The Expanding Role of Imaging in Prion Disease
The transmissible spongiform encephalopathies (TSEs) are invariably fatal rare neurodegenerative diseases. The most accepted theory is that TSEs are caused by abnormal forms of a naturally occurring glycoprotein. These proteinaceous infectious agents (or prions) are altered forms (PrPTSE) of the normal cellular prion protein (PrPC).3,4 Both isoforms have the same primary sequence, but their three-dimensional conformations are quite different (PrPC has ∼3% β-sheets, whereas PrPTSE has ∼43%), as are their biochemical and biophysical properties.3,5 In particular, the PrPTSE form is extremely resistant to inactivation by methods ranging from chemical (such as formalin) to heat (including autoclaving) to radiation (both ionizing and ultraviolet).3 According to the prion theory, infection spreads as a result of PrPTSE-induced unfolding and refolding of the normal PrPC into the infectious form. Chaperone proteins may also play a role. There are a number of different PrPTSE strains with distinctly different clinical and pathological phenotypes. This has been difficult to reconcile with the protein-only prion hypothesis, but may relate to differences in three-dimensional conformation.3,5,6 The sporadic form of CJD (sCJD), for instance, has at least six major variations based on a combination of different forms of the protease-resistant core of PrPTSE (type 1 and type 2) and the genotype at codon 129 of the PrP gene (homozygous or heterozygous for methionine or valine).7,8 These strains vary along many dimensions, including average age at onset, disease duration, constellation of clinical symptoms, neuropathological characteristics, presence of EEG abnormality, diagnostic imaging appearance, and CSF findings.7,9
The TSEs are caused by spontaneous conversion of PrPC to PrPTSE (sporadic form), by inherited germ-line mutation (genetic or familial form), and by inoculation (iatrogenic or dietary forms). Sporadic cases (unknown etiology) are the most common (>85%), followed by familial cases (12%–14%); iatrogenic cases are least common.5 Many different mutations in the PrP gene (PRNP) have been identified. The predominant forms in humans are Creutzfeldt-Jakob disease (CJD), Gerstmann-Straüssler-Scheinker syndrome (GSS), fatal insomnia (FI), kuru, and new variant Creutzfeldt-Jacob disease (vCJD). Familial GSS is associated with codon 102, 105, 117, 198, and 217 mutations.10 The familial form of CJD (fCJD) is associated with codon 53, 178, and 200 mutations. The familial form of FI (fFI) is also associated with mutation at codon 178. In addition, the genotype at codon 129 of the PrP gene (homozygous or heterozygous for methionine or valine) drastically affects disease course.11 Individuals with the codon 178 mutation who are homozygous for valine express fCJD, while those that are heterozygous or homozygous for methionine express fFI.10,12 Patients heterozygous at codon 129 have a slower disease progression (21±15 months) than those who are homozygous for methionine (12±4 months).11,1314
There is much debate in the medical literature about the existence of some variants of the disease, the true epidemiology, and transmissibility of the infectious agent from one species to another.15–17 Scrapie has been recognized in sheep from the 1700s onward.18 A few cases of what would now be considered human TSE were reported in the 1920s and 1930s. The discovery in the 1950s of kuru, with its clinical similarities to scrapie, resulted in a wider recognition of the human forms of TSE. The emergence of bovine spongiform encephalopathy (BSE) in cattle in Great Britain in the 1980s heightened awareness of the TSEs and created concern about the potential for transmission to humans. The identification of vCJD in humans has spurred a resurgence of both concern and research. A PubMed search of TSEs in humans produced 3,636 articles in English; 464 of these were written since January 2001. Currently, many nations have “surveillance units” to follow any suspected cases and to monitor for potential outbreaks.
The worldwide incidence of these rare diseases is estimated as 1 case per million population. Iatrogenic cases have been described in patients receiving cadaver pituitary growth hormone extracts (cadaver extracts are now illegal in most countries, replaced by synthetic growth hormone), insertion of electroencephalographic (EEG) depth electrodes, and corneal and dura mater transplants from persons later found to have infection.19 Sporadic cases generally occur in the elderly (sixth to seventh decade); the transplant and new variant cases occur in younger adults. Incubation time can be years, with death rapidly occurring once symptoms begin (generally within 9 months of symptom onset).9,20
Although prion diseases are rare, the differential diagnosis includes common conditions such as Alzheimer's disease and vascular dementia. They serve as examples of the importance of newer imaging techniques and diagnostic testing in clinical neuropsychiatry. Additionally, the public health implications of a missed identification of a patient with prion disease are troublesome.
CLINICAL FEATURES
As noted earlier, prion diseases are fatal encephalopathies. The clinical features vary according to the individual diagnosis, and many patients exhibit variations of a central theme according to the form of protein mutation. However, common clinical characteristics exist. An excellent clinical review can be found in the monograph series by Knight and Collins.21 The following brief overview of this summary can be helpful as an initial framework for study or patient screening in suspected cases.
A diagnostic progression has been developed for CJD in which the diagnosis is definite (confirmed by biopsy or autopsy), probable, or possible. The probables are approximately 95% certain for sporadic CJD, with clinical and EEG or cerebrospinal fluid (CSF) markers. The possibles have less specific clinical and diagnostic findings. The hallmark features of sCJD include a rapidly progressive dementia with visual/cerebellar features (including ataxia), myoclonus, pyramidal/extrapyramidal features, and progression to akinetic mutism. Other vague markers include initial depression, insomnia, or headaches. The variant form has a slower progression, and is reported to have unexplained pain and more psychiatric findings (including delusions, hallucinations, agitation, anxiety, and depression) with less akinetic mutism. Positive EEG and CSF findings are less likely.21
GSS symptoms include a pancerebellar dysfunction with dementia, akinetic mutism, and occasionally aggression or emotional lability. Classic EEG findings are present in some cases. FI has a progressive loss of the normal sleep–wake cycle with eventual inability to attain normal sleep. The patients have hallucinations, parasomnias, and severe sympathetic overactivity (including salivation, rhinorrhea, hyperthermia, tachycardia, and hypertension). Hormonal abnormalities include increased serum cortisol and decreased melatonin or thyrotropin. Abnormal cycles of other hormones including prolactin, and the gonadonal hormones occur. Eventual cerebellar dysfunction, myoclonus, altered respirations, dementia, coma, and death ensue. Polysomnographic testing demonstrates lack of stage 2 and non-REM sleep and only brief REM sleep episodes (often associated with oneiric behavior). Classic EEG and CSF findings are not present. Kuru has a prominent pancerebellar dysfunction with initial ataxia and eventual loss of all voluntary muscle control, gross tremors, movements similar to shivering, euphoria, and late-onset cognitive decline and emotional lability.21
DIAGNOSTIC TESTING
To date, limited medical testing is available to support a proposed diagnosis of TSE.7 Periodic sharp and slow wave complexes (PSWCs) are frequently found on EEGs but may not be present in the early or late stages of the disease. They are not pathognomonic, as they have been reported in many conditions including Alzheimer's disease. Serial EEGs are recommended in order to have the best chance for demonstration of PSWCs. Test sensitivity is 30% to 100%, and eventual identification of the complexes occurs in approximately two-thirds of sCJD cases. Variant CJD and FI patients do not generally have PSWCs.21,22
Routine CSF studies are normal in prion diseases. The TSEs are unusual in that the immune system is not activated by the infection.3 Two CSF neuronal proteins (neuron-specific enolase [NSE] and S-100) have been found commonly in patients with prion diseases. However, recent comparisons to another marker, 14-3-3, have established it to be more reliable. 14-3-3 has a sensitivity of 89% to 93% and a specificity of 93% to 100% for sCJD.22,23 Several conditions may give false positives, including herpes encephalitis, multiple sclerosis, and cerebral infarction, but they can be eliminated by other diagnostic tests. Although 14-3-3 results are helpful, particularly in cases that do not show all of the classic clinical symptoms, definitive confirmation of the diagnosis requires neuropathological demonstration of the characteristic spongiform changes (Figure 1).
Computed tomography (CT) is used as screening tool to rule out other illnesses with similar initial presentations. Magnetic resonance (MR) imaging is becoming increasingly useful in the diagnosis of TSEs. A recent large study assessed the presence of high signal intensity within the basal ganglia on T2-weighted MR imaging in patients with sCJD and those with non-CJD-related dementia.24 This appearance was found in 67% (109/162) of patients with sCJD but only 7% (4/58) of patients with dementia due to another cause. A similar frequency was found in a previous review of case reports of sCJD with extrapyramidal signs, while an earlier study found increased signal intensity within the basal ganglia in 79% (23/29) of CJD patients.25,26 Thus the presence of high signal intensity within the basal ganglia has reasonable sensitivity and high specificity for diagnosis of sCJD. High signal intensity may also found in the thalamus (particularly in vCJD) and within the cortical ribbon (Figure 1). Several studies indicate that a variation on T2-weighted MR in which the high signal intensity from CSF is removed (fluid attenuated inversion recovery [FLAIR]) is even more sensitive to these changes (Figure 1).27 Neuropathological studies indicate that this appearance is associated with gliosis more than spongiform changes.24,26 Thus T2-weighted and FLAIR MR imaging would be expected to be most sensitive to TSEs in the later stages of disease progression.
A new type of MR imaging, diffusion-weighted (DW) MR imaging, may provide a way to image changes much earlier in disease progression. Several studies have shown that high signal intensity areas (indicating restricted diffusion) are present on DW MR images prior to the appearance of abnormality on other MR imaging methods (Figure 1).28–31 In one study they were present even before the appearance of PSWCs in the EEG or increased protein 14-3-3 in the CSF.32 Neuropathological studies indicate that this appearance is associated with the microvesicular spongiform changes that occur earlier in disease progression.31,32 DW MR images are also affected by T2 weighting, so areas with both restricted diffusion and prolonged T2 will be even brighter.28 This may be an additional reason why DW MR images are more sensitive early in disease progression. Sequential imaging in one case demonstrated that very late in disease progression this distinctive appearance on DW MR images can disappear.30 No histopathology was presented, but the authors suggest that this may be due to the development of gliosis. Thus, DW MR imaging shows promise for both the early diagnosis of TSEs and for monitoring of disease progression.
Functional brain imaging (positron emission tomography [PET], single-photon emission computed tomography [SPECT]) also appears to be quite sensitive to the changes that occur early in the TSEs. Several studies have shown functional decreases (in blood flow or metabolic rate) prior to development of clear abnormalities on structural imaging, EEG, and/or level of 14-3-3 in CSF.2,33–38 The pattern of abnormalities may be relatively specific to the TSE clinical variant. In sCJD hypoperfusion/hypometabolism primarily in frontal and temporoparietal cortices early in the disease progresses to global decreases by later stages.33,36,39 Benzodiazepine receptor binding (as measured with 123I-iomazenil SPECT) is also severely decreased, suggesting widespread neuronal degeneration.39 In iatrogenic CJD (iCJD) cerebellar clinical signs are common.19 Cerebellar hypoperfusion has been shown early in disease progression.36 In sCJD-Heidenhain variant, which is characterized by visual disturbances (including isolated homonymous hemianopsia), hypometabolism/hypoperfusion is found in occipital cortex (Figure 2).2,37,38 In familial fatal insomnia (fFI) the most common early metabolic changes are in thalamus (particularly anterior) and cingulate cortex (Figure 3).11 Neuropathological studies strongly link the autonomic dysfunction and disruption of the sleep–waking cycle that characterize this disease with anterior thalamic degeneration. The authors of this study comment: “The presence of significant hypometabolism in the thalamus and cingulate cortex in all fFI patients, regardless of symptom duration and the codon 129 genotype, indicates that the disease arises relatively early in these structures and subsequently spreads to other areas of the brain.” If this holds true for all the TSE variants, as seems likely, functional imaging may provide a basis for early differential diagnosis.
MR spectroscopy provides another way of assessing cerebral function, by measuring the levels of certain brain metabolites. Most of the few TSE cases examined with this technique were in a fairly advanced state of the disease, with widespread neuronal loss that was reflected by decreases in measured N-acetylaspartate (NAA, a neuronal marker).40–42 Two cases have been reported in which NAA was decreased in brain regions that still appeared normal by other techniques.41,43 In a case of CJD the cortical neuronal number (assessed from a brain biopsy) was normal, but NAA was decreased by 27%.41 Similarly, NAA was decreased in all areas surveyed (frontal cortex, putamen, cerebellum) in a case of GSS at a time when cerebral blood flow in these areas was normal (as measured by SPECT).43 Thus MR spectroscopy shows some promise for earlier diagnosis of TSEs and may also help to differentiate TSE from other dementing illnesses.
CONCLUSION
Diagnosis of the TSEs is challenging because there is a wide range of potential presentations, many of which do not fulfill the most widely used criteria, and there is no definitive imaging or laboratory finding. Thus it is likely that TSEs are under-reported. Both DW MR and functional imaging show promise for early differential diagnosis. They may also be useful in improving targeting of brain biopsy sites. Other tests also show some promise, such as tonsillar biopsy for vCJD and peripheral blood immunoassays for PrPTSE.22 Additionally, one in vitro study suggests that chlorpromazine and quinacrine (an antimalarial agent) reduce the conversion of PrPC to PrPTSE and may enhance clearance of the abnormal form, perhaps in part by blocking transport across the plasma membrane.44,45 Initial trials in two patients resulted in symptom reduction in one.45,46
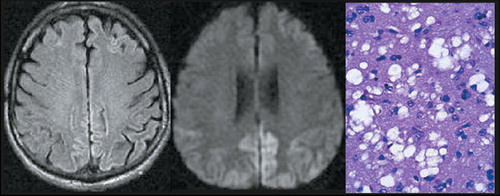
Figure 1. Increased signal is clearly present in the cortical ribbon on the FLAIR (left) and the diffusion-weighted (middle) magnetic resonance images, particularly in the posterior regions. Diagnosis of Creutzfeldt-Jakob disease was confirmed by presence of marked spongiform changes (right) in the brain at biopsy.1
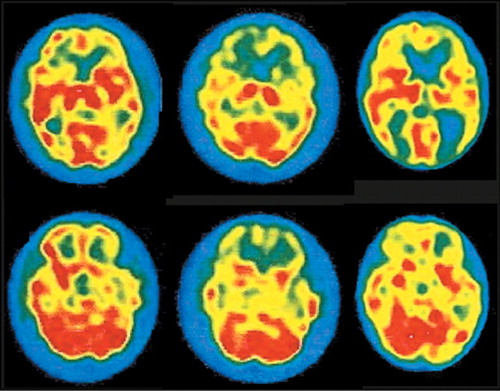
Figure 2. Images of regional cerebral blood flow (rCBF) were acquired monthly for 3 months by using technetium-99m hexamethylpropyleneamine oxime ([99mTc]HMPAO) SPECT in a patient with sporadic Creutzfeldt-Jakob disease (sCJD), Heidenhain subtype. One month after symptom onset (left) there was clearly decreased rCBF in the frontal and left temporal cortices. Decreases were more widespread one month later (middle). By one month before death (right) rCBF was diminished throughout the cerebral cortex, particularly in the temporo-occipital regions (modified with permission from Staffen et al.2).
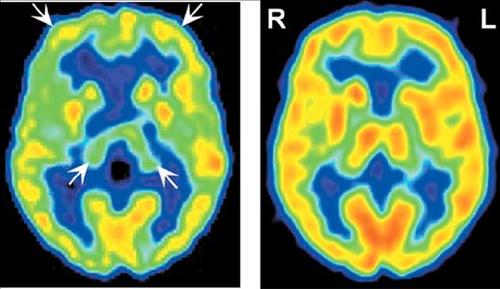
Figure 3. Regional cerebral glucose metabolism was imaged utilizing [18F]-2-fluoro-2-deoxy-d-glucose (FDG) PET in a patient with familial fatal insomnia (fFI) of 10 months duration (left). Note the severe thalamic hypometabolism and milder secondary impairment in frontal and cingulate cortices (arrows). An image of a normal individual (right) is shown for comparison (images courtesy of Dr. Perani, HSR Milan).
1 Case courtesy of Drs. Fatima Janjua and Dennis Mosier, Baylor College of Medicine: www.bcm.tmc.edu/neurol/challeng/pat52/menu.htmlGoogle Scholar
2 Staffen W, Trinka E, Iglseder B, et al: Clinical and diagnostic findings in a patient with Creutzfeldt-Jakob disease (type Heidenhain). J Neuroimaging 1997; 7:50-54Crossref, Medline, Google Scholar
3 Prusiner SB: An introduction to prion biology and diseases, in Prion Biology and Diseases, edited by Prusiner SB. Cold Spring Harbor, NY, Cold Spring Harbor Laboratory Press, 1999, pp 1-66Google Scholar
4 Riesner D: The prion theory: background and basic information, in Prions: A Challenge for Science, Medicine and Public Health System, edited by Rabenau HF, Cinatl J, Doerr HW. Basel, Karger, 2001, pp 7-20Google Scholar
5 Cappai R, Jobling MF, Barrow CJ, et al: Structural biology of prions, in Prions: A Challenge for Science, Medicine and Public Health System, edited by Rabenau HF, Cinatl J, Doerr HW. Basel, Karger, 2001, pp 32-47Google Scholar
6 Hill AF, Collinge J: Strain variations and species barriers, in Prions: A Challenge for Science, Medicine and Public Health System, edited by Rabenau HF, Cinatl J, Doerr HW. Basel, Karger, 2001, pp 48-57Google Scholar
7 Zerr I, Schulz-Schaeffer WJ, Giese A, et al: Current clinical diagnosis in Creutzfeldt-Jakob disease: identification of uncommon variants. Ann Neurol 2000; 48:323-329Crossref, Medline, Google Scholar
8 Parchi P, Capellari S, Gambetti P: Intracerebral distribution of the abnormal isoform of the prion protein in sporadic Creutzfeldt-Jakob disease and fatal insomnia. Microscopy Research and Technique 2000; 50:16-25Crossref, Medline, Google Scholar
9 Kordek R: The diagnosis of human prion diseases. Folia Neuropathol 2000; 38:151-160Medline, Google Scholar
10 Monari L, Chen SG, Brown P, et al: Fatal familial insomnia and familial Creutzfeldt-Jakob disease: different prion proteins determined by a DNA polymorphism. Proc Natl Acad Sci USA 1994; 91:2839-2842Crossref, Medline, Google Scholar
11 Cortelli P, Perani D, Parchi P, et al: Cerebral metabolism in fatal familial insomnia: relation to duration, neuropathology, and distribution of protease-resistant prion protein. Neurology 1997; 49:126-133Crossref, Medline, Google Scholar
12 Parchi P, Petersen RB, Chen SG, et al: Molecular pathology of fatal familial insomnia. Brain Pathol 1998; 8:539-548Crossref, Medline, Google Scholar
13 Montagna P, Cortelli P, Avoni P, et al: Clinical features of fatal familial insomnia: phenotypic variability in relation to a polymorphism as codon 129 of the prion protein gene. Brain Pathol 1998; 8:515-520Crossref, Medline, Google Scholar
14 Cortelli P, Gambetti P, Montagna P, et al: Fatal familial insomnia: clinical features and molecular genetics. J Sleep Res 1999; 8:23-29Crossref, Medline, Google Scholar
15 van Duijn CM, Delasnerie-Lauprêtre N, Masullo C, et al: Case-control study of risk factors of Creutzfeldt-Jakob disease in Europe during 1993-95. Lancet 1998; 351:1081-1085Crossref, Medline, Google Scholar
16 Coulthart MB, Cashman NR: Variant Creutzfeldt-Jakob disease: a summary of current scientific knowledge in relation to public health. CMAJ Can Med Assoc J 2001; 165:51-58Google Scholar
17 Venters GA: New variant Creutzfeldt-Jakob disease: the epidemic that never was. BMJ 2001; 323:858-861Crossref, Medline, Google Scholar
18 Brown P, Bradley R: 1755 and all that: a historical primer of transmissible spongiform encephalopathy. BMJ 1998; 317:1688-1692Crossref, Medline, Google Scholar
19 Brown P, Preece M, Brandel J-P, et al: Iatrogenic Creutzfeldt-Jakob disease at the millennium. Neurology 2000; 55:1075-1081Crossref, Medline, Google Scholar
20 Zerr I, Poser S: Epidemiology and risk factors of transmissible spongioform encephalopathies in man, in Prions: A Challenge for Science, Medicine and Public Health System, edited by Rabenau HF, Cinatl J, Doerr HW. Basel, Karger, 2001, pp 93-104Google Scholar
21 Knight R, Collins S: Human prion diseases: cause, clinical and diagnostic aspects, in Prions: A Challenge for Science, Medicine and Public Health System, edited by Rabenau HF, Cinatl J, Doerr HW. Basel, Karger, 2001, pp 68-92Google Scholar
22 Collins S, Boyd A, Fletcher A, et al: Recent advances in the pre-mortem diagnosis of Creutzfeldt-Jakob disease. J Clin Neurosci 2000; 7:195-202Crossref, Medline, Google Scholar
23 Poser S, Mollenhauer B, Kraub A, et al: How to improve the clinical diagnosis of Creutzfeldt-Jakob disease. Brain 1999; 122:2345-2351Crossref, Medline, Google Scholar
24 Schröter A, Zerr I, Henkel K, et al: Magnetic resonance imaging in the clinical diagnosis of Creutzfeldt-Jakob disease. Arch Neurol 2000; 57:1751-1757Crossref, Medline, Google Scholar
25 Finkenstaedt M, Szudra A, Zerr I, et al: MR imaging of Creutzfeldt-Jakob disease. Radiology 1996; 199:793-798Crossref, Medline, Google Scholar
26 Urbach H, Klisch J, Wolf HK, et al: MRI in sporatic Creutzfeldt-Jakob disease: correlation with clinical and neuropathological data. Neuroradiology 1998; 40:65-70Crossref, Medline, Google Scholar
27 de Priester JA, Jansen GH, de Kruijk JR, et al: New MRI findings in Creutzfeldt-Jakob disease: high signal in the globus pallidus on T1-weighted images. Neuroradiology 1999; 41:265-268Crossref, Medline, Google Scholar
28 Bahn MM, Parchi P: Abnormal diffusion-weighted magnetic resonance images in Creutzfeldt-Jakob disease. Arch Neurol 1999; 56:577-583Crossref, Medline, Google Scholar
29 Demaerel P, Heiner L, Robberecht W, et al: Diffusion-weighted MRI in sporatic Creutzfeldt-Jakob disease. Neurology 1999; 52:205-208Crossref, Medline, Google Scholar
30 Matoba M, Tonami H, Miyaji H, et al: Creutzfeldt-Jakob disease: serial changes on diffusion-weighted MRI. J Comput Assist Tomogr 2001; 25:274-277Crossref, Medline, Google Scholar
31 Mao-Draayer Y, Braff SP, Nagle KJ, et al: Emerging patterns of diffusion-weighted MR imaging in Creutzfeldt-Jakob disease: case report and review of the literature. AJNR Am J Neuroradiol 2002; 23:550-556Medline, Google Scholar
32 Mittal S, Farmer P, Kalina P, et al: Correlation of diffusion-weighted magnetic resonance imaging with neuropathology in Creutzfeldt-Jakob disease. Arch Neurol 2002; 59:128-134Crossref, Medline, Google Scholar
33 Ogawa T, Inugami A, Fujita H, et al: Serial positron emission tomography with fludeoxyglucose F 18 in Creutzfeldt-Jakob disease. AJNR Am J Neuroradiol 1995; 16:978-981Medline, Google Scholar
34 de Silva R, Patterson J, Hadley D, et al: Single photon emission computed tomography in the identification of new variant Creutzfeldt-Jakob disease: case reports. BMJ 1998; 316:593-594Crossref, Medline, Google Scholar
35 Miller DA, Vitti RA, Maslack MM: The role of 99m-Tc HMPAO SPECT in the diagnosis of Creutzfeldt-Jakob disease. AJNR Am J Neuroradiol 1998; 19:454-455Medline, Google Scholar
36 Matsuda M, Tabata K-I, Hattori T, et al: Brain SPECT with 123I-IMP for the early diagnosis of Creutzfeldt-Jakob disease. J Neurol Sci 2001; 183:5-12Crossref, Medline, Google Scholar
37 Jacobs DA, Lesser RL, Mourelatos Z, et al: The Heidenhain variant of Creutzfeldt-Jakob disease: clinical, pathologic, and neuroimaging findings. J Neuro-opthalmology 2001; 21:99-102Crossref, Medline, Google Scholar
38 Mathews D, Unwin DH: Quantitative cerebral blood flow imaging in a patient with the Heidenhain variant of Creutzfeldt-Jakob disease. Clin Nucl Med 2001; 26:770-773Crossref, Medline, Google Scholar
39 Itoh Y, Amano T, Shimizu T, et al: Single-photon emission computed tomography image of benzodiazepine receptors in a patient with Creutzfeldt-Jakob disease. Intern Med 1998; 37:896-900Crossref, Medline, Google Scholar
40 Bruhn H, Weber T, Thorwirth V, et al: In-vivo monitoring of neuronal loss in Creutzfeldt-Jakob disease by proton magnetic resonance spectroscopy. Lancet 1991; 337:1610-1611Crossref, Medline, Google Scholar
41 Graham GD, Petroff OAC, Blamire AM, et al: Proton magnetic resonance spectroscopy in Creutzfeldt-Jakob disease. Neurology 1993; 43:2065-2068Crossref, Medline, Google Scholar
42 Shyu W-C, Lee C-C, Hsu Y-D, et al: Panencephalitic Creutzfeldt-Jakob disease: unusual presentation of magnetic resonance imaging and proton magnetic resonance spectroscopy. J Neurol Sci 1996; 138:157-160Crossref, Medline, Google Scholar
43 Konaka K, Kaido M, Okuda Y, et al: Proton magnetic resonance spectroscopy of a patient with Gerstmann-Straussler-Scheinker disease. Neuroradiology 2000; 42:662-665Crossref, Medline, Google Scholar
44 Korth C, May BCH, Cohen F, et al: Acridine and phenothiazine derivatives as pharmacotherapeutics for prion disease. Proc Natl Acad Sci USA 2001; 98:9836-9841Crossref, Medline, Google Scholar
45 Amaral L, Kristiansen JE: Phenothiazines: potential management of Creutzfeldt-Jacob disease and its variants. Int J Antimicrob Agents 2001; 18:411-417Crossref, Medline, Google Scholar
46 Josefson D: Drugs for malaria and psychosis may offer hope to people with CJD. BMJ 2001; 323:416-416Crossref, Medline, Google Scholar



