The Incidence of Mental and Behavioral Disturbances in Dementia: The Cache County Study
Abstract
A population-based prevalence sample of 355 residents of Cache County, Utah, who were diagnosed with dementia, was rated on the Neuropsychiatric Inventory (NPI). Of the 355 residents, 119 had no neuropsychiatric symptoms at baseline and were, consequently, at risk for incident mental and behavioral disturbances. The NPI was readministered approximately 18 months later to 61 surviving participants. Sixty-nine percent developed at least one mental or behavioral symptom. Delusions were most common (28%), followed by apathy (21%), and aberrant motor behavior (21%). When this incidence rate of 69% was combined with a previously estimated prevalence rate of 61%, the cumulative 18-month prevalence approached 90%. These results argue for a routine assessment of psychiatric disturbances in all patients with dementia, even among those who have never experienced symptoms of mental or behavioral disturbances.
Among the elderly, dementing illnesses and their consequences are pressing concerns. Alzheimer's disease (AD) is the most common cause of dementia in elderly patients, accounting for 50%–65% of cases.1,2 Vascular dementia (10%–15%) is probably the second most common, and Dementia with Lewy Bodies (15%–25%) is likely the third.1,2 Most dementias are irreversible and progressive, causing global cognitive and functional decline and, ultimately, death. Mental and behavioral disturbances, such as delusions, hallucinations, depression, or agitation typically occur, causing additional burden and suffering for patients and their caregivers. According to estimates from clinical samples, as many as 70%–90% of patients with dementia experience at least one mental or behavioral disturbance during the course of their illness.3–7 Such psychiatric disturbances are closely associated with impairment in quality of life and caregiver distress.4,8–10 This association may exceed that of the cognitive and functional symptoms of dementia.11 Psychiatric disturbances are also associated with more rapid cognitive decline, earlier nursing home placement, and increased cost of care.7,8,12
Two large population-based studies have estimated the prevalence of mental and behavioral disturbances in dementia. The first study assessed 178 individuals with AD in the United Kingdom.13–16 The following prevalence since the onset of illness was reported: apathy (41%), major depression (24%), agitation/aggression (20%), wandering (19%), hallucinations (17%), delusions (16%), and mania (3.5%). More recently, the Cache County study used the Neuropsychiatric Inventory (NPI)17 to assess the prevalence of 10 mental and behavioral disturbances in a cohort of 329 individuals with dementias of various etiologies.6 Sixty-one percent of participants experienced at least one disturbance in the month prior to interview. In order of decreasing frequency, the following disturbances were reported: apathy (27%), depression (24%), agitation/aggression (24%), irritability (20%), delusions (19%), anxiety (17%), aberrant motor behavior (14%), hallucinations (14%), disinhibition (9%), and elation (1%).
In the general population of persons with dementia, less is known about the incidence of mental and behavioral disturbances. One study estimated the incidence of major depression in AD patients in two large community-dwelling convenience series. Findings revealed a 0%, 3-year incidence in one series and a 1.3%, 2-year incidence in the other.18 No population-based studies to date have assessed the incidence of mental or behavioral disturbances in dementia. Population-based studies of incidence are important, as they avoid referral biases inherent in studies from clinical samples, which would tend to overestimate incidence. This study reports findings on the incidence of mental and behavioral disturbances among participants with dementia who were followed in the Cache County study.
METHODS
Sampling and Screening
The methods of the Cache County study are reported in detail elsewhere.6,19 We contacted all permanent residents of Cache County, Utah, who were 65 years old or older in January 1995 (N = 5,677) and enrolled 5,092 residents (90%) who were screened for dementia with the Modified Mini-Mental State Examination.20 Individuals who could not participate directly were characterized using the Informant Questionnaire on Cognitive Decline in the Elderly21 that was administered by a knowledgeable informant. Individuals whose screening scores suggested possible cognitive impairment were studied further with the Dementia Questionnaire.22 The Dementia Questionnaire was also used to assess a weighted, stratified probability sample of the total population, irrespective of the above screening results. The latter probability subsample, along with all other participants with a Dementia Questionnaire rating of 4 (suspected dementia) or 5 (probable dementia), underwent a comprehensive clinical assessment performed by a research nurse and a trained psychometrician. This assessment was conducted in the presence of a collateral informant at the subject's place of residence (including nursing homes). The informant was a spouse, close relative, or other person whom the subject identified as knowledgeable of his or her memory and daily functioning. The evaluation consisted of a medical and cognitive history, mental state and standardized neurological exams, a brief physical evaluation, and a 1-hour neuropsychological battery, including the Mini-Mental State Examination (MMSE).23 The NPI17 was administered by either the nurse or the psychometrician.
The study was approved by the institutional review boards of Duke University Medical Center, the Johns Hopkins University School of Public Health, and Utah State University. All study participants, or their next of kin, signed an informed consent document after a complete description of the study for each stage of the screening and assessment was given.
Procedure
After screening and careful case ascertainment, we identified 355 participants with dementia. Of these, 119 had a baseline NPI score of 0 (no symptoms in any domain). These are the populations of interest here, as they were at risk for the incidence of NPI-ascertained disturbances at follow-up examinations conducted approximately 18 months later (mean interval to follow-up 17.1, SD = 4.2). Therefore, NPI data were obtained on all of the remaining 61 (51%). Thirty-three (27%) died in the interim, and one (0.8%) moved away from the area. Twenty-two (18%) had no collateral informant available for NPI questions, and two (1.6%) refused the NPI or discontinued early. Stated otherwise, NPI follow-up was obtained on 61/85 (72%) of those who were alive and available for investigation. The same informant was used for the 18-month follow-up in 75.8% of the cases.
Similar to the methods we used to estimate psychiatric prevalence,6 we reviewed data from these 18-month follow-up evaluations in order to classify study participants into distinct clinical diagnostic categories. These assignments were made at initial diagnostic conferences that included the staff who conducted the evaluation and a geriatric psychiatrist (D.C.S, M.S., J.C.S.B., or C.G.L.). A Clinical Dementia Rating (CDR) level was assigned to each participant during this time. The CDR is a standardized global rating of dementia severity based on all available information about a patient.24 Cases were then reviewed by a panel of experts for assignment of final diagnosis. The panel included three geriatric psychiatrists, a board-certified neurologist, a senior neuropsychologist, and a cognitive neuroscientist.
Assessment of Mental and Behavioral Disturbances
The reliability and validity of the NPI have been established17 and have been used in a variety of studies, including our study of the prevalence of mental and behavioral disturbances in Cache County.6
The NPI version assesses 10 categories of mental and behavioral disturbances that occur in dementia: delusions; hallucinations; agitation; depression; anxiety; elation; apathy; disinhibition; irritability; and aberrant motor behavior, such as wandering or pacing. The instrument examines whether symptoms have occurred over a period of one month from the time the assessment is conducted. Each operationally defined disturbance is ascertained by a trained examiner through a structured interview with the caregiver. Within each domain, the NPI includes a screening question. If the screening item is answered in the negative, the rater is instructed to move to the next domain. Otherwise, the rater asks the informant a series of individual questions describing behavior reflective of disturbance in that domain. The informant is then asked about the frequency of symptoms in the domain on a 4-point scale from 1 (occasionally: less than once a week) to 4 (very frequent: more than once a day). The informant is also asked to rate the severity (disruptiveness, burden) of the behavior on a 3-point scale (mild, moderate, or severe). By multiplying the severity and frequency scores, the NPI yields a domain rating with a range from 0 to 12.
Analysis
Our primary goal was to determine the incidence of disturbance in each NPI domain. Baseline demographics (e.g., age, sex, education) and clinical variables (e.g., MMSE score, dementia duration, percentage with AD diagnosis, CDR level) of the 61 participants with completed NPIs at follow-up and the 58 participants without completed NPIs at follow-up were compared in order to assess obvious bias in loss to follow-up. Similarly, baseline variables were compared for those with an NPI score of 0 (no disturbances) at 18-month follow-up versus those with an NPI score of 1 or greater. We determined the proportion of participants who had symptoms in at least one domain at 18-month follow-up. We further determined the proportion with symptoms in each of the 10 NPI domains. A mean severity score was estimated for those with symptoms in any given domain.
RESULTS
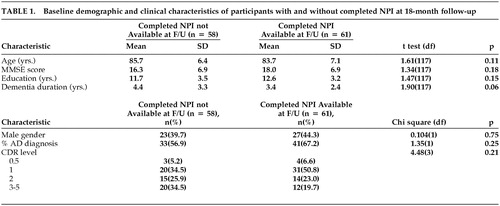
Table 1 compares demographic and clinical characteristics of the 61 participants who had a baseline NPI score of 0 and completed an 18-month follow-up evaluation, versus the 58 participants who did not. Those who did not complete follow-up NPIs showed a trend toward a longer duration of dementia (p = .06), but the two groups were otherwise similar.
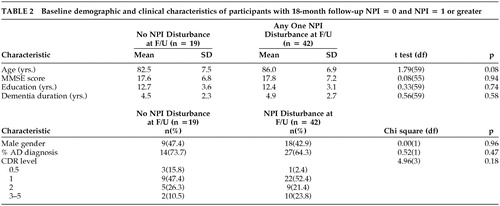
Of the 61 participants with follow-up NPI data, 42 (68.9%) exhibited at least one NPI-ascertained disturbance. Table 2 compares the baseline characteristics of the 19 participants who did not exhibit any NPI-assessed disturbance, as compared with the 42 who did. Those with incident NPI-assessed disturbances tended to be older, although this distinction did not reach statistical significance (p = .08). Of the 42 with an NPI disturbance at follow-up, 27 (64.3%) were diagnosed with AD, 1 (2.4%) with vascular dementia, 6 (14.3%) with a mixed AD and vascular dementia and 8 (19.0)% with other dementias: dementia of undetermined etiology (2), neuropsychiatric disorder (1), Parkinson's disease (1), amyotrophic lateral sclerosis with dementia (1), normal-pressure hydrocephalus (1), dementia with Lewy bodies (1), and dementia lacking distinctive histology on autopsy (1).
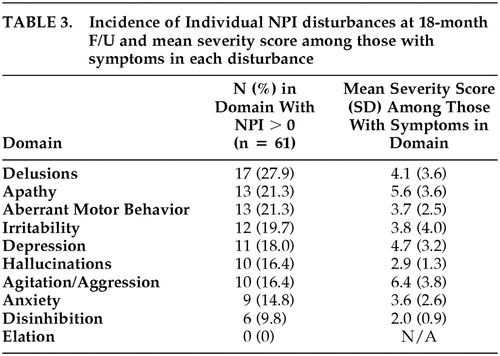
We also estimated the incidence of each individual NPI disturbance and the mean NPI severity score of subjects who were symptomatic in each domain. Table 3 shows these results. Incidence of delusions was highest, followed by apathy, aberrant motor behavior, and irritability. Incidence was lowest for elation, followed by disinhibition. All other domains had an 18-month incidence of at least 14.8%. Among those with symptoms in any given domain, the mean severity on the NPI 12-point domain scale was highest for agitation/aggression (6.4), followed by apathy (5.6) and depression (4.7). Lowest mean severity was noted for hallucinations (2.9) and disinhibition (2.0). Clinical significance of a psychiatric disturbance has been defined as a domain score of 4 or greater {personal communication, Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study}.
DISCUSSION
We report findings on the incidence of mental and behavioral disturbance in a population-based panel of elderly individuals with dementia who had been free from such comorbid symptoms approximately 18 months earlier. To our knowledge, no other study has reported similar estimates from a population sample. We found that 69% of participants developed at least one mental or behavioral symptom during the 18-month interval. For eight of the 10 NPI domains assessed, symptoms occurred in at least 15% of all subjects. Elation was the only symptom to have a 0% incidence, consistent with other findings of the relative rarity of manic and euphoric symptoms in dementia.6,15,25 These estimates likely underestimate true incidence, as we only inquired about symptoms occurring during the month prior to assessment. It is likely that some participants developed psychiatric symptoms during the 18-month interval that resolved prior to reassessment, either spontaneously or after treatment.
Additionally, we observed that 61% of participants who were initially identified with dementia had one or more NPI-ascertained mental or behavioral symptoms. Those subjects were not studied here, but combining their original prevalence of 61% with the 69% showing incident, symptoms indicate that the cumulative prevalence of mental or behavioral disturbances in dementia during 18 months may be as much as 88.6%.
Incident symptom severity was highest among subjects who developed agitation/aggression, depression, or apathy. Such symptoms are among the most distressing to both patients and caregivers and typically require a multidimensional treatment plan involving pharmacotherapy and/or environmental modification.8,26 Prompt diagnosis and intervention may alleviate the need for placement in an assisted living or nursing facility. Disinhibition and hallucinations were of mildest severity when they occurred. Often, these latter symptoms respond best to reassurance and redirection.7,8
Several limitations of our study are worthy of discussion. First, as previously mentioned, we studied a population that may not be fully representative. The Cache County population is older and less ethnically diverse than the rest of the United States, and it has higher rates of religious affiliation and lower rates of alcoholism and substance abuse than elsewhere.19 Second, the principal method of assessing mental and behavioral disturbances, the NPI, relied on informants rather than the direct examination of participants. Third, there was a fairly wide range in the time that elapsed between baseline visits and “18-month” follow-up visits. This limits our ability to draw precise estimates about incidence, but does not compromise our study's central finding of a high incidence of psychiatric disturbances in subjects who were previously assessed as symptom-free. Finally, the present sample was small, especially as compared with our prevalence study. Nearly one-half of the original panel of symptom-free participants did not have a follow-up NPI. Most losses to follow-up were due to intercurrent death or lack of a collateral informant. NPI data were obtained on three-quarters of those who were alive and available. Those subjects who did not complete the follow-up NPIs showed a trend toward a longer duration of dementia but otherwise had similar baseline characteristics.
These findings add to the growing body of evidence for the high occurrence of comorbid psychiatric symptoms in dementia and show that even patients who are asymptomatic at a given time are still at very high risk for developing psychiatric disturbances later. Psychiatric comorbidity in dementia is often complex to treat and typically requires a rigorous assessment and sophisticated treatment plan. Management may involve behavioral and/or pharmacological interventions. Successful strategies often involve some combination of the two.7,8,26 Behavioral treatments may include modifying the environment, altering the caregiving approach, and providing structured activities. Such techniques alone may be useful with symptoms of milder severity or when psychotropic use is contraindicated.7,8
Two large placebo-controlled studies have documented the benefits of atypical neuroleptics in the treatment of behavioral symptoms in AD.27,28 Placebo-controlled studies have also demonstrated the benefits of carbamazepine29 and divalproex sodium30 for agitation and aggression. Preliminary data from another placebo-controlled trial demonstrate the effectiveness of sertraline in the treatment of depression in AD.31 Recent data also suggest that cholinesterase inhibitors, which are approved by the Food and Drug Administration (FDA) to treat the cognitive symptoms of AD, may also provide behavioral benefits.32,33 The potential for effective treatment options and the high incidence rates observed in our study make early evaluation of psychiatric disturbances in dementia a vital necessity.
ACKNOWLEDGMENTS
Supported by National Institute on Aging grant AG-11380 for the Cache County Study on Memory in Aging and by NIMH grant MH-56511 for the Depression in Alzheimer's Disease Study (DIADS).
 |
 |
 |
1 Bolla LR, Filley CM, Palmer RM: Office diagnosis of the four major types of dementia. Geriatrics 2000; 55:34–46Medline, Google Scholar
2 Small GW, Rabins PV, Barry PP, et al: Diagnosis and treatment of Alzheimer's disease and related disorders: consensus statement of the American Association for Geriatric Psychiatry, the Alzheimer's Association, and the American Geriatrics Society. JAMA 1997; 278:1363–1371Crossref, Medline, Google Scholar
3 Tariot PN, Mack JL, Patterson MB, et al: The Behavior Rating Scale for Dementia of the Consortium to Establish a Registry for Alzheimer's Disease. Am J Psychiatry 1995; 152:1349–1357Crossref, Medline, Google Scholar
4 Teri L, Logsdon RG, Peskind E, et al: Treatment of agitation in AD: a randomized, placebo-controlled clinical trial. Neurology 2000; 55:1271–1278Crossref, Medline, Google Scholar
5 Rao V, Lyketsos CG: Delusions in Alzheimer's disease: a review. J Neuropsychiatry Clin Neurosci 1998; 10:373–382Link, Google Scholar
6 Lyketsos C, Steinberg M, Tschanz J, et al: Mental and behavioral disturbances in dementia: findings from the Cache County Study on Memory in Aging. Am J Psychiatry 2000; 157:708–714Crossref, Medline, Google Scholar
7 Lyketsos C, Steele C, Steinberg M: Behavioral disturbances in dementia, in Reichel's Care of the Elderly. Edited by Gallo JJ, Busby-Whitehead J, Rabins PV, Silliman R, Murphy J. Baltimore, Williams & Wilkins, 1999, pp 214–218Google Scholar
8 Rabins PV, Lyketsos CG, Steele CD: Practical Dementia Care. New York, Oxford University Press, 1999Google Scholar
9 Lyketsos CG, Breitner JCS, Rabins PV: An evidence-based proposal for the classification of neuropsychiatric disturbance in Alzheimer's disease. Int J Geriatr Psychiatry 2001; 16:1037–1042Crossref, Medline, Google Scholar
10 Deimling GT, Bass DM: Symptoms of mental impairment among elderly adults and their effects on family caregivers. J Gerontology 1986; 41:778–784Crossref, Medline, Google Scholar
11 Gonzales-Salvador T, Arango C, Lyketsos CG, et al: The stress and psychological morbidity of the Alzheimer patient caregiver. Int J Geriat Psychiatry 1999; 14:701–710Crossref, Medline, Google Scholar
12 Teri L, Hughes JP, Larson EB: Cognitive deterioration in Alzheimer's disease: behavioral and health factors. J Gerontol B: Psychol Sci 1990; 45:P58-P63Google Scholar
13 Burns A, Jacoby R, Levy R: Psychiatric phenomena in Alzheimer's disease I: disorders of thought content. Br J Psychiatry 1990; 157:72–76Crossref, Medline, Google Scholar
14 Burns A, Jacoby R, Levy R: Psychiatric phenomena in Alzheimer's disease II: disorders of perception. Br J Psychiatry 1990; 157:76–81Crossref, Medline, Google Scholar
15 Burns A, Jacoby R, Levy R: Psychiatric phenomena in Alzheimer's disease III: disorders of mood. Br J Psychiatry 1990; 157:81–86Crossref, Medline, Google Scholar
16 Burns A, Jacoby R, Levy R: Psychiatric phenomena in Alzheimer's disease IV: disorders of behavior. Br J Psychiatry 1990; 157:86–94Crossref, Medline, Google Scholar
17 Cummings JL, Mega M, Gray K, et al: The Neuropsychiatric Inventory: comprehensive assessment of psychopathology in dementia. Neurology 1994; 44:2308–2314Crossref, Medline, Google Scholar
18 Weiner M, Edland S, Luszczynska H: Prevalence and incidence of major depression in Alzheimer's disease. Am J Psychiatry 1994; 151:1006–1009Crossref, Medline, Google Scholar
19 Breitner JCS, Wyse BW, Anthony JC, et al: APOE-epsilon 4 count predicts age when prevalence of Alzheimer's disease increases- then declines: the Cache County Study. Neurology 1999; 52:321–331Crossref, Medline, Google Scholar
20 Teng EL, Chui HC: The Modified Mini-Mental State (3MS) examination. J Clin Psychiatry 1987; 48:314–318Medline, Google Scholar
21 Jorm AF: A short form of the Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE): development and cross-validation. Psychol 1994; 24:145–153Google Scholar
22 Silverman JM, Breitner JCS, Mohs RC, et al: Reliability of the family history method in genetic studies of Alzheimer's disease and related dementias. Am J Psychiatry 1986; 143:1279–1282Crossref, Medline, Google Scholar
23 Folstein M, Folstein S, McHugh P: “Mini-Mental State”: a practical method of grading the cognitive state of patients for clinicians. J Psychiatr Res 975; 12:189–198Google Scholar
24 Morris J: Clinical Dementia Rating Scale. St. Louis, Washington University, 1994Google Scholar
25 Lyketsos CG, Corazzini K, Steele C: Mania in Alzheimer's disease. J Neuropsychiatry Clin Neurosci 1998; 10:440–447Link, Google Scholar
26 Leroi I, Steele C, Lyketsos CG: The care of patients with dementia. Reviews in Clinical Gerontology 1999; 9:235–255Crossref, Google Scholar
27 Katz IR, Jeste DV, Mintzer JE, et al: Comparison of risperidone and placebo for psychosis and behavioral disturbances associated with dementia: a randomized, double-blind trial. J Clin Psychiatry 1999; 60:107–115Crossref, Medline, Google Scholar
28 Street J, Clark S, Gannon K, et al: Olanzapine treatment of psychotic and behavioral symptoms in patients with Alzheimer's disease in nursing care facilities: a double-blind randomized placebo-controlled trial. Arch Gen Psychiatry 2000; 57:968–976Crossref, Medline, Google Scholar
29 Tariot PN, Erb R, Podgorski CA, et al: Efficacy and tolerability of carbamazepine for agitation and aggression in dementia. Am J Psychiatry 1998; 155:54–61Crossref, Medline, Google Scholar
30 Porsteinsson AP, Tariot PN, Erb R, et al: Placebo-controlled study of divalproex sodium for agitation in dementia. Am J Geriatr Psychiatry 2001; 9:58–66Crossref, Medline, Google Scholar
31 Lyketsos CG, Sheppard J, Steele C, et al: A randomized, placebo-controlled, double-blind clinical trial of sertraline in the treatment of depression complicating Alzheimer's disease: initial results from the Depression in Alzheimer's Disease Study (DIADS). Am J Psychiatry 2000; 57:968–976Google Scholar
32 Cummings JL, Arnand R, Koumaris B, et al: Rivastigmine provides behavioral benefits to Alzheimer's disease patients residing in a nursing home: findings from a 26-week trial. Neurology 2000; 54(suppl 3):A468Google Scholar
33 Tariot PN, Solomon PR, Morris JC, et al: A 5-month randomized, placebo-controlled trial of galantamine in AD. Neurology 2000; 15:586–593Google Scholar



