Electrical Injury, Part I: Mechanisms
This report is the first of a two-part series on the mechanisms and consequences of electrical injury. Part II, addressing clinical sequelae and symptoms, will appear in the Fall 2009 issue of the Journal of Neuropsychiatry and Clinical Neurosciences.
MECHANISMS OF INJURY
Four mechanisms of cellular injury by electricity are presently known. They are the direct effects of the current, thermal burns, mechanical injury due to falls, and electroporation. 13 The passage of the current through tissue can cause intense muscle contractions. Current pathways including the chest can be life-threatening as a result of induced asystole and/or apnea. Electricity may also be converted into heat, resulting in the varying degrees of burns particularly associated with high-voltage electrical injury. Loss of consciousness is possible, which may lead to a fall and mechanical blunt force trauma to the head. Finally, electricity can directly cause pore formation in the lipid bilayers that form cell membranes, a process called electroporation ( Figure 1 ). Previously, thermal injuries were presumed to be the primary cause of any cellular damage. More recently, electroporation has been identified as the cause of the more rapid and diffuse necrosis associated with electrical injury. This discussion will focus on thermal burns and electroporation, the two mechanisms presently believed to be responsible for most direct injury to the nervous system.
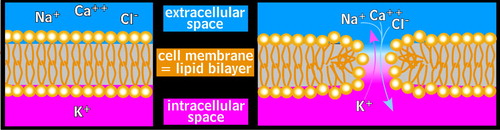
In simplistic terms, electricity passes through the body from the entrance site to the exit site. The entrance site for most electrical injuries is the point of contact. Bodily orifices (e.g., eyes, ears, nose, mouth) have been shown to be important entry sites for lightning strikes. 14 Current will course through the body via multiple parallel paths. The amount is in inverse proportion to the resistance of each path, so the majority of the current courses through the paths of least resistance. In real life these paths are not always the physically shortest or the most obvious paths from the entry to exit site. 15 Both nerves and blood within vessels, for example, have lower resistance than either bone or fat. 5 CSF has lower resistance even than blood and both may readily conduct an electrical current. 14 Neurons and the brain may therefore be uniquely vulnerable to electrical injury. The unpredictability of electrical paths is particularly evident when an electrical injury victim loses consciousness despite the current’s path not appearing to involve the brain (e.g., foot to foot). 16
When electricity encounters resistance, heat is generated based on the level of current (squared), tissue resistance, and duration of exposure. Thermal damage associated with high-voltage electrical injury can be massive, especially should a “no-let-go” series of muscle contractions occur. Thermal injuries may only be on the surface or may extend deep into the tissue. Lightning is a useful example of the complex interplay of current, resistivity, and exposure time. Despite the massive voltage, lightning rarely causes significant burns because contact time is extremely brief. By the time thermal burns are evident, considerable electroporation (see below) will have occurred. Paralysis and other acute neurological complications are quite possible in the absence of surface burns. 17 Thus, the presence of thermal burns is not a good predictor of the severity of the neurological injury. In fact, the absence of thermal burns could indicate that the electrical current was more efficiently transferred through the skin and into the body. 13 Only in prolonged electrical exposures with higher electrical fields does thermal damage begin to outweigh cellular damage due to electroporation.
As noted above, electroporation is the process by which an electrical field induces formation of pores through the cell membrane, allowing free passage of ions and fluid ( Figure 1 ). 1 – 4 The cell will attempt to maintain its ionic gradients through great expenditures of energy. If the pores do not reseal, the cell will eventually exhaust its energy reserves and die. If the pores seal, the cell may eventually recover. However, the sealing process is considerably slower than the pore opening. Even if the pores close, secondary injury processes may be disruptive or even fatal to the cell. The influx of sodium ions in a permeabilized cell prevents the transmission of signals. 18 The influx of calcium ions could cause the cytoskeleton of the cell to collapse, much like what occurs in diffuse axonal injury (DAI). 19 , 20 In addition, a recent study indicates that a bioelectric current can be generated in an area of nervous system injury, driven by the potential difference between nearby intact cell membranes and the disrupted cells. 20 Induction of large intracellular voltages along the long axis of involved neurons could contribute to ongoing secondary injury. It has also been suggested that electroporation may activate genetic messengers (e.g., c- fos mRNA transmitters) which in turn activate the hypothalamic-pituitary-adrenal (HPA) axis. While not yet demonstrated, there is also a high likelihood of cortisol activation, which could interact with both the HPA axis and other factors (e.g., brain-derived neurotrophic factor). Consequent hippocampal damage has been implicated in the genesis of depression and associated psychological features (C.J. Andrews, personal communication). Thus, injury remote from the actual pathway of the current (e.g., brain injury remote from peripheral current) becomes realistic.
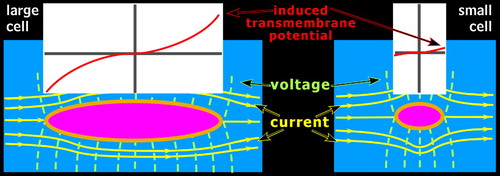
Unlike thermal heating, electroporation may not affect every cell in a specific region. In the human body, electricity encounters the greatest resistance when passing through any cellular membrane. For small cells, electricity may more readily flow around rather than through the cells. However, nerve and muscle cells are larger than other cells. Thus, electricity is more likely to pass through rather than around these cells. 4 The larger the cell, the more likely it is to be injured by this mechanism ( Figure 2 ). In an experiment focusing on the refractory period (time required for an axon to reset before firing again), larger myelinated axons were shown to be more vulnerable to electroporation than smaller axons. 18 Consequently, neurons with large diameter axons that project long distances are much more likely to be injured by exposure to electrical currents than neurons with fine axons or that project short distances. The spatial orientation of the cell in relation to the electrical path also influences the likelihood of injury. If the current approaches the narrow, or thin side, of the cell, the electricity will have a smaller effect as it passes quickly through or around the cell as opposed to traversing the cell’s entire length. 1 Thus, electricity could in effect skip over some cells based on their size and/or spatial orientation while fatally damaging others in a relatively small area. These factors may account for some of the variability and diffuse nature of sequelae from electrical injury.
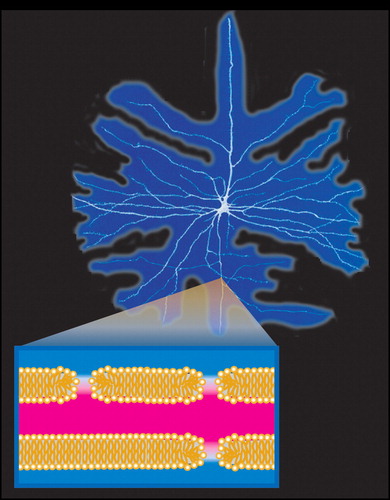
In addition to being a primary injury mechanism for electrical injuries, electroporation is also gaining prominence as a useful research and medical process. Reversible electroporation allows cells to take in large molecules that otherwise could not pass through the cell’s membrane. Once the controlled electrical field is turned off, the cell will reseal. This method is now used to insert genes or drugs (e.g., electrochemotherapy) into cells. 3 , 21 Irreversible electroporation involves use of a much stronger current that opens pores too large to reseal, and can be used to remove injured or cancerous tissue. Application of this technique in medicine is particularly promising because of its speed, ease of use, and path specificity. 22
Neuroimaging
While electrical exposure clearly can cause measurable acute neurological injury and even death, the long-term sequelae for survivors are more controversial. Imaging evidence (e.g., MRI, CT) of structural changes in the brain following electrical injury would support an “organic” rather than “psychogenic” origin of long-term symptoms ( Figure 3 ). However, no large-scale neuroimaging studies have yet been done. A 2001 review of the literature reported that 32% (16/50) of structural imaging studies (either MRI or CT) were abnormal. 5 The most common abnormalities were edema, mild atrophy, and hemorrhage in the basal ganglia. The authors noted that scans were obtained at various times post-injury and some injuries could have included mechanical traumatic brain injuries (TBIs). A study comparing patients with high-voltage electrical injuries to patients with TBI found that neuroimaging was more likely to be positive in the TBI group, and electroencephalography was more likely to be positive in the electrical injury group at all times examined (<1 month, 1–6 months, >6 months). 23 Initial imaging is often abnormal in case reports in which the electrical path clearly involved the head. 24 – 27 Imaging abnormalities have also been reported when the current path clearly did not involve the head. 28 Only a few reports include longitudinal imaging. Improvement has been noted in some but not all cases. 25 , 29 , 30 Thus, electrical injury does appear to have the potential to cause transitory or permanent structural changes in the brain.
CONCLUSION
Electroporation has only recently been identified as a primary cause of neurological injury following electrical exposure, and is incompletely understood. Other mechanisms of injury may also be present, such as the breakdown of the cytoskeleton due to excessive intracellular calcium or from systematic alterations of the HPA axis. Of particular interest is how seemingly remote electrical exposures affect the brain. 31 Furthermore, the impact of electrical injuries on the developing nervous system of a child is not known. 32 Additional longitudinal studies are sorely needed to determine the progression of electrical injuries over time.
1. Lee RC, Zhang D, Hannig J: Biophysical injury mechanisms in electrical shock trauma. Annu Rev Biomed Eng 2000; 2:477–509Google Scholar
2. Tieleman DP: The molecular basis of electroporation. BMC Biochem 2004; 5:10Google Scholar
3. Teissie J, Golzio M, Rols MP: Mechanisms of cell membrane electropermeabilization: a minireview of our present (lack of?) knowledge. Biochim Biophys Acta 2005; 1724:270–280Google Scholar
4. Lee RC: Cell injury by electric forces. Ann NY Acad Sci 2005; 1066:85–91Google Scholar
5. Duff K, McCaffrey RJ: Electrical injury and lightning injury: a review of their mechanisms and neuropsychological, psychiatric, and neurological sequelae. Neuropsychol Rev 2001; 11:101–116Google Scholar
6. Rai J, Jeschke MG, Barrow RE, et al: Electrical injuries: a 30-year review. J Trauma 1999; 46:933–936Google Scholar
7. Cherington M, Yarnell PR, Lane J, et al: Lightning-induced injury on an airplane. J Trauma 2002; 52:579–581Google Scholar
8. Stockwell B, Bellis G, Morton G, et al: Electrical injury during “hands on” defibrillation: a potential risk of internal cardioverter defibrillators? Resuscitation 2009; 80:832–834Google Scholar
9. Tkachenko TA, Kelley KM, Pliskin NH, et al: Electrical injury through the eyes of professional electricians. Ann N Y Acad Sci 1999; 888:42–59Google Scholar
10. Taylor AJ, McGwin G Jr, Valent F, et al: Fatal occupational electrocutions in the United States. Inj Prev 2002; 8:306–312Google Scholar
11. Ritenour AE, Morton MJ, McManus JG, et al: Lightning injury: a review. Burns 2008; 34:585–594Google Scholar
12. Centers for Disease Control and Prevention: Lightning-associated injuries and deaths among military personnel, United States, 1998–2001. MMWR 2002; 51:859–862Google Scholar
13. Spies C, Trohman RG: Narrative review: Electrocution and life-threatening electrical injuries. Ann Intern Med 2006; 145:531–537Google Scholar
14. Andrews CJ: Structural changes after lightning strike, with special emphasis on special sense orifices as portals of entry. Semin Neurol 1995; 15:296–303Google Scholar
15. Morse MS, Berg JS, TenWolde RL: Diffuse electrical injury: A study of 89 subjects reporting long-term symptomatology that is remote to the theoretical current pathway. IEEE Trans Biomed Eng 2004; 51:1449–1459Google Scholar
16. Pliskin NH, Fink J, Malina A, et al: The neuropsychological effects of electrical injury. Annals of the New York Academy of Science 1999; 888:140–149Google Scholar
17. Fan K, Zhu Z, Den Z: An experimental model of an electrical injury to the peripheral nerve. Burns 2005; 31:731–736Google Scholar
18. Abramov GS, Bier M, Capelli-Schellpfeffer M, et al: Alteration in sensory nerve function following electrical shock. Burns 1996; 22:602–606Google Scholar
19. Hurley RA, McGowan JC, Arfanakis K, et al: Traumatic axonal injury: Novel insights into evolution and identification. J Neuropsychiatry Clin Neurosci 2004; 16:1–7Google Scholar
20. Zuberi M, Liu-Snyder P, ul Haque A, et al: Large naturally produced electric currents and voltage traverse damaged mammalian spinal cord. J Biol Eng 2008; 2:17Google Scholar
21. Sersa G, Miklavcic D, Cemazar M, et al: Electrochemotherapy in treatment of tumours. Eur J Surg Oncol 2008; 34:232–240Google Scholar
22. Rubinsky B: Irreversible electroporation in medicine. Technol Cancer Res Treat 2007; 6:255–260Google Scholar
23. Barrash J, Kealey GP, Janus TJ: Neurobehavioral sequelae of high voltage electric injuries: comparison with traumatic brain injury. Appl Neuropsychol 1996; 3:75–81Google Scholar
24. Aslan S, Yilmaz S, Karioglu O: Lightning: an unusual cause of cerebellar infarction. Emerg Med J: 2004; 21:750–751Google Scholar
25. Kleiter I, Luerding R, Diendorfer G, et al: A lightning strike to the head causing a visual cortex defect with simple and complex visual hallucinations. J Neurol Neurosurg Psychiatry 2007; 78:423–426Google Scholar
26. Ozgun B, Castillo M: Basal ganglia hemorrhage related to lightning strike. AJNR Am J Neuroradiol 1995; 16:1370–1371Google Scholar
27. Scholz T, Rippmann V, Wojtecki L, et al: Severe brain damage by current flow after electrical burn injury. J Burn Care Res 2006; 27:917–922Google Scholar
28. Kalita J, Jose M, Misra UK: Myelopathy and amnesia following accidental electrical injury. Spinal Cord 2002; 40:253–255Google Scholar
29. Caksen H, Yuca SA, Demirtas I, et al: Right thalamic hemorrhage resulting from high-voltage electrical injury: a case report. Brain Dev 2004; 26:134–136Google Scholar
30. Cherington M: Spectrum of neurologic complications of lightning injuries. NeuroRehabilitation 2005; 20:3–8Google Scholar
31. Andrews CJ: Further documentation of remote effects of electrical injuries, with comments on the place of neuropsychological testing and functional scanning. IEEE Trans Biomed Eng 2006; 53:2102–2113Google Scholar
32. Primeau M: Neurorehabilitation of behavioral disorders following lightning and electric trauma. NeuroRehabilitation 2005; 20:25–33Google Scholar



