New Techniques for Understanding Huntington's Disease
Huntington's disease (HD) is a progressive neurodegenerative disorder that is inherited in an autosomal dominant fashion. It occurs in approximately 3 to 8 per 100,000 population in the United States. HD has been reported in all races and cultures, but it occurs less in individuals of African or Japanese descent. HD was first formally described by George Huntington in 1872 and has been studied as a classic neurodegenerative disorder since that time. An excellent recent review summarizes historical, diagnostic, and clinical aspects of the disease.1 Folstein's text provides a more comprehensive treatment of this subject.2 In brief, HD presents with either an adult or a juvenile onset. The adult form is more common, with onset between 35 and 45 years of age and progression to death within 15 to 20 years. The juvenile form (usually paternally transmitted) presents before age 20 and has a more rapid progression. Although most clinicians are aware of the classic motor findings in HD (involuntary choreiform movements, dysarthrias, dystonias, and rigidity), the psychiatric and cognitive findings, almost always present, are often overlooked.
All patients with HD develop a progressive subcortical dementia that is characterized by frontal lobe executive dysfunction and memory deficits. Neuropsychological testing reveals deficits in recent and remote memory; impaired visuospatial function; difficulty with shifting sets, planning, and organization; and overall decreasing IQ. Psychiatric syndromes (present in up to 79% of patients) most commonly include impulse control disorders, depression, personality changes, and, more rarely, psychosis or mania. Symptoms include disinhibition, irritability, aggression, apathy, and neurovegetative markers of depression. The suicide rate has been reported to be up to 20 times that of the general population over age 50.1 Increased criminal behavior and hypersexuality have often been reported in HD patients. A recent Danish study found that there is a statistically significant increase in nonviolent crime in male HD patients compared with their nonaffected relatives and control subjects.3 These criminal behaviors, as well as the psychiatric syndromes, are felt to be related to the destruction of the medial caudate, which disrupts the memory and emotion tracts descending from the frontal lobes.1,4
In 1993, the Huntington's Disease Collaborative Research Group discovered the genetic defect responsible for HD. It is a long repetition of the trinucleotide CAG (cytosine-adenosine-guanosine) sequence in the first exon of a gene on chromosome 4. This repeating sequence leads to the production of a protein called huntingtin. Accumulation of this protein is theorized to lead to nuclear inclusions and cell death in medium spiny GABAergic neurons of the caudate.5 Currently, probability studies are under way to coordinate the number of CAG repeats with expected age at onset, symptoms, and length of survival.6
Several methods of studying brain functioning, including magnetic resonance spectroscopy (MRS), single-photon emission computed tomography (SPECT), and positron emission tomography (PET) have been applied to HD. MRS provides a relative measure of certain brain metabolites, most commonly presented as the spectrum of the amount of signal produced by the metabolites being measured rather than as an image. MRS uses the same scanners and magnets as traditional MR imaging, but with special hardware and software that allow substances other than water to be studied. Using 1H (proton) MRS, several groups have found decreased levels of N-acetylaspartate (NAA, a neuronal marker) in the cerebrum of symptomatic gene-positive patients.7–12 This change was particularly common in the occipital cortex and striatum.7,9,11,12 Some have also reported increased levels of lactate (LA; an indication of metabolic distress), although this is still controversial.13 Increased LA was asymmetric in the striatum, with levels in the left hemisphere exceeding levels in the right.7 The degree of decrease in NAA and increase in LA in the striatum has been shown to correlate with the duration of symptoms.7 Experimental treatment with coenzyme Q10 resulted in significant decreases in cortical LA in symptomatic patients,11 indicating a possible defect in energy metabolism. Asymptomatic carriers typically are characterized by normal 1H MRS spectra, although elevated LA has been reported in some.7,8 A study using 31P (phosphorus) MRS found a decrease in the phosphocreatine (PCR) to inorganic phosphate (Pi) ratio in the resting calf muscle of symptomatic patients, another indication of a defect in energy metabolism.
In both SPECT and PET, radioactive tracers are used to measure quantities such as cerebral blood flow, glucose metabolism, and receptor density. It has been known since the 1980s that these methods can demonstrate reductions in caudate glucose metabolism (by PET scanning) and caudate blood flow (by PET and SPECT scanning) in patients with HD prior to clear evidence of structural changes on magnetic resonance imaging (MRI) or computed tomography (CT). More recent studies have suggested that reduced putamen volume can also be seen quite early in the disease by using MRI.14 Cortical damage can also be demonstrated earlier in the course of the disease on SPECT or PET than on MRI, as is the case with other dementias, such as Alzheimer's disease.15 There is a good correlation between the reductions in striatal and cortical blood flow and the degree and type of neuropsychological impairment, indicating that SPECT scans may be useful in assessing the degree of neuronal damage and disease progression.16 PET scanning with radiolabeled neuroreceptor ligands, such as the D2 ligand carbon-11 raclopride, has revealed markedly reduced dopamine receptor density in the striatum of HD patients.17 D2 radioligands for SPECT imaging have shown similar results and may soon be available for routine clinical use. These results are especially interesting clinically, given that SPECT, unlike PET, is now available at nearly all medical centers.
The role that these new approaches to HD will play in clinical management is not yet clear. However, the results obtained thus far with functional brain imaging demonstrate the potential usefulness of these modalities for evaluating both asymptomatic and symptomatic HD patients, particularly in monitoring both disease progression and the effects of therapy.
To date, there is no treatment for Huntington's disease. Typical antipsychotics such as fluphenazine and haloperidol have been used to decrease the choreiform movements early in the course of the disease, but they produce significant side effects, including tardive dyskinesia and worsening cognition. There are limited reports of improvements with both clozapine and electroconvulsive therapy. However, the most innovative and potentially useful treatment is neuronal cell transplantation via stereotaxic injection.18 This method is ethically controversial because embryonic donors provide the only viable source of stem cells for this therapy. Extensive research is currently under way to develop new strategies to grow early stem cells in the laboratory.
The techniques discussed here are certainly exciting and are leading to a new understanding of Huntington's disease and possible treatments. The potential now exists to apply these concepts to other neurodegenerative diseases.
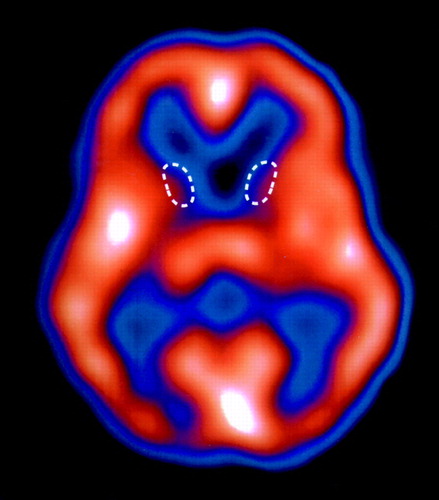
ATransaxial slice of a single-photon emission computed tomography (SPECT) brain perfusion scan from a 23-year-old male with juvenile-onset, genetically proven Huntington's disease. The patient was placed in a darkened, quiet room, where he received 25 mCi of [99mTc]HMPAO intravenously prior to SPECT imaging of the brain. Areas of highest tracer uptake appear white/orange (high blood flow); lowest uptake is blue/black (low blood flow). Note the markedly reduced uptake in the caudate nuclei bilaterally (outlined by white dashed lines). The adjacent thalami are normal. Activity in the cortex is essentially normal in this patient, but it typically becomes abnormal (reduced perfusion) as the disease progresses. High activity in the visual cortex is secondary to visual stimulation the patient received in the partially darkened room. This patient presented with a history of progressive dyskinetic and choreiform movements, hyperkinesia, dysarthria, anger dyscontrol, very inappropriate speech, depressed mood, and episodic crying. The dyskinesias first appeared at age 16.
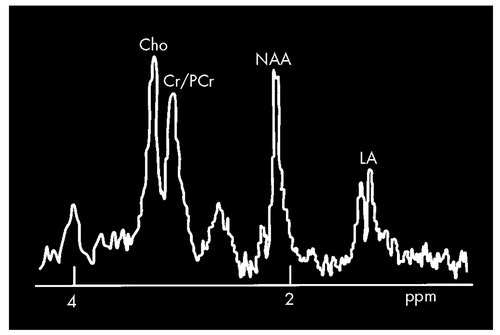
BSchematic drawing of a short-echo-time proton magnetic resonance spectrum (MRS) illustrating the most common findings in symptomatic gene-positive Huntington's disease patients, particularly from regions in the left striatum and occipital cortex. The lactic acid (LA) peak, which is not detectable in normal subjects, is typically elevated, and the N-acetylaspartate (NAA) peak is typically substantially decreased. (At this echo time, the NAA peak should be approximately a factor of 1.6 times that shown.) Normal peaks labeled for orientation in the spectrum include the mixed creatine/phosphocreatine peak (Cr/PCr) and choline (CHO).
1 Haddad MS, Cummings JL: Huntington's disease. Psychiatr Clin North Am 1997; 20:791–807Crossref, Medline, Google Scholar
2 Folstein SE: Huntington's Disease: A Disorder of Families. Baltimore, John Hopkins University Press, 1989Google Scholar
3 Jensen P, Fenger K, Bolwig TG, et al: Crime in Huntington's disease: a study of registered offenses among patients, relatives, and controls. J Neurol Neurosurg Psychiatry 1998; 65:467–471Crossref, Medline, Google Scholar
4 Burrus JW, Hurley RA, Taber KH, et al: A functional neuroanatomy of the frontal lobe circuits. Radiology (in press)Google Scholar
5 Walling HW, Baldassare JJ, Westfall TC: Molecular aspects of Huntington's disease. J Neurosci Res 1998; 54:301–308Crossref, Medline, Google Scholar
6 Brinkman RR, Mezei MM, Theilmann J, et al: The likelihood of being affected with Huntington disease by a particular age, for a specific CAG size. Am J Hum Genet 1997; 60:1202–1210Google Scholar
7 Jenkins BG, Rosas HD, Chen YC, et al: 1H NMR spectroscopy studies of Huntington's disease: correlations with CAG repeat numbers. Neurology 1998; 50:1357–1365Google Scholar
8 Harms L, Meierkord H, Timm G, et al: Decreased N-acetyl-aspartate/choline ratio and increased lactate in the frontal lobe of patients with Huntington's disease: a proton magnetic resonance spectroscopy study. J Neurol Neurosurg Psychiatry 1997; 62:27–30Crossref, Medline, Google Scholar
9 Taylor-Robinson SD, Weeks RA, Bryant DJ, et al: Proton magnetic resonance spectroscopy in Huntington's disease: evidence in favor of the glutamate excitotoxic theory. Mov Disord 1996; 11:167–173Crossref, Medline, Google Scholar
10 Tsai G, Coyle JT: N-acetylaspartate in neuropsychiatric disorders. Prog Neurobiol 1995; 46:531–540Crossref, Medline, Google Scholar
11 Koroshetz WJ, Jenkins BG, Rosen BR, et al: Energy metabolism defects in Huntington's disease and effects of coenzyme Q10. Ann Neurol 1997; 41:160–165Crossref, Medline, Google Scholar
12 Jenkins BG, Koroshetz WJ, Beal MF, et al: Evidence for impairment of energy metabolism in vivo in Huntington's disease using localized 1H NMR spectroscopy. Neurology 1993; 43:2689–2695Google Scholar
13 Hoang TQ, Bluml S, Dubowitz DJ, et al: Quantitative proton-decoupled 31P MRS and 1H MRS in the evaluation of Huntington's and Parkinson's diseases. Neurology 1998; 50:1033–1040Google Scholar
14 Harris GJ, Aylward EH, Peyser CE, et al: Single photon emission computed tomographic/blood flow and magnetic resonance volume imaging of basal ganglia in Huntington's disease. Arch Neurol 1996; 53:316–324Crossref, Medline, Google Scholar
15 Sax DS, Powsner R, Kim A, et al: Evidence of cortical metabolic dysfunction in early Huntington's disease by single-photon-emission computed tomography. Mov Disord 1996; 11:671–677Crossref, Medline, Google Scholar
16 Rauch SL, Savage CR: Neuroimaging and neuropsychology of the striatum. Psychiatr Clin North Am 1997; 20:741–768Crossref, Medline, Google Scholar
17 Andrews TC, Brooks DJ: Advances in the understanding of early Huntington's disease using the functional imaging techniques of PET and SPET. Mol Med Today 1998; 4:532–539Crossref, Medline, Google Scholar
18 Dunnett SB, Kendall AL, Watts C, et al: Neuronal cell transplantation for Parkinson's and Huntington's diseases. Br Med Bull 1997; 53:757–776Crossref, Medline, Google Scholar



