerged as the principal neurotransmitters involved in both disorders. In schizophrenia, the dopamine hypothesis has been considered as the fundamental neurochemical model, and this is most strongly supported by successful treatment of the disorder with dopamine-receptor antagonists.30 However, the efficacy of the serotonin—dopamine receptor antagonists in the treatment of schizophrenia additionally supports the importance of the serotonergic system in the pathophysiology of this disorder and may reflect the modulation of dopaminergic pathways by serotonin receptors.31 In OCD, the serotonin hypothesis is well established and is supported by successful treatment of the disorder with serotonin-reuptake inhibitors.32 Since only 40%–60% of patients with OCD exhibit response to monotherapy with selective serotonin-reuptake inhibitors, other neurotransmitter systems may be involved in the pathophysiology of OCD.32 Evidence suggesting the role of dopaminergic system in the pathophysiology of OCD includes the role of dopamine in stereotypic behaviors in animal models, the etiologic role of dopamine in Tourette's disorder, and successful treatment of refractory OCD with dopamine-receptor antagonists.33–35 It is, therefore, reasonable to presume that people with schizophrenia with OC symptoms have abnormalities in both serotonin and dopaminergic systems and hence require drugs that affect both neurotransmitter systems. However, this requires confirmation in large, randomized trials.
Schizophrenia and OCD are both considered as disorders of cortico-striato-thalamo-cortical circuitry,36,37 but the cortical areas involved in these disorders are different. In schizophrenia, the main cortical area involved is the dorsolateral prefrontal cortex, and, in OCD, it is the orbitofrontal cortex.36,37 The NES scale does not specifically assess orbitofrontal cortex functions, and this could explain the lack of significant differences in many of the NES items or the total score. This leaves us with the possibility that the significant difference between the two groups in the motor coordination subscale on the left side, if replicated in larger samples or evaluated via other investigative approaches, is due to differences in fronto-cerebellar circuitry in patients with schizophrenia with obsessive-compulsive symptoms, with possible neurodevelopmental origins.
1. : Disability and family burden in obsessive-compulsive disorder. Can J Psychiatry 1997; 42:919–928Crossref, Medline, Google Scholar
2. : The prognostic significance of obsessive compulsive symptoms in schizophrenia. Am J Psychiatry 1986; 143:437–441Crossref, Medline, Google Scholar
3. : Obsessive compulsive disorder with psychotic features: a phenomenologic analysis. Am J Psychiatry 1986; 143:1527–1533Crossref, Medline, Google Scholar
4. : Obsessive-compulsive disorder in patients with schizophrenia or schizoaffective disorder. Am J Psychiatry 1997; 154:271–273Crossref, Medline, Google Scholar
5. : Prevalence of obsessive-compulsive disorder in schizophrenia and significance of motor symptoms. J Neuropsychiatry Clin Neurosci 2000; 12:16–24Link, Google Scholar
6. : The cross-national epidemiology of obsessive compulsive disorder: The Cross National Collaborative Group. J Clin Psychiatry 1994; 55:5–10Medline, Google Scholar
7. : Clinical and neuropsychological profiles of obsessive-compulsive schizophrenia: a pilot study. J Neuropsychiatry Clin Neurosci 2000; 12:91–94Link, Google Scholar
8. : Olanzapine-sertraline combination in schizophrenia with obsessive-compulsive disorder. J Clin Psychiatry 2003; 64:611Crossref, Medline, Google Scholar
9. : Obsessive and compulsive symptoms in schizophrenia: clinical and neurocognitive correlates. J Nerv Ment Dis 2000; 188:78–83Crossref, Medline, Google Scholar
10. : Association of obsessions and compulsions in schizophrenia with neurocognition and negative symptoms. J Neuropsychiatry Clin Neurosci 2002; 14:449–453Link, Google Scholar
11. : Features of obsessive-compulsive disorder in patients primarily diagnosed with schizophrenia. Psychiatry Clin Neurosci 2003; 57:67–74Crossref, Medline, Google Scholar
12. : Auditory working memory and Wisconsin Card Sorting Test performance in schizophrenia. Arch Gen Psychiatry 1997; 54:159–165Crossref, Medline, Google Scholar
13. : Functional neuroimaging and the neuroanatomy of obsessive–compulsive disorder. Psychiatr Clin North Am 2000; 23:563–586Crossref, Medline, Google Scholar
14. : Significance and meaning of neurological soft signs in schizophrenia. Am J Psychiatry 1988; 145:11–18Crossref, Medline, Google Scholar
15. : Significance and meaning of neurological signs in schizophrenia: two decades later. Schizophr Bull 2005; 4:962–977Crossref, Google Scholar
16. : Brain imaging in childhood and adolescence-onset schizophrenia associated with obsessive compulsive symptoms. Acta Psychiatr Scand 2000; 102:32–37Crossref, Medline, Google Scholar
17. : Neuroimaging commonality between schizophrenia and obsessive compulsive disorder: a putative basis for schizo-obsessive disorder? World J BiolPsychiatry 2003; 4:129–134Crossref, Medline, Google Scholar
18. : Morphometric abnormality of the insula in schizophrenia: a comparison with obsessive-compulsive disorder and normal control using MRI. Schizophr Res 2003; 60:191–198Crossref, Medline, Google Scholar
19. : Ventricular enlargement in schizophrenia spectrum patients with prodromal symptoms of obsessive-compulsive disorder. Psychiatry Res 2000; 99:83–91Crossref, Medline, Google Scholar
20. : Similarity and disparity of obsessive-compulsive disorder and schizophrenia in MR volumetric abnormalities of the hippocampus-amygdala complex. J Neurol Neurosurg Psychiatry 2003; 74:962–964Crossref, Medline, Google Scholar
21. : Is there room for a new diagnostic subtype: the schizo-obsessive subtype? CNS Spectr 1997; 2:49–50Crossref, Google Scholar
22. : Patterns of obsessive-compulsive symptoms and social function in schizophrenia. Psychiatry Res 2004; 125:139–146Crossref, Medline, Google Scholar
23.
24. : The Neurological Evaluation Scale (NES): a structured instrument for the assessment of neurological signs in schizophrenia. Psychiatry Res 1989; 27:335–350Crossref, Medline, Google Scholar
25. : The Yale-Brown Obsessive Compulsive Scale: development, use, and reliability Arch Gen Psychiatry 1989; 46:1006–1011Crossref, Medline, Google Scholar
26. : The Positive and Negative Syndrome Scale (PANSS) for schizophrenia. Schizophr Bulletin 1987; 2:261–176Crossref, Google Scholar
27. : Early Clinical Drug Evaluation (ECDEU) Assessment Manual for Psychopharmacology. Rockville, Md, US Department of Health and Human Services, 1976Google Scholar
28. : Dilapidation in schizophrenia: relationship to negative syndrome and prefrontal impairment. Psychiatry Res 1990; 34:221–222Crossref, Medline, Google Scholar
29. : Neurological soft signs in neuroleptic-naive and neuroleptic-treated schizophrenic patients and in normal comparison subjects. Am J Psychiatry 1995; 152:191–196Crossref, Medline, Google Scholar
30. : Dopamine hypothesis of schizophrenia: making sense of it all. Curr Psychiatry Rep 2007; 9:329–336Crossref, Medline, Google Scholar
31. : Neuropharmacology of second-generation antipsychotic drugs: a validity of the serotonin-dopamine hypothesis. Prog Brain Res 2008; 172:199–212Crossref, Medline, Google Scholar
32. : Psychobiology and psychopharmacology of compulsive spectrum disorders, in Impulsivity and Compulsivity, Edited by Oldham JMHollander ESkodol AE. Washington, DC, American Psychiatric Publishing, 1996, pp 143–161Google Scholar
33. : Considerations of the relevance of ethological animal models for human repetitive behavioral spectrum disorders. Biol Psychiatry 1997; 41:226–229Crossref, Medline, Google Scholar
34. : Tourette syndrome, associated conditions and the complexities of treatment. Brain 2000; 123:425–462Crossref, Medline, Google Scholar
35. : Neuroleptics addition in fluvoxamine refractory obsessive–compulsive disorder. Am J Psychiatry 1990; 147:652–654Crossref, Medline, Google Scholar
36. : Dopamine, schizophrenia, mania, and depression: toward a unified hypothesis of cortico-stratio-pallido-thalamic function. Behav Brain Sci 1997; 10:197–245Crossref, Google Scholar
37. : Cerebral glucose metabolic rates in nondepressed patients with obsessive compulsive disorder. Am J Psychiatry 1988; 145:1560–1563Crossref, Medline, Google Scholar



