Longitudinal Improvement in Psychomotor Processing Speed Is Associated With Potent Combination Antiretroviral Therapy in HIV-1 Infection
Abstract
This longitudinal natural history study aimed to assess the pattern and durability of neurocognitive benefits of progressively more potent combination antiretroviral therapy in HIV-1 infection. A cohort of 141 homosexual or bisexual men were assessed semiannually for CD4 count, HIV RNA viral load, medical and depressive symptoms, and a neuropsychological test battery, including psychomotor speed, verbal memory, and executive function. In a mixed-effects model, increasingly potent antiretroviral therapy was associated with improvement in tests of psychomotor processing speed. This study contributes to the growing literature documenting the longitudinal benefit provided by potent antiretroviral therapy for neuropsychological function, particularly psychomotor processing speed, in patients with HIV illness.
HIV-1 enters the central nervous system early in the course of infection and may produce a range of neuropsychological symptoms over time. These symptoms include impairments in psychomotor processing speed, executive function, and memory that may progress to clinical disorders, including minor cognitive motor disorder and dementia,1 which independently predict shortened survival.2 With the introduction of combination antiretroviral therapy (ART) that includes a protease inhibitor—or highly active antiretroviral therapy (HAART)—AIDS-related morbidity and mortality in populations with access to treatment have declined dramatically.3 However, the impact of these therapies on CNS disease is in the early stages of investigation.
Recent evidence suggests that an elevation in CNS (cerebrospinal fluid and brain) viral load, but not in plasma viral load, is associated with HIV-related neurocognitive disorders.4–6 HIV found in the CNS also exhibits genotypic and phenotypic variation compared with that found in the systemic circulation.4,7 This evidence suggests that the CNS is an independent reservoir for HIV-1 replication, particularly in the later stages of disease.8 This has important implications for ART, because sufficient blood-brain barrier penetration to achieve adequate HIV-1 inhibitory concentrations in the CNS may be necessary for durable antiretroviral efficacy against neurocognitive disorders.9
The nucleoside analog reverse transcriptase inhibitors (NRTIs) zidovudine, stavudine, lamivudine, and abacavir and the non-nucleoside reverse transcriptase inhibitors (NNRTIs) nevirapine and efavirenz have been found to penetrate the blood-brain barrier and achieve sufficient concentration to inhibit HIV replication.9–13 Zidovudine (AZT) monotherapy at current standard and higher doses has been shown to improve neuropsychological performance14,15 and severity of dementia16 and was associated with the decline in the incidence of dementia from the late 1980s to the early 1990s.17 However, the benefits of AZT monotherapy may be temporary because of the emergence of viral resistance.18
The protease inhibitors, with the exception of indinavir,19 have poor blood-brain barrier penetration, which has generated concern that HAART might not confer any benefit in CNS disease. However, HAART regimens containing various protease inhibitors have been associated with reductions of CSF viral load to undetectable levels,7,20 with reversal of white matter lesions on magnetic resonance imaging,21 and with reversal of brain metabolic abnormalities detected by proton MR spectroscopy.22 In a cross-sectional assessment of men with symptomatic HIV and AIDS, HAART, compared with less potent ART, was associated with superior performance on tasks of verbal memory, psychomotor speed, and executive function.23 An open-label trial of HAART with 26 patients who had advanced HIV illness reported improvements in psychomotor speed and memory in 75% of patients after a 15-month follow-up.24 Analysis of data collected over 1 year from 411 homosexual men in the Multicenter AIDS Cohort Study revealed that combination ART with or without a protease inhibitor was associated with improved psychomotor speed.25
In this study we aimed to further assess the pattern and durability of the potential neurocognitive benefits of progressively more potent ART after accounting for other factors that might affect cognitive performance, including practice—that is, the effect of repeatedly undergoing the battery of neuropsychological tests. The primary questions for this analysis are: Is progressively more potent ART associated with neuropsychological improvement over the course of 1 year? If so, what is the pattern of this improvement?
METHOD
Sample
Data for this study were derived from a longitudinal study of homosexual and bisexual men initiated in July 1995 to examine factors associated with psychosocial adjustment to AIDS.26 Initially, 173 subjects were recruited from advertisements in community-based organizations. Although individuals at all stages of HIV infection were included, recruitment of those with symptomatic illness was emphasized. Men reporting a past or current history of intravenous drug use were excluded, as were men with insufficient English fluency to complete the testing. Laboratory evaluations, HIV symptom measures and illness staging, depression rating, and medication treatment data were collected at six semiannual visits over 2.5 years. Neuropsychological testing was initiated at visit 4 (18 months) with 141 subjects, and repeated at visits 5 and 6. The 141 subjects who were assessed at visit 4, which constitutes the neuropsychological baseline, are included in this analysis.
Measures
Laboratory measures. Assays to determine CD4 lymphocyte count and peripheral HIV RNA viral load (Roche, Amplicor; lower limit of detection 400 copies/ml) were performed by Corning-Metpath (now Quest) Laboratory.
Medical symptoms. A medical symptom checklist adapted from earlier HIV-positive cohort studies was used.26 This self-report scale consists of 14 signs and symptoms commonly associated with HIV infection, such as night sweats and oral candidiasis. The total score, which ranges from 0 to 14, indicates the number of symptoms currently present.
Physical limitations. The Rand Physical Limitations Scale was used to assess physical functional ability.27 This self-report scale requires respondents to rate their ability to accomplish ten physical tasks listed in descending order of difficulty. Higher scores indicate more physical limitations.
Depressive symptoms. The Beck Depression Inventory (BDI) was used as a self-report measure of depressive symptoms, with scores ranging 0 to 63.28 Scores greater than 10 indicate clinically significant depressive symptoms.
Neuropsychological assessment. The neuropsychological test battery assessed the domains of attention, concentration, verbal memory, psychomotor speed, and executive function. Tests administered at visits 4 through 6 were the Trail Making Test, Parts A and B,29 the California Verbal Learning Test (CVLT),30 the Digit Symbol Substitution Test,31 and the Stroop Color Word Test.32 The Grooved Pegboard Test was administered at visits 4 and 6.33 For data analysis, neuropsychological test scores were used in two ways: raw scores were used as continuous measures, and subjects were also rated as neuropsychologically impaired or unimpaired on the basis of their performance relative to published normative data. Subjects were classified as neuropsychologically impaired according to American Academy of Neurology criteria for HIV-associated cognitive motor disorders—that is, if they scored one or more standard deviations in the impaired direction relative to age-matched population-based norms on two or more neuropsychological tests or two or more standard deviations in the impaired direction on one or more neuropsychological tests.1
Antiretroviral treatment potency. Given the naturalistic design of this study, the heterogeneity of ART regimens and the high rate of switching precluded analysis of the impact of stable regimens on cognitive performance. Thus, in order to provide a measure of regimen potency at each visit, five antiretroviral potency groups were established on the basis of recommendations of the International AIDS Society–USA Panel at the time of data collection.34 Subjects were grouped as follows: category 1, zero to one antiretroviral; category 2, two NRTIs; category 3, three NRTIs + the original formulation of saquinavir (which had low bioavailability); category 4, two NRTIs + nevirapine or delavirdine (NNRTIs); and category 5, two NRTIs with or without one NNRTI + ritonavir, indinavir, or nelfinavir (HAART).
Procedure
Between July 1995 and December 1997, subjects were seen for six semiannual visits, each lasting 3 to 4 hours, during which self-ratings, neuropsychological assessments, and blood tests were completed. Medical information was elicited by clinicians under the supervision of a physician who worked in the hospital HIV/AIDS clinic (S.J.F.), and neuropsychological assessments were administered under the supervision of a board-certified neuropsychologist (W.v.G.). Written informed consent was obtained under the institutional review board guidelines of Weill Medical College of Cornell University. Subjects were paid $30 or $40 for each study visit.
Statistical Analyses
Outcome (dependent) variable distributions were examined for normality, and five variables were found to be significantly skewed. Log10 transformations were used for HIV RNA, Trail Making Test A and B scores, and Grooved Pegboard Dominant and Nondominant hand scores. Cross-sectional and preliminary longitudinal comparisons were made using t tests, χ2 tests, and analysis of variance.
The longitudinal data were fitted to mixed-effect models35 with the SAS Institute's mixed procedure composed of fixed effect and random effect.36 The data were fitted to the model with random effects of intercept and time, and unstructured covariance of random coefficients and constant error variances. The maximum likelihood method was used for the estimation. The type 3 F test was used in significance testing. In these analyses, all available data were used, without estimations for missing data. Subjects for whom there were missing data were retained in the analyses.
As a preliminary analysis to determine whether missing values resulting from dropout were missing at random, the logistic regression approach outlined by Ridout37 and illustrated by Vonesh and Chinchilli38 was applied. The results revealed that dropout was dependent on the observed outcome measurement history of the CVLT sum and long-delay free recall tests. In addition, time (measured as the number of semiannual visits) was correlated with all neuropsychological outcome measures, indicating informative censoring. Hence, for analyzing these variables, the conditional mixed-effects model representing a class of pattern mixture model39 was employed. To take into account missing data, the subject's time in the study was used as a covariate for all measures. For the CVLT sum and long-delay free recall subtests, indicators representing death or other dropout or study completion and the number of missing visits for adjusting intermittent missing values were included as additional covariates in the model. In the modeling, change in individual response was focused, and subject-specific random effects were included in the model. Consequently, the intercept and time factor were assumed as random effects. For analyzing the dichotomous neuropsychologically impaired versus unimpaired outcome, as a linear function of covariates, we employed the generalized estimating equation (GEE) approach.40 The analyses were performed with SAS PROC MIXED.36
In the longitudinal mixed-effects models, scores for neuropsychological tests at visits 4, 5, and 6 were the dependent variables. We used the following covariates to assess associations with neuropsychological test scores over time: number of visits, age, race, level of education, CD4 count, log10 HIV RNA, Physical Limitation score, HIV symptom score, BDI score, antidepressant or anxiolytic use (yes or no), and ART potency. With the exception of race and years of education, all covariates were time variant.
RESULTS
Demographics and HIV Illness Measures
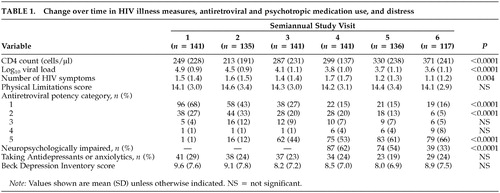
At study baseline, the 141 subjects had a mean age of 40 (SD = 8), 59 (42%) were nonwhite (mostly African American or Hispanic), and 88% had some post–high school education (mean, 14 ± 2 years). Sixty-five (46%) were working more than 20 hours per week. Average time since notification of HIV status was approximately 5 years. Mean CD4 count at baseline was 249 ± 228 cells/μl; mean log10 HIV RNA was 4.9 ± 0.9 copies/ml; 115 (82%) of the subjects had AIDS; and nearly all subjects had symptomatic HIV illness.
As Table 1 shows, for the entire sample, mean CD4 count increased 122 cells/μl during the 2.5-year period, and mean log10 HIV RNA dropped 1.3 log10 copies. The mean number of HIV symptoms also dropped significantly. These favorable changes coincided with a dramatic rise in the number of subjects taking three or more antiretrovirals (potency categories 3, 4, 5), increasing from 6% at study baseline to 79% at time 6.
Between visits 4 and 6, 20 men dropped out of the study and four died. These subjects were more likely than those who remained to be neuropsychologically impaired at their last visit (71% vs. 36%, χ2 = 10, P<0.01), although they did not differ on other measures (data not shown).
Use of antidepressants and anxiolytics. Across the six time points, mean BDI scores were in the “not depressed” range and did not change significantly over time. Approximately 25 to 30% of subjects were taking antidepressants or anxiolytics, or both, at any given time point.
Neuropsychological function. As seen in Table 1, nearly two-thirds of subjects had some neuropsychological impairment at baseline, and the impairment rate decreased significantly over the year of follow-up. At the final study visit, only 36% of subjects had impairment. At that time, subjects with neuropsychological impairment did not differ from those without on mean CD4 count (314 ± 255 vs. 381 ± 210, P = 0.16), mean log10 HIV RNA (3.7 ± 1.2 vs. 3.6 ± 1.0, P = 0.53), or proportion with undetectable virus (33% vs. 49%, P = 0.13). Sixty-seven percent of both the neuropsychologically impaired group and the unimpaired group were on HAART (potency category 5), and 81% and 77%, respectively, were taking any triple combination ART regimen (categories 3–5). Five (9%) of 54 subjects without neuropsychological impairment at baseline (three on HAART, one on saquinavir + two NRTIs and one on dual NRTIs) became neuropsychologically impaired by their last study visit. Under the multivariate GEE model, antiretroviral potency was not significantly associated with the dichotomous outcome of neuropsychologically impaired versus unimpaired (data not shown).
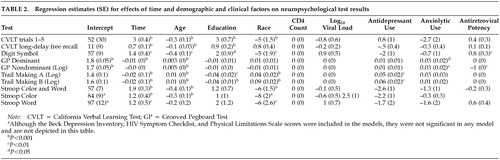
In contrast to the dichotomous outcome, in the mixed-effects models (Table 2), after controlling for factors that might affect neuropsychological test performance over time, greater antiretroviral potency was associated with significant improvements in tests of psychomotor speed (digit symbol and grooved pegboard nondominant hand tests). Time (that is, over the course of repeated test administrations) was associated with improvement in neuropsychological test scores, and older age, lower education, and nonwhite race were consistently associated with poorer performance. Antidepressant use was associated with poorer performance on both parts of the Trail Making Test (psychomotor speed, cognitive flexibility), and anxiolytic use was associated with poorer performance on the grooved pegboard test (psychomotor speed). Depression, HIV symptoms, physical limitations, CD4 count, and viral load did not significantly affect neuropsychological performance.
DISCUSSION
In this longitudinal natural history study of HIV-positive men with symptomatic HIV and AIDS, the rate of neuropsychological impairment dropped by nearly 50% over the course of 1 year, but overall impairment was not related to ART regimen potency. However, longitudinal improvement in psychomotor speed was associated with progressively more potent ART, after controlling for multiple factors that might affect neuropsychological test performance, including practice from taking the tests repeatedly over time. Impairment in psychomotor speed is a cognitive domain consistently reported to be associated with HIV,1 and improvement in this domain has been most consistently associated with ART.15,23,25 Thus, subcortical brain structures mediating psychomotor speed may not only be sensitive to HIV-associated neuropathological changes but also to the benefits of progressively more potent ART.
Although the cohort as a whole experienced significant increases in CD4 count and reductions in plasma viral load with increasing use of combination ART, changes in neuropsychological performance were not associated with these HIV illness markers. This finding is consistent with the notion that CNS immunological and viral dynamics differ from those in plasma and that these markers are insufficient to monitor the impact of ART in the CNS. Thus, alternative markers, such as serial neuropsychological testing, CSF viral load measurement,8 MR spectroscopy22 and MR diffusion tensor imaging,41 are more likely to be useful in monitoring the impact of ART in the CNS.
Although neuropsychological impairment was reduced by about 50% overall, more than one-third of this sample continued to be neuropsychologically impaired at the end of the study, and 9% of the patients who were unimpaired at baseline became neuropsychologically impaired after 6–12 months of follow-up. Moreover, about two-thirds of them had neuropsychological impairment despite being on HAART and one-third despite having plasma viral loads below detectable limits. The rate of neuropsychological test impairment seen in symptomatic HIV and AIDS before the advent of combination ART was found to exceed 50%.42 In this cohort with symptomatic HIV and AIDS, after 1 year of follow-up, the prevalence of neuropsychological impairment approximated the median prevalence of impairment seen in asymptomatic HIV-positive samples before combination ART was used.43 Residual neuropsychological impairment or the development of new neuropsychological impairment may reflect a number of factors that were not assessed in this study, including the irreversibility of HIV-associated neuropathological changes, variable CNS ARV penetration, variable adherence to the medication regimen, and the development of ARV-resistant HIV in the CNS. Such persistent neuropsychological impairment despite potent ART may impede functional ability, including capacity to work,44 and may still constitute a risk factor for shortened survival.2
A number of limitations in this study might affect the generalizability of these findings. The sample did not include women and intravenous drug users. Given rapid changes in the availability and prescribing patterns of ART, it may be difficult to determine whether findings from the period of this study will be generalizable to the medications used currently or in the future. Finally, given the heterogeneity of ART and the high rate of ARV switching in this cohort, we were unable to compare the effects of different stable ARV regimens on HIV CNS disease. However, this naturalistic study very likely reflects the trajectory of CNS disease in the real-world context of variable ART.
This study contributes to the growing literature documenting the longitudinal benefit provided by potent ART for neuropsychological function, particularly psychomotor processing speed, in patients with HIV illness. In future research, studies that extend beyond 1 year of follow-up will be crucial in determining whether extended systemic improvement is accompanied by similar improvement in neurocognitive function.
ACKNOWLEDGMENTS
This research was presented in part at the NIMH conference on “HIV and the Nervous System: Emerging Issues” held in Washington, DC, in April 1999.
 |
 |
1 Dana Consortium on Therapy for HIV Dementia and Related Disorders: Clinical Confirmation of American Academy of Neurology algorithm for HIV-1-associated cognitive/motor disorder. Neurology 1996; 47:1247-1253Crossref, Medline, Google Scholar
2 Sacktor NC, Bacellar H, Hoover DR, et al: Psychomotor slowing in HIV infection: a predictor of dementia, AIDS, and death. J Neurovirol 1996; 2:404-410Crossref, Medline, Google Scholar
3 Palella FJ, Delaney KM, Moorman AC, et al: Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. N Engl J Med 1998; 338:853-860Crossref, Medline, Google Scholar
4 Di Stefano M, Monno L, Riore JR, et al: Neurological disorders during HIV-1 infection correlated with viral load in cerebrospinal fluid but not with virus phenotype. AIDS 1998; 12:737-743Crossref, Medline, Google Scholar
5 Ellis RJ, Hsia K, Spector SA, et al: Cerebrospinal fluid human immunodeficiency virus type 1 RNA levels are elevated in neurocognitively impaired individuals with acquired immunodeficiency syndrome. Ann Neurol 1997; 42:679-688Crossref, Medline, Google Scholar
6 McArthur JC, McClernon DR, Cronin MF, et al: Relationship between human immunodeficiency virus-associated dementia and viral load in cerebrospinal fluid and brain. Ann Neurol 1997; 42:689-698Crossref, Medline, Google Scholar
7 Staprans S, Marlowe N, Glidden D, et al: Time course of cerebrospinal fluid responses to antiretroviral therapy: evidence for variable compartmentalization of infection. AIDS 1999; 13:1051-1061Crossref, Medline, Google Scholar
8 Price RW, Staprans S: Measuring the viral load in cerebrospinal fluid in human immunodeficiency virus infection: window into brain infection [editorial]. Ann Neurol 1997; 42:675-678Crossref, Medline, Google Scholar
9 Enting RH, Hoetelmans RMW, Lange JMA, et al: Antiretroviral drugs and the central nervous system. AIDS 1998; 12:1941-1955Crossref, Medline, Google Scholar
10 Ravitch JR, Jarrett JL, White HR, et al: CNS penetration of the anti-retroviral 1592 in human and animal models. Fifth Conference on Retroviruses and Opportunistic Infections, February 1998, abstract 636Google Scholar
11 Viramune package insert, Roxane Laboratories, Inc. Columbus, OHGoogle Scholar
12 Tashima K: Cerebrospinal fluid (CSF) HIV-RNA levels are undetectable (<400 copies/ml) in patients taking Sustiva (efavirenz) in combination therapy. Eighth Annual Neuroscience of HIV Infection, Basic Research and Clinical Frontiers meeting, June 3-6, 1998Google Scholar
13 Limoges J, Persidsky Y, Rasmussen J, et al:1592 prevents viral spread in brain tissue of SCID mice with HIV-1 encephalitis. Fifth Conference on Retroviruses and Opportunistic Infections, February 1998, abstract 635Google Scholar
14 Schmitt FA, Bigley JW, McKinnis R, et al, and the AZT Collaborative Working Group. Neuropsychological outcome of zidovudine (AZT) treatment of patients with AIDS and AIDS-related complex. N Engl J Med 1988; 319:1573-1578Crossref, Medline, Google Scholar
15 Martin EM, Pitrak DL, Novak RM, et al: Reaction times are faster in HIV-seropositive patients on antiretroviral therapy: a preliminary report. J Clin Exp Neuropsychol 1999; 21:730-735Crossref, Medline, Google Scholar
16 Sidtis JJ, Gatsonis C, Price RW, et al, and the AIDS Clinical Trials Group. Zidovudine treatment of the AIDS dementia complex: results of a placebo-controlled trial. Ann Neurol 1993; 33:343-349Crossref, Medline, Google Scholar
17 Baldweg T, Catalan J, Gazzard BG, et al: Risk of HIV dementia and opportunistic brain disease in AIDS and zidovudine therapy. J Neurol Neurosurg Psychiatry 1998; 65:34-41Crossref, Medline, Google Scholar
18 Gulevich SJ, McCutchan JA, Thal LJ, et al: Effect of antiretroviral therapy on the cerebrospinal fluid of patients seropositive for the human immunodeficiency virus. J AIDS 1993; 6:1002-1007Medline, Google Scholar
19 Brinkman K, Kroon F, Hugen PWH, et al: Therapeutic concentrations of indinavir in cerebrospinal fluid of HIV-1 infected patients. AIDS 1998; 12:537Crossref, Medline, Google Scholar
20 Gisslen M, Hagberg L, Svennerholm B, et al: HIV-1 RNA is not detectable in the cerebrospinal fluid during antiretroviral combination therapy. AIDS 1997; 11:1194Crossref, Medline, Google Scholar
21 Filippi C, Sze G, Farber SJ, et al: Regression of HIV encephalopathy and basal ganglia signal intensity abnormality at MR imaging in patients with AIDS after the initiation of protease inhibitor therapy. Radiology 1998; 206:491-498Crossref, Medline, Google Scholar
22 Chang L, Ernst T, Leonido-Yee M, et al: Highly active antiretroviral therapy reverses brain metabolite abnormalities in mild HIV dementia. Neurology 1999; 53:782-789Crossref, Medline, Google Scholar
23 Ferrando S, van Gorp W, McElhiney M, et al: Highly active antiretroviral treatment (HAART) in HIV infection: benefits for neuropsychological function. AIDS 1998; 12:F65-F70Google Scholar
24 Tozzi V, Balestra P, Galgani S, et al: Positive and sustained effects of highly active antiretroviral therapy on HIV-1-associated neurocognitive impairment. AIDS 1999; 13:1889-1897Crossref, Medline, Google Scholar
25 Sacktor NC, Lyles RH, Skolasky RL, et al: Combination antiretroviral therapy improves psychomotor speed performance in HIV-seropositive homosexual men. Multicenter AIDS Cohort Study (MACS). Neurology 1999; 52:1640-1647Crossref, Medline, Google Scholar
26 Rabkin JG, Ferrando SJ, Jacobsberg L, et al: Prevalence of axis I disorders in an AIDS cohort: a cross-sectional, controlled study. Compr Psychiatry 1997; 38:1-10Crossref, Google Scholar
27 Brook RH, Ware JE, Davies-Avery A, et al: Overview of adult health status measures fielded in Rand's Health Insurance Study. Med Care 1979; 17(7 suppl):20Google Scholar
28 Beck AT, Ward CH, Mendelson M, et al: An inventory for measuring depression. Arch Gen Psychiatry 1961; 4:53-63Crossref, Google Scholar
29 Reitan R: Validity of the Trail Making Test as an indication of organic brain damage. Percept Mot Skills 1958; 8:271-276Crossref, Google Scholar
30 Delis DC, Kramer JH, Kaplan E, et al: California Verbal Learning Test, Research Edition. Cleveland, Psychological Corporation, 1983Google Scholar
31 Wechsler D: Wechsler Adult Intelligence Scale-Revised. San Antonio, TX, Psychological Corporation/Harcourt Brace Jovanovich, 1985, pp 32-33Google Scholar
32 Golden CJ: Stroop Color and Word Test: A Manual for Clinical and Experimental Uses. Chicago, Stoetling, 1978Google Scholar
33 Lezak MD: Neuropsychological Assessment, 3rd edition. New York, Oxford University Press, 1983, 683-684Google Scholar
34 Carpenter CCJ, Fischl MA, Hammer SM, et al: Antiviral therapy for HIV infection in 1997: updated recommendations of the International AIDS Society-USA panel. JAMA 1997; 277:1962-1969Crossref, Medline, Google Scholar
35 Searle SR, Casella G, McCulloch CE: Variance Components. New York: John Wiley and Sons, 1992Google Scholar
36 SAS Institute, Inc: SAS/STAT Software: Changes and Enhancements. Cary, NC, SAS Institute, 1996, 531-556Google Scholar
37 Ridout M: Testing for random dropouts in repeated measurement data. Biometrics 1991; 47:1617-1621Crossref, Medline, Google Scholar
38 Vonesh EF, Chinchilli VM: Linear and non-linear models for the analysis of repeated measurements. New York, Marcel Dekker, 1997Google Scholar
39 Little RJA: Modeling the drop-out mechanism in repeated-measures studies. J Amer Statistical Assn 1995; 90:1112-1121Crossref, Google Scholar
40 Zeger SL, Liang KY: Longitudinal data analysis for discrete and continuous outcomes. Biometrics 1986; 42:121-130Crossref, Medline, Google Scholar
41 Filippi CG, Ulug AM, Ryan E, et al: Diffusion tensor imaging in HIV patients with normal-appearing white matter on brain MR scans. Am J Neuroradiol 2001; 22:277-283Medline, Google Scholar
42 Heaton RK, Grant I, Butters N, et al: The HNRC 500: Neuropsychology of HIV infection at different disease stages. JINS 1995; 1:231-251Crossref, Medline, Google Scholar
43 White DA, Heaton RK, Monsch AU, et al: Neuropsychological studies of asymptomatic human immunodeficiency virus-type-1 infected individuals. International Neuropsychological Society 1995; 1:304-315Crossref, Medline, Google Scholar
44 van Gorp WG, Baerwald JP, Ferrando SJ, et al: The relationship between employment and neuropsychological impairment in HIV infection. J International Neuropsychological Society 1999; 5:534-539Crossref, Medline, Google Scholar



