Strong Association Between Migraine and Transient Global Amnesia: A National Inpatient Sample Analysis
Abstract
The purpose of this article was to explore sex- and race-specific variables and comorbidities associated with transient global amnesia (TGA) using a nationally representative database. Data were obtained from the Nationwide Inpatient Sample using ICD-9 and procedure codes. Descriptive and survey logistic regression analyses were conducted and adjusted for influence of comorbidities, demographic characteristics, and hospitalization-related factors. Patients with migraines were 5.98 times more likely to also have a diagnosis of TGA compared with patients without migraines. Similarly, patients with TGA were more likely to have hypertension, precerebral disease, and hyperlipidemia. The odds of being diagnosed with TGA was lower among African Americans and Hispanics as well as among patients classified as Asian/Other, compared with Caucasians. TGA was associated with lower hospital charges ($14,242 versus $21,319), shorter hospital stays (mean days: 2.49 [SE=0.036] versus 4.72 [SE=0.025]), and routine hospital discharges (91.4% versus 74.5%). Patients with migraines and patients classified as Caucasian had higher odds of being diagnosed with TGA. All minority populations showed a lower rate of diagnosis that fell short of statistical significance.
Transient global amnesia (TGA) is an uncommon amnestic syndrome characterized by a combined anterograde and retrograde amnesia of sudden onset.1–3 Though generally self-limited with rare recurrences, this striking presentation has spurred efforts to link TGA to more consequential and treatable conditions. Previous studies on TGA have been inconsistent regarding sex prevalence.4–6 The average age of onset has ranged between 50–80 years, with an incidence of approximately 5–11 per 100,000 per year.7,8
Despite the general acceptance of TGA as a clinical entity, its etiology has yet to be determined.9 Past studies have attempted to correlate TGA to vascular risk factors, migraine, and epilepsy, with variable results. Because of the implicated loss of declarative memory, ischemia in bilateral hippocampi has been suggested as a possible cause. High-resolution brain MRI of TGA patients suggests diffusion restriction in the hippocampi.10–13 More recent studies have focused on the discovery that a large percentage of TGA patients have jugular venous insufficiency.14,15 Migraines and epilepsy have also been implicated as potential correlates of TGA.16 Numerous associations with other possible triggers have been reported2 yet, none of these correlations have been validated in a nationwide database.
In an effort to better characterize TGA, we have examined the Nationwide Inpatient Sample, which is uniquely suited to evaluate the incidence of rare diseases, such as TGA.
Methods
Data were obtained from the Nationwide Inpatient Sample (NIS) for the years 1999–2008. The NIS is a database maintained as a part of the Healthcare Cost and Utilization Project by the Agency for Healthcare Research and Quality. It represents 20% of U.S. community hospitals, comprising 5–8 million inpatient records per year. The data were collected in a manner whereby we could perform weighted analysis representing the total population of the United States. Information collected by the NIS includes basic demographic data, ICD-9 diagnoses, procedural coding, financial data, and discharge data, including the length of stay and total cost of hospitalization. The sampling strategy selects hospitals nationwide from the State Inpatient Database according to defined strata based on ownership, bed size, teaching status, urban or rural location, and region. All discharges from sampled hospitals for the calendar year are then selected for inclusion into NIS. To allow extrapolation for national estimates, both hospital and discharge weights are provided. Detailed information on the design of the NIS is available online (http://www.hcup-us.ahrq.gov). The unit of analysis was the discharge or encounter rather than the individual. We assumed that each discharge or encounter appeared only once per patient. A unique hospital identifier allows linkage of discharge data to an NIS data set with hospital characteristics.
To analyze TGA hospitalizations, we identified all hospital discharges for which an ICD-9-CM code of 437.7 (transient global amnesia) was listed. Searching the literature, we were able to identify eight comorbidities previously associated with TGA: epilepsy (ICD-9 code 345), migraine headache (ICD-9 code 346), precerebral disease (atherosclerosis) (CCS code 110), stroke (CCS code 109), atrial fibrillation (CCS code 106), hypertension (CCS code 98), hyperlipidemia (CCS code 53), and diabetes mellitus (CCS codes 49, 50). Missing data were not included in the analyses. The variables included were a binary outcome (TGA), four main binary predictors (precerebral disease, migraine headache, epilepsy, and stroke), and three potential binary confounders (atrial fibrillation, hypertension, and diabetes mellitus) as well as other secondary variables (sex [binary], race [nominal], age [continuous], hyperlipidemia [binary], and hospital classification [region, location, and teaching status; binary]). From the original hospital discharge records (from 1999 to 2008), we were able to identify 58,191,569 patients without TGA and 9,723 patients with TGA. A causal diagram that graphically represents assumed associations and compounding relationships among selected variables and TGA is shown in Figure 1.
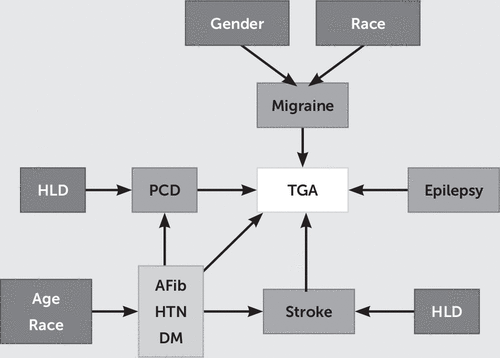
FIGURE 1. Causal Diagram of Assumed Associations and Compounding Relationships Among Selected Variables and Transient Global Amnesia (TGA)a
a AFib=atrial fibrillation, DM=diabetes mellitus, HLD=hyperlipidemia, HTN=hypertension, PCD=precerebral disease.
Statistical Analysis
Using chi-square tests, we explored the differences between TGA and non-TGA hospitalizations. Differences in the distribution of the study variables were tested by Chi-square test for independence. Independent predictors and covariates examined included: a) socio-demographic characteristics b) clinical factors and c) selected two-way interactions. Multiple logistic regression analyses were performed to explore the relationship between TGA status and selected predictors. Since the NIS database uses a stratified, single-stage random cluster sampling methodology, we used a survey logistic regression to account for discharge weights and effects of stratification and clustering. We combined NIS data from 1999 to 2008 and ran the final model with all demographic variables and comorbidities. Adjustments in the multivariable models were made for socio-demographic factors, including age, sex, and race, clinical comorbidities and hospital regions. Age in years was centered at 65, and a squared term was added to the model to determine if TGA has a quadratic relationship with age. We ran two models, one with race-comorbidity interaction and the other without the interaction, with the aim being to ascertain the average effect for race.
Only patient records with no missing data for selected model variables were included for analyses.
NIS sampling and design parameters were employed during this analysis. Demographic statistics were processed with SPSS, version 20, with the complex samples module. Logistic regressions were conducted with Statistical Analysis System, 9.3. This study was deemed exempt by the institutional review board at Loma Linda University.
Results
From the original hospital discharge records for all potentially eligible patients, between 1999 and 2008, the study included 58,191,569 unweighted patients without TGA, which was equivalent to 283,228,304 weighted patients without TGA, and 9,723 unweighted patients with TGA, equivalent to 47,432 weighted patients with TGA (Table 1). Any hospital discharge for which any chosen variables were missing was excluded from the analyses.
| Ordered Value | Transient Global Amnesia (N) | Total Frequency (Unweighted) | Total Weight |
|---|---|---|---|
| 1 | 0 | 58,191,569 | 283,228,304 |
| 2 | 1 | 9,723 | 47,432 |
TABLE 1. Response Profile of the Study Participants (1999–2008)
The 47,432 patients in the TGA group differed from the patients in the non-TGA group in age (TGA group: mean age, 64.93 years [SD=0.18], non-TGA group: mean age, 47.74 years [SD=0.25]). Most patients in the TGA group were ≥65 years old (55.04%). Overall, Caucasians outnumbered other ethnicities in both groups, with more Caucasians in the TGA group (86.28% versus 67.86%). Other racial and ethnic populations were represented at noticeably lower proportions in both groups (TGA group: African American, 4.43%, Hispanic, 5.30%, and Asian/Other, 4.0%; non-TGA group: African American, 13.6%, Hispanic, 12.42%, and Asian/Other, 6.12%). There was a higher proportion of women in both groups, with more women in the non-TGA group (51.99% versus 58.8%).
Total in-hospital charges were less for the TGA group compared with the non-TGA group ($14,242 versus $21,319), but the cost trend increased over the 10-year period. Overall, TGA hospitalization was associated with a reduced number of procedures (TGA group: mean, 0.69 [SD=0.036]; non-TGA group: mean, 2 [SD=0.016]), yet patients in the TGA group were seven times more likely to have brain MRI (7.66% versus 0.44%) and head CT scans (7.31% versus 0.87%). Similarly, more lumbar punctures were performed for patients with TGA (3.23% versus 0.91%). Length of hospital stay was shorter for patients in the TGA group, with a mean of 2.49 days (SD=0.036), compared with 4.72 days (SD=0.25) for patients without TGA. More patients with TGA were evaluated at urban hospital settings (urban setting, 90.16%; rural setting, 9.84%) and in the Southern region (37.45%).
We were able to identify differences in comorbidities using Charlson’s comorbidity index.17 Hypertension was twice as prevalent among patients with TGA (51.25% versus 27.62%). Valvular disease was detected more frequently in the TGA group (8.26% versus 5.0%) as well as anxiety (5.98% versus 2.99%) and depression (8.88% versus 6.81%). Overall, TGA hospitalization was associated with lower mortality (0.1% versus 2.24%), routine discharges from the hospital (91.46% versus 74.49%), and less frequent discharge to a facility or home health care (6.82% versus 20.06%).
Significant differences were observed in the distribution of medical comorbidities. Of particular importance was the high prevalence of migraine headache, hypertension, precerebral disease, and hyperlipidemia among patients with TGA, when compared with non-TGA patients. Migraine was almost five times more prevalent in the TGA group compared with the non-TGA group (4.94% versus 0.76%), and hypertension was twice as high (51.25% versus 27.62%), precerebral disease almost four times as high (3.94% versus 1.0%), and hyperlipidemia nearly three times more prevalent (33.66% versus 12.55%). However, this pattern was not seen for epilepsy (0.79% versus 0.64%) (Tables 2 and 3).
| Without TGAa | With TGAb | |||
|---|---|---|---|---|
| Characteristic | % | p | % | p |
| Age (years) | <0.0001 | <0.0001 | ||
| <45 years | 43.19 | 6.04 | ||
| 45–54 years | 10.65 | 11.15 | ||
| 55–64 years | 11.1 | 27.77 | ||
| ≥65 years | 35.06 | 55.04 | ||
| Race/ethnicity | <0.0001 | <0.0001 | ||
| Caucasian | 67.86 | 86.28 | ||
| African American | 13.6 | 4.43 | ||
| Hispanic | 12.42 | 5.3 | ||
| Asian/Otherc | 6.12 | 4 | ||
| Gender | <0.0001 | <0.0001 | ||
| Male | 41.22 | 48.01 | ||
| Female | 58.78 | 51.99 | ||
| Primary payer (%) | <0.0001 | <0.0001 | ||
| Medicare | 37.42 | 50.64 | ||
| Medicaid | 18.51 | 2.24 | ||
| Private (including HMO) | 35.79 | 42.26 | ||
| Otherd | 8.27 | 4.87 | ||
| Comorbid medical conditions | <0.0001e | <0.0001e | ||
| Cancer | 13.39 | 10.83 | ||
| Cerebrovascular disease (stroke) | 1.9 | 2.88 | ||
| Chronic pulmonary disease | 10.48 | 4.61 | ||
| Congestive heart failure | 10.65 | 3.08 | ||
| Connective tissue disease | 5.22 | 0.148 | 5.56 | 0.148 |
| Diabetes with complications | 4.82 | 2.3 | ||
| Diabetes without complications | 11.79 | 9.63 | ||
| Hypertension | 27.62 | 51.25 | ||
| Metastatic carcinoma | 2.58 | 0.4 | ||
| Mild liver disease | 1.04 | 0.47 | ||
| Moderate to severe liver disease | 0.5 | 0.1 | ||
| Paraplegia/hemiplegia | 1.23 | 0.47 | ||
| Peptic ulcer disease | 1.23 | 0.32 | 1.12 | 0.32 |
| Valvular heart disease | 5 | 8.26 | ||
| HIV/AIDS | 0.7 | 0.11 | ||
| Urinary tract infection | 6.89 | 3.61 | ||
| Delirium | ||||
| Delirium with dementia | 0.04 | 0.129 | 0.08 | 0.129 |
| Nondementia, nondrug delirium | 0.29 | <0.0001 | 2.13 | <0.0001 |
| Drug-induced delirium | 0.18 | 0.909 | 0.19 | 0.909 |
| Mental health comorbidity | <0.0001 | <0.0001 | ||
| Depression | 6.81 | 8.88 | ||
| Anxiety | 2.99 | 5.98 | ||
| Primary in-hospital procedures (CPT codes) | <0.0001 | <0.0001 | ||
| MRI brain | 0.44 | 7.66 | ||
| CT brain | 0.87 | 7.31 | ||
| Lumbar puncture | 0.91 | 3.23 | ||
| Number of procedures | <0.0001 | <0.0001 | ||
| 0–1 | 63.38 | 83.24 | ||
| 2–3 | 24.48 | 11.32 | ||
| 4–5 | 7.26 | 3.61 | ||
| ≥6 | 4.88 | 1.83 | ||
| Hospital type | <0.0001 | <0.0001 | ||
| Rural | 12.16 | 9.84 | ||
| Urban nonteaching | 43.46 | 51.02 | ||
| Urban teaching | 44.38 | 39.14 | ||
| Mortality status | <0.0001 | <0.0001 | ||
| Died in hospital | 2.24 | 0.1 | ||
| Discharge disposition | <0.0001 | <0.0001 | ||
| Routine | 74.49 | 91.46 | ||
| Short-term hospital | 2.3 | 0.85 | ||
| Another type of facility/home health care | 20.06 | 6.82 | ||
| Against medical advice | 0.91 | 0.77 | ||
| Died | 2.24 | 0.1 | ||
| Hospital region | <0.0001 | <0.0001 | ||
| Northeast | 25.28 | 31.86 | ||
| Midwest | 14.69 | 15.26 | ||
| South | 40.34 | 37.45 | ||
| West | 19.69 | 15.43 | ||
| Hospital location | <0.0001 | <0.0001 | ||
| Rural | 12.16 | 9.84 | ||
| Urban | 87.84 | 90.16 | ||
| Hospital teaching status | <0.0001 | <0.0001 | ||
| Nonteaching | 54.86 | 60.18 | ||
| Teaching | 45.14 | 39.82 | ||
| Comorbidities in the model | <0.0001f | <0.0001f | ||
| Epilepsy | 0.64 | 0.09 | 0.79 | 0.09 |
| Migraine headache | 0.76 | 4.94 | ||
| Hypertension | 27.62 | 0.001 | 51.25 | 0.001 |
| Atrial fibrillation | 12.21 | 0.001 | 13.41 | 0.001 |
| Stroke | 1.9 | 2.88 | ||
| Precerebral disease | 1 | 3.94 | ||
| Hyperlipidemia | 12.55 | 33.66 | ||
| Diabetes mellitus | 16.59 | 11.91 | ||
TABLE 2. Demographic Characteristics of Participants With and Without Transient Global Amnesia (TGA)
| Without TGAa | With TGAb | |||||||
|---|---|---|---|---|---|---|---|---|
| Variable | Mean | SD | SE | p | Mean | SD | SE | p |
| Age (years) | 47.74 | 0.25 | <0.0001 | 64.93 | 0.18 | <0.0001 | ||
| Total in-hospital charge | 21.319 | 32.97 | 14.242 | 292.77 | ||||
| Length of hospital stay | 4.72 | 0.025 | 2.49 | 0.036 | ||||
| Number of procedures | 2 | 0.016 | 0.69 | 0.036 | ||||
TABLE 3. Hospital Data Among Participants With and Without Transient Global Amnesia (TGA)
Using survey logistic analysis, we found that the odds of having TGA was higher for men (odds ratio=1.13, 95% confidence interval [CI]=1.08–1.18) (Table 4). The squared-age term was significant, and thus it was retained in the model. In the adjusted regression model, patients with migraine headache had a sixfold greater odds of TGA compared with patients in the non-TGA group (odds ratio=5.98, 95% CI=5.42–6.60), and migraine headache was followed by precerebral disease (odds ratio=1.92, 95% CI=1.71–2.14), hyperlipidemia (odds ratio=1.88, 95% CI=1.79–1.97), and hypertension (odds ratio=1.35, 95% CI=1.29–1.41). The adjusted odds ratio for epilepsy was 1.16 but was not statically significant (95% CI=0.92–1.45.). TGA was not associated with stroke (odds ratio=1.00, 95% CI=0.89–1.14). Patients with atrial fibrillation and diabetes had lower odds of TGA (atrial fibrillation: odds ratio=0.69, 95% CI=0.64–0.73; diabetes: odds ratio=0.36, 95% CI=0.33–0.39).
| Variables | Adjusted Odds Ratio | 95% CI | p |
|---|---|---|---|
| Male (reference: female) | 1.13 | 1.08–1.18 | <0.0001 |
| Age (years), centered at age 65 | 1.00 | 0.38 | |
| Age (years), squared | 1.00 | <0.0001 | |
| Comorbidities | |||
| Epilepsy | 1.16 | 0.92–1.45 | 0.22 |
| Migraine headache | 5.98 | 5.42–6.60 | <0.0001 |
| Hypertension | 1.35 | 1.29–1.41 | <0.0001 |
| Atrial fibrillation | 0.69 | 0.64–0.73 | <0.0001 |
| Stroke | 1.00 | 0.89–1.14 | 0.95 |
| Precerebral diseasea | a | ||
| Hyperlipidemiaa | a | ||
| Diabetesa | a | ||
| Race (without interaction)b | |||
| African American | 0.363 | 0.23–1.34 | <0.0001 |
| Hispanic | 0.697 | 0.09–0.56 | <0.0001 |
| Asian/Other | 0.819 | 0.12–1.10 | 0.001 |
TABLE 4. Adjusted Odds Ratios of Transient Global Amnesia Prevalence for Selected Variables With Race-Comorbidity Interactions and for Race Without Race-Comorbidity Interactions
Compared with Caucasians, the odds of having TGA was lower among African Americans (odds ratio=0.36, 95% CI=0.23–1.34) and Hispanics (odds ratio=0.69, 95% CI=0.09–0.56) as well as among individuals classified as Asians/Other (odds=0.82, 95% CI=0.12–1.10) (Table 4).
African Americans with precerebral disease had almost a fourfold increased odds of having TGA (odds ratio=3.72, 95% CI=2.15–6.42), and Hispanics with precerebral disease had almost three times the odds (odds ratio=2.72, 95% CI=1.40–5.28), while Caucasians with the disease had almost twice the odds (odds ratio=1.92, 95% CI=1.71–2.14). For patients with hyperlipidemia, the odds of TGA was almost twice as high among Caucasians (odds ratio=1.88, 95% CI=1.79–1.97) and Hispanics (odds ratio=2.11, 95% CI=1.70–2.61), but a statistically significant association was not seen among African Americans (odds ratio=1.24, 95% CI=0.96–1.58). Diabetes seemed to be protective for TGA in all races but more for Hispanics, Asians/Other, and Caucasians than for African Americans (Caucasians, odds ratio=0.36, 95% CI=0.33–0.39; African Americans, odds ratio=0.56, 95% CI=0.43–0.71; Hispanics, odds ratio=0.31, 95% CI=0.24–0.340; Asians/Other, odds ratio=0.36, 95%=CI 0.27–0.47) (Table 5).
| Race | Adjusted Odds Ratioa | 95% CI | p |
|---|---|---|---|
| Caucasian | |||
| With or without precerebral disease | 1.916 | 1.713–2.1 | 0.000 |
| With or without hyperlipidemia | 1.880 | 1.790–1.97 | 0.000 |
| With or without diabetes mellitus | 0.358 | 0.33–0.39 | 0.000 |
| African American | |||
| With or without precerebral disease | 3.717 | 2.15–6.42 | 0.000 |
| With or without hyperlipidemia | 1.230 | 0.96–1.58 | 0.106 |
| With or without diabetes mellitus | 0.555 | 0.43–0.71 | 0.000 |
| Hispanic | |||
| With or without precerebral disease | 2.719 | 1.40 –5.28 | 0.003 |
| With or without hyperlipidemia | 2.112 | 1.71–2.61 | 0.000 |
| With or without diabetes mellitus | 0.308 | 0.24–0.40 | 0.000 |
| Asian/Other | |||
| With or without precerebral disease | 1.810 | 0.77–4.22 | 0.169 |
| With or without hyperlipidemia | 1.828 | 1.49–2.23 | 0.000 |
| With or without diabetes mellitus | 0.358 | 0.27–0.47 | 0.000 |
TABLE 5. Race- and Comorbidity-Specific Adjusted Odds Ratios
Discussion
Consistent with previous data, the average age of patients with TGA was approximately 65 years,4 and our adjusted regression model revealed that men had modestly higher odds of having TGA. Our most significant finding was that those with a diagnosis of migraine had six time greater odds of having TGA. Migraine and TGA, as historically noted, share a number of characteristics, including paroxysmal presentation and associated triggers. It has been proposed that cortical depression in the hippocampal region is the common denominator in both disease processes.18–21 Several studies have gone as far as to refer to TGA as a migrainous aura.20,22 Despite the proposed shared pathophysiologic mechanisms, there are several unavoidable differences, such as TGA being self-limiting and most often affecting elderly men.
Because there was significant interaction between comorbidities and race, the effect of comorbidities was not consistent when considering racial differences. We found that for African Americans, diagnosis of precerebral disease increased the odds of having TGA by 3.7 times, and the odds was increased by 2.7 times for Hispanics and almost two times for Caucasians. Similarly, the odds of hyperlipidemia were elevated among Caucasians, Hispanics, and Asians but not African Americans. Contrary to previous studies, we did not find a statistically significant association between epilepsy and TGA. The low recurrence and self-limiting nature of TGA make it highly unlikely to be related to epilepsy. TGA patients typically have normal EEG recordings during and after a genuine episode of TGA.23–25 At times, the postictal amnesia associated with partial complex seizures may last longer than usual. However, such events are uncommon and are associated with ictal manifestations, and therefore, must be considered as a distinct diagnosis of epilepsy, rather than TGA. The finding of elevated odds of TGA with hyperlipidemia, precerebral disease, and hypertension as opposed to Seizures speaks to a more vascular possible etiology to TGA than an electrophysiological one.
Conflicting reports exist with regards to thromboembolic processes and their correlation with TGA symptomatology. Multiple neuroimaging studies have noted the presence of ischemic changes in the hippocampi during different stages of TGA.26 In a case-control study, Lauria et al.5 describe decreased frequency of patent foramen ovale in TGA patients compared with TIA patients, and concluded that cryptogenic thromboembolic processes were unlikely to be responsible for TGA. Similar to previous studies, we did not find a correlation between TGA and stroke. This is an important finding, as it suggests that patients with a clear diagnosis of TGA do not require inpatient admission and extensive diagnostic workup.
We found that the cost of TGA hospitalization has increased disproportionately over the last ten years. One could posit that increase in the use of sophisticated and costly neuroimaging modalities is largely responsible for the climbing costs of TGA care, and suggest that a better understanding of the disease process and definitive diagnostic criteria might obviate the need for such extensive workup.
We also found geographic differences in TGA diagnosis. Patients in hospitals located in the Southern region were more likely to be diagnosed with TGA, with relatively fewer diagnoses in the Midwestern and Northeastern regions, and least frequently in Western region of the country. There is indication in the Healthcare Cost and Utilization Project database that the Southern territory has a relatively larger number of hospitals, and more academic institutions per capita than the West. The distribution of neurology specialists and neurology training programs is similarly skewed. Large centers in the East Coast are more likely to be located in proximity to larger population. Due to stratified and single-stage cluster sampling of this database, our findings can be generalized for the greater U.S. population.
Limitations
Although the numerical power of our study is unprecedented, it is likely that this number is still an underestimation of the true prevalence of the disease, due to a variety of factors, including misdiagnosis, noninpatient evaluations, and regional differences. Because the diagnosis of TGA is self-limited and typically lasts only 4–6 hours, we must consider the possibility that patients with TGA may never present to the hospital, let alone be admitted.
It is very difficult to account for the inverse relationship between TGA and such comorbidities as atrial fibrillation and diabetes, other than the possibility of physician bias, where those physicians, in an emergency room setting, are more likely to diagnose patients with symptoms consistent for TGA as having stroke, or transient ischemic attach if they also have a comorbidity of diabetes or atrial fibrillation. This also may speak to the limitations of this database for diagnosis of TGA, but the fact that this category of bias would bias toward the null, would only strengthen the possible degree of relationships that we were able to observe.
The NIS, by virtue of its focus on the inpatient population, is unable to account for the possibility of diagnosis and conservative management in the outpatient clinic and emergency room settings. Additionally, the inherent weakness of NIS is that it relies upon unverified physician diagnosis. We also acknowledge that diagnostic criteria have varied in the past and are not consistently implemented. Despite these limitations, we believe that our results provide great insight into this enigmatic disease as well as direction for future research.
Conclusions
We demonstrated that Caucasians and men had greater odds of developing TGA, and patients with migraines had almost six times higher odds of having TGA. Additionally, we showed a strong possible vascular etiological cause for TGA, which should be further explored.
Despite the progressive decline in the length of hospital stay, the cost of management has grown exponentially. Our findings from this nationally representative sample will help us to better plan patient care and future research related to this enigmatic disease.
1 : Transient global amnesia: an often misconceived disorder. MMW Fortschr Med 2008; 150(Suppl 2):29–31Medline, Google Scholar
2 : Transient global amnesia: functional anatomy and clinical implications. Lancet Neurol 2010; 9:205–214Crossref, Medline, Google Scholar
3 : The transience and nature of cognitive impairments in transient global amnesia: a meta-analysis. J Clin Exp Neuropsychol 2009; 31:8–19Crossref, Medline, Google Scholar
4 : Syndromes of transient amnesia: towards a classification: a study of 153 cases. J Neurol Neurosurg Psychiatry 1990; 53:834–843Crossref, Medline, Google Scholar
5 : Transient global amnesia and transient ischemic attack: a community-based case-control study. Acta Neurol Scand 1998; 97:381–385Crossref, Medline, Google Scholar
6 : Transient global amnesia: a case control study. Brain 1992; 115:261–270Crossref, Medline, Google Scholar
7 : The aetiology of transient global amnesia: a case-control study of 114 cases with prospective follow-up. Brain 1990; 113:639–657Crossref, Medline, Google Scholar
8 : Incidence of transient global amnesia in the Belluno province, Italy: 1985 through 1995: results of a community-based study. Acta Neurol Scand 1997; 95:303–310Crossref, Medline, Google Scholar
9 : Transient global amnesia. Neurol Clin 2011; 29:1045–1054Crossref, Medline, Google Scholar
10 : Transient global amnesia: diffusion-weighted imaging lesions and cerebrovascular disease. Stroke 2008; 39:2219–2225Crossref, Medline, Google Scholar
11 : Focal MR spectroscopy of hippocampal CA-1 lesions in transient global amnesia. Neurology 2008; 70:1030–1035Crossref, Medline, Google Scholar
12 : Transient global amnesia: hippocampal magnetic resonance imaging abnormalities. Funct Neurol 2008; 23:149–152Medline, Google Scholar
13 : Transient global amnesia and brain lesions: new hints into clinical criteria. Eur J Neurol 2008; 15:981–984Crossref, Medline, Google Scholar
14 : Incompetence of internal jugular vein valve in patients with transient global amnesia. Vojnosanitetski pregled. 2011;68(1):35–40Crossref, Medline, Google Scholar
15 : Disturbance of venous flow patterns in patients with transient global amnesia. Lancet 2000; 356:1982–1984Crossref, Medline, Google Scholar
16 : Transient global amnesia, epilepsy, and migraine: diagnostic comparisons and distinctions. Mil Med 1989; 154:424–427Crossref, Medline, Google Scholar
17 : A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987; 40:373–383Crossref, Medline, Google Scholar
18 : Selective affection of hippocampal CA-1 neurons in patients with transient global amnesia without long-term sequelae. Brain 2006; 129:2874–2884Crossref, Medline, Google Scholar
19 : The transient global amnesia-migraine connection. Arch Neurol 1984; 41:75–79Crossref, Medline, Google Scholar
20 : Leao’s spreading depression in the hippocampus explains transient global amnesia: a hypothesis. Acta Neurol Scand 1986; 73:219–220Crossref, Medline, Google Scholar
21 : Transient global amnesia: a genetic disorder? Eur Neurol 2010; 63:186–187Crossref, Medline, Google Scholar
22 : Pathophysiology of the migraine aura: the spreading depression theory. Brain 1994; 117:199–210Crossref, Medline, Google Scholar
23 : Transient global amnesia and epilepsy. Electroencephalographic distinction. Arch Neurol 1987; 44:629–633Crossref, Medline, Google Scholar
24 : EEG studies in the syndrome of isolated episodes of confusion with amnesia “transient global amnesia.” J Neurol Neurosurg Psychiatry 1966; 29:472–474Crossref, Medline, Google Scholar
25 : Transient global amnesia: the electroencephalogram at onset. Ann Neurol 1987; 22:771–772Crossref, Medline, Google Scholar
26 : Detection of delayed focal MR changes in the lateral hippocampus in transient global amnesia. Neurology 2004; 62:2165–2170Crossref, Medline, Google Scholar



