Prevention of Post-Stroke Generalized Anxiety Disorder, Using Escitalopram or Problem-Solving Therapy
Abstract
This study examined the efficacy of antidepressant treatment for preventing the onset of generalized anxiety disorder (GAD) among patients with recent stroke. Of 799 patients assessed, 176 were randomized, and 149 patients without evidence of GAD at the initial visit were included in this double-blind treatment with escitalopram (N=47) or placebo (N=49) or non-blinded problem-solving therapy (PST; 12 total sessions; N=53). Participants given placebo over 12 months were 4.95 times more likely to develop GAD than patients given escitalopram and 4.00 times more likely to develop GAD than patients given PST. Although these results should be considered preliminary, the authors found that both escitalopram and PST were effective in preventing new onset of post-stroke GAD.
Patients who have survived a stroke are an ideal group for selective prevention (i.e., prevention among individuals at increased risk for developing illness),1 for generalized anxiety disorder (GAD), because the prevalence of GAD in the first 6 months after stroke is 21.3%. Although GAD is often comorbid with depression, the prevalence of GAD without depression is still 10.7%2
We have previously demonstrated that post-stroke depression was selectively prevented by administration of escitalopram (10 mg/day <age 65, 5 mg ≥age 65) or problem-solving therapy (PST) over 1 year, as compared with placebo.3 We examined our prevention study data3 for development of new-onset GAD during 1 year of treatment with escitalopram, problem-solving therapy (PST), or placebo. We hypothesized that both escitalopram and PST, as compared with placebo, would constitute effective preventive interventions for post-stroke GAD.
Methods
Patients
Patients were enrolled within 3 months of an index stroke between July 9, 2003, and October 1, 2007, from the Department of Neurology, University of Iowa, Iowa City; the Department of Neurology, University of Chicago; and Burke Rehabilitation Hospital, White Plains, NY, and also recruited through newspaper advertisements. Protocols were approved by the institutional review boards at each study site, and written informed consent was obtained from each participant. A total of 799 patients were evaluated for eligibility, and 200 signed an informed consent: 28 from Burke Rehabilitation Hospital, 24 from University of Chicago, and 148 from University of Iowa. There were 149 patients randomized to treatment (Figure 1).
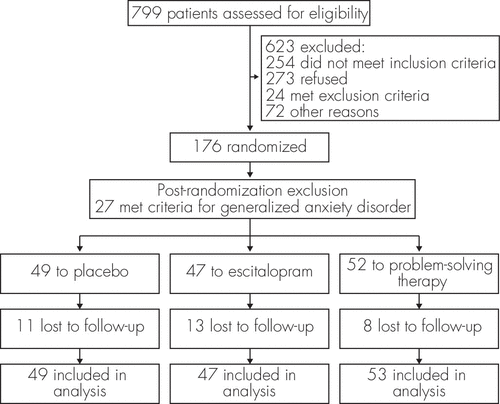
FIGURE 1. A Schematic Display of the Flow of Patients Included in This Study
As described in previous publication,3 inclusion criteria included age (>50 years and <90 years) with clinical and neuroradiological findings consistent with either ischemic or hemorrhagic stroke. Patients were excluded if they met DSM-IV diagnostic criteria for major or minor depressive disorder or GAD at intake (excluding the 6-month duration criterion; N=27). Other exclusion criteria included severe comprehension deficits, demonstrated by failing one or more items on Part 1 of the Token Test;4 or stroke secondary to complications from other vascular disease or medical illness. Thus, data from 149 subjects are included in this analysis (Figure 1).
Neurological and Neuroradiological Evaluations
A complete physical and neurological examination were performed at intake. Vital signs and body weight were recorded at baseline, 12 weeks, and 6-, 9-, and 12-month follow-ups. There was no standardized imaging protocol.
Experimental Design and Treatment
Patients were centrally randomized by a team member uninvolved in any evaluation, using permuted block sizes of 3, 6, and 9. Patients were assigned to escitalopram (10 mg/day in the morning for patients ≤65 years and 5 mg/day for patients >65 years), placebo (all pills were identical), or PST. Escitalopram was selected because it is indicated for treatment of anxiety disorder as well as depression.5,6
PST is a manual-based intervention that was selected because it has been modified for use in medically ill elderly patients with depression.7,8 PST consisted of six treatment sessions over Weeks 1, 2, 3, 4, 6, and 10, and six reinforcement sessions (Months 4, 5, 6, 8, 10, and 12) using a protocol described in a previous publication.3 All therapy sessions were videotaped for review of consistency and fidelity of administration. Patients were seen for evaluation by raters who were blinded to drug assignment and not involved in administration of PST at 3, 6, 9, and 12 months. Patients receiving PST were aware of their treatment.
Assessment Instruments and GAD Diagnosis
Patients were administered the Structured Clinical Interview for DSM-IV (SCID)9 at initial evaluation and 3-, 6-, 9-, and 12-month follow-up. Diagnosis of GAD was based on symptoms elicited by the SCID during the 3 months since the last evaluation and applied to DSM-IV criteria for GAD, excluding the requirement of 6-month duration. The Hamilton Rating Scale for Anxiety (Ham–A)10 has been shown to be valid and reliable in patients with stroke.11,12 At each follow-up interview, or if a patient reported a problem with anxiety, the patient was examined for GAD. Socioeconomic status was determined by the Hollingshead and Redlich classification.13
The Functional Independence Measure (FIM)14 was used to assess activities of daily living (ADL) at baseline . The FIM is an 18-item, 7-level, ADL scale, assessing interpersonal, familial, and occupational functioning. The maximum score is 126 points; higher scores indicate less impairment. The FIM has been shown to be valid and reliable among patients with stroke.14 The Social Functioning Exam15 is a 28-item scale that assesses patients’ satisfaction with their social functioning during the 2 weeks before each examination. Scores on the Social Functioning Exam range from 0 to 100, with higher scores indicating greater severity of social impairment. Reliability and validity of this instrument have been demonstrated in a previous publication.16
Adverse Events
Patients, family members, and primary care physicians were asked about medication adverse effects (specifically, gastrointestinal, sexual, and cardiovascular) at 3-month intervals, using a standardized checklist developed for this study. A data and safety monitoring board, consisting of investigators not involved in this trial, assessed adverse events related to the three treatments.
Statistical Analysis
Categorical data were analyzed with Fisher’s exact test. For continuous measures, the Kruskal-Wallis test was used to compare all groups. In order to analyze the time to the onset of GAD, a proportional-hazards Cox regression model was used. Various baseline characteristics, such as age, were considered as possible confounders. Two-way interactions between covariates and treatment group were considered. The final model was chosen by Akaike’s information criterion. The robust sandwich estimate of Lin and Wei17 for the covariance matrix was used for inference to account for the correlation of each data-point within each site. Responses of participants in the same site were correlated. When standard errors were calculated, the within-site correlation was considered. The proportional-hazards assumption was assessed and met. This analysis included patients who completed the study as well as those who dropped out after randomization. Patients who dropped out after randomization were considered in the model as censored observation at the time they stopped participating in the study. To investigate the effect of treatment on secondary outcomes, the FIM, and the Social Functioning Exam, we used a linear mixed model that included secondary outcome scores as the dependent variable, and Treatment Group, Time, and the interaction between Group and Time as explanatory variables. All analyses were performed with R 2.5.1 (R Foundation for Statistical Computing; Vienna, Austria) and SAS Version 9.2 for Windows (SAS Institute Inc., Cary, NC). All p values reported are two-tailed. Significance level was set at p<0.05.
Results
Participants and Baseline Measures
The patient flow diagram is shown in Figure 1. With the exception of age and frequency of diabetes mellitus, there were no significant differences in demographic or baseline variables among the three treatment groups or between those who dropped out and those who completed the study (Table 1).
| Placebo (N=49) | Escitalopram (N=47) | PST (N=53) | |
|---|---|---|---|
| Age, mean (SD), yearsa | 64.8 (13.5) | 61.5 (13.7) | 68.3 (10.4) |
| Male, % | 67.3 | 63.8 | 54.7 |
| Married, % | 59.2 | 53.2 | 47.2 |
| Education, mean (SD), years | 13.4 (2.9) | 13.4 (3.1) | 14.1 (2.9) |
| Socioeconomic Class IV, Vb, % | 26.5 | 19.1 | 24.5 |
| Previous history of GAD, % | 6.1 | 6.4 | 5.7 |
| Baseline Hamilton Rating Scale for Anxiety score, mean (SD) | 6.8 (4.4) | 7.1 (5.6) | 8.3 (5.4) |
| Baseline Functional Independence Measure (FIM) score, mean (SD) | 114.9 (20.7) | 115.8 (16.6) | 114.5 (13.7) |
| Baseline Social Functioning Exam score, mean (SD) | 0.13 (0.12) | 0.12 (0.09) | 0.09 (0.09) |
| Stroke characteristics, % | |||
| Hemorrhagic stroke | 8.2 | 4.3 | 13.2 |
| Ischemic stroke | 91.8 | 95.7 | 86.8 |
| Large-artery atherosclerosis | 30.6 | 36.2 | 20.8 |
| Small-artery occlusion | 32.7 | 17.0 | 30.2 |
| Cardioembolism/Other | 28.6 | 42.6 | 35.8 |
| Left-side lesion | 51.0 | 48.9 | 41.5 |
| Medical comorbidity | |||
| Systolic blood pressure, mean (SD), mm Hg | 144.0 (24.4) | 139.3 (24.1) | 146.9 (20.6) |
| Low-density lipoprotein cholesterol, mean (SD), mg/dl | 122.5 (47.7) | 116.3 (37.0) | 109.9 (38.0) |
| Cumulative Illness Rating Scale total score, mean (SD) | 9.7 (4.6) | 10.2 (5.4) | 9.5 (4.8) |
| Diabetes mellitusc, % | 20.4 | 42.6 | 17.0 |
| Coronary artery disease, % | 22.4 | 25.5 | 24.5 |
| Cognitive heart failure, % | 6.1 | 14.9 | 13.2 |
| Atrial fibrillation, % | 14.3 | 14.9 | 22.6 |
TABLE 1. Characteristics of Post-Stroke Patients Randomized to Receive Placebo, Escitalopram, or Problem-Solving Therapy (PST)
Effect of Preventive Intervention on GAD
Using 149 patients randomized to treatment, we built a Cox proportional-hazards model using time-to-GAD onset as the dependent variable. Covariates included in the model were age, gender, previous history of GAD, and FIM. After adjusting for covariates, participants receiving placebo (9 cases of GAD; total: 18.4%) were 4.95 times more likely to develop GAD than patients receiving escitalopram (2 cases of GAD; total: 4.3%) and 4.00 times more likely than patients who received PST (3 cases of GAD; total: 5.7%; Table 2; Figure 2). On the basis of these findings, the number needed to treat (NNT) for the combined escitalopram-and-PST group was 7.46 (95% confidence interval [CI]: 4.62–33.91) and for escitalopram-alone was 7.09 (95% CI: 4.31–94.08).
| Adjusted Hazard Ratio (HR) | 95% Confidence Interval (CI) for Hazard Ratio (HR) | p | |
|---|---|---|---|
| Placebo versus escitalopram | 4.95 | 1.54–15.93 | 0.0073 |
| Placebo versus PST | 4.00 | 1.84–8.70 | 0.0005 |
| Escitalopram versus PST | 0.81 | 0.12–5.26 | NS |
| Age | 1.03 | 1.01–1.06 | 0.0097 |
| Previous history of GAD | 3.59 | 7.74–4.64 | 0.0005 |
| Female vs. male | 1.39 | 0.59–3.07 | NS |
| Baseline Functional Independence Measure score | 1.03 | 0.96–1.10 | NS |
TABLE 2. The Cox Hazard Ratio (HR) Model Results for Risk Comparison of GAD Onset
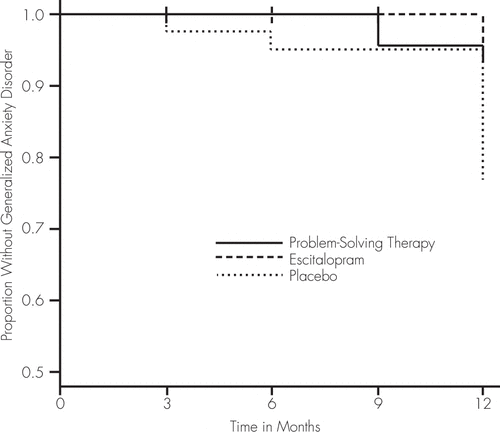
FIGURE 2. Kaplan-Meier Plot of Generalized Anxiety Disorder (GAD) Onset for Patients Receiving Placebo, Escitalopram, or Problem-Solving Therapy (PST) Over 1 Year
After adjusting for age, gender, previous history of GAD, and the Functional Independence Measure, participants who received placebo were 4.95 times more likely to develop GAD than patients who received escitalopram (adjusted hazard ratio [HR]: 4.95; 95% confidence interval [CI]: 1.54–15.93; p=0.0073) and 4.00 times more likely than patients who had PST (adjusted HR: 4.00; 95% CI: 1.84–8.70; p=0.0005).
Among patients with GAD, there were 7 who also developed comorbid depression. As a subgroup analysis, we excluded patients with comorbid depression and conducted a Cox proportional-hazards model (HR) analysis using time to GAD-only onset as the dependent variable. There were 7 patients with GAD without depression: 5 received placebo, 0 got escitalopram, and 2 got PST. To increase the power of our analysis, we combined the escitalopram and PST subjects into a single treatment group. Similar to our previous finding, after adjusting for the same covariates as the above analysis, participants receiving placebo were 6.63 times more likely to develop GAD than patients who were treated (adjusted HR: 6.63; 95% CI: 2.85–15.40; p<0.0001).
Adverse Events and Adverse Effects
There were no significant differences among groups in frequency of any of adverse events. Also, there were no significant differences among groups in frequency of hospital admissions.
Discussion
This study demonstrated, over the first year after an acute stroke, that development of GAD can be effectively prevented by either escitalopram or PST.
Before discussing implications of these findings, the limitations of this study need to be acknowledged. First, patients selected for the study did not include all patients with acute stroke. Patients with life-threatening comorbid physical illness, such as cancer or severe cardiac arrhythmia, were excluded, as well as patients with severe impairment in verbal comprehension. Thus, our findings might not be applicable to all patients with stroke. As noted in Table 1, we included patients with multiple system illnesses and a representative range of stroke mechanisms and severity. Second, the study had a relatively small sample size, and the number of incident GAD cases was also relatively small. Thus, our findings should be considered preliminary, and further studies of prevention of post-stroke anxiety are needed. Finally, our psychological treatment group could not be blinded.
Given these limitations, what are the implications of these findings? The most important question may be why it is important to prevent GAD among stroke patients. Patients in this study were elderly, and late-life onset anxiety disorder is frequently associated with physical disability,18 memory impairment,19,20 poor quality of life,18,21,22 increased risk of medical illness,23 and increased use of healthcare resources.18 Furthermore, a randomized, controlled study found that non–brain-injured older adults with GAD who received escitalopram had significantly greater response rate for reduction of anxiety symptoms and role-functioning, as compared with patients who received placebo over 12 weeks.24 Thus, prevention of late-life-onset GAD may have augmented recovery (although this was not demonstrated by FIM scores), improved quality of life, and decreased morbidity.
Another important question is: what mechanism allowed escitalopram or PST to effectively prevent GAD? Although the mechanism is not known, it seems likely to be different for escitalopram and PST. In a previous study, compared with placebo, escitalopram 20 mg/day significantly reduced the risk of relapse in non–brain-injured patients with GAD during 24–76 weeks after 12 weeks of treatment.25 Antidepressants have been associated with enhanced recovery in executive functioning26 and enhanced recovery from memory impairment,27 as well as increased neuroplasticity in several brain regions, including the dentate gyrus of the hippocampus and the prefrontal cortex.28
Problem-solving therapy was as effective as escitalopram for prevention of post-stroke GAD, but there is no evidence that PST enhances neuroplasticity or improves cognitive recovery from stroke; in fact, PST patients had less cognitive recovery than placebo patients.27 Rovner et al.29 reported that PST was effective in preventing depression over 8 weeks in older adults with macular degeneration and speculated that PST may prevent depression by facilitating the ability of these elderly patients to maintain or regain valued activities. Recent studies have shown that PST in elderly patients leads to effective treatment of depression30 and reduction in maladaptive, avoidant coping.31 Avoidant behavior is a risk factor for onset and maintenance of anxiety disorders, including GAD, and, therefore, reduction of avoidant behavior might have been an important factor among our group of stroke patients. Van’t Veer-Tazelaar et al.32 also demonstrated that PST, as part of a stepped-care program, prevented both depression and anxiety disorders (including GAD and other anxiety disorders) in elderly subjects without major physical illness.
To our knowledge, however, this is the first demonstration of specific selective prevention of first-episode GAD in a randomized, controlled trial. The clinical implications of our findings are that patients given escitalopram or PST after acute stroke may be prevented from developing GAD and perhaps its adverse consequences.
1 : Reducing Risks for Mental Disorders: Frontiers for Preventive Intervention Research. Edited by Mrazek PJ, Haggerty RJ. Washington, DC, National Academy Press, 1994Google Scholar
2 : The Clinical Neuropsychiatry of Stroke. Cambridge, UK, Cambridge University Press, 2006Crossref, Google Scholar
3 : Escitalopram and problem-solving therapy for prevention of poststroke depression: a randomized controlled trial. JAMA 2008; 299:2391–2400Crossref, Medline, Google Scholar
4 : The token test: a sensitive test to detect receptive disturbances in aphasics. Brain 1962; 85:665–678Crossref, Medline, Google Scholar
5 : Efficacy comparison of escitalopram and citalopram in the treatment of major depressive disorder: pooled analysis of placebo-controlled trials. CNS Spectr 2002; 7(Suppl 1):40–44Crossref, Medline, Google Scholar
6 : Escitalopram and paroxetine in the treatment of generalised anxiety disorder: randomised, placebo-controlled, double-blind study. Br J Psychiatry 2006; 189:264–272Crossref, Medline, Google Scholar
7 : Effectiveness of problem-solving therapy for older, primary care patients with depression: results from the IMPACT project. Gerontologist 2008; 48:311–323Crossref, Medline, Google Scholar
8 : Predictors of response to problem-solving treatment of depression in primary care. Behav Ther 2002; 33:511–527Crossref, Google Scholar
9 : Structured Clinical Interview for DSM-IV (SCID). New York, Biometric Research, New York State Psychiatric Institute, 1995Google Scholar
10 : The assessment of anxiety states by rating. Br J Med Psychol 1959; 32:50–55Crossref, Medline, Google Scholar
11 : Treatment of poststroke generalized anxiety disorder comorbid with poststroke depression: merged analysis of nortriptyline trials. Am J Geriatr Psychiatry 2003; 11:320–327Crossref, Medline, Google Scholar
12 : Effects of anxiety disorder on impairment and recovery from stroke. J Neuropsychiatry Clin Neurosci 1998; 10:34–40Link, Google Scholar
13 : Social Class and Mental Illness: A Community Study. New York, Wiley, 1958Crossref, Google Scholar
14 : Functional Independence Measure. Buffalo, NY, The Buffalo General Hospital State University of New York at Buffalo, 1987Google Scholar
15 : The social functioning exam: an assessment for stroke patients. Soc Work Res Abstr 1982; 18:28–33Crossref, Medline, Google Scholar
16 : Reliability, validity, and clinical utility of the social functioning exam in the assessment of stroke patients. Exp Aging Res 1983; 9:101–106Crossref, Medline, Google Scholar
17 : The robust inference for the Cox proportional-hazards model. J Am Stat Assoc 1989; 84:1074–1079Crossref, Google Scholar
18 : The burden of late-life generalized anxiety disorder: effects on disability, health-related quality of life, and healthcare utilization. Am J Geriatr Psychiatry 2009; 17:473–482Crossref, Medline, Google Scholar
19 : Cognitive impairment in late-life generalized anxiety disorder. Am J Geriatr Psychiatry 2007; 15:673–679Crossref, Medline, Google Scholar
20 : Cognitive errors, symptom severity, and response to cognitive behavior therapy in older adults with generalized anxiety disorder. Am J Geriatr Psychiatry 2007; 15:680–689Crossref, Medline, Google Scholar
21 : Cognitive-behavioral treatment of late-life generalized anxiety disorder. J Consult Clin Psychol 2003; 71:309–319Crossref, Medline, Google Scholar
22 : Quality of life in geriatric generalized anxiety disorder: a preliminary investigation. J Psychiatr Res 2004; 38:305–312Crossref, Medline, Google Scholar
23 : Generalized anxiety disorder and medical illness. J Clin Psychiatry 2009; 70(Suppl 2):20–24Crossref, Medline, Google Scholar
24 : Escitalopram for older adults with generalized anxiety disorder: a randomized, controlled trial. JAMA 2009; 301:295–303Crossref, Medline, Google Scholar
25 : Prevention of relapse in generalized anxiety disorder by escitalopram treatment. Int J Neuropsychopharmacol 2006; 9:495–505Crossref, Medline, Google Scholar
26 : Effect of antidepressant therapy on executive function after stroke. Br J Psychiatry 2007; 190:260–265Crossref, Medline, Google Scholar
27 : Escitalopram and enhancement of cognitive recovery following stroke. Arch Gen Psychiatry 2010; 67:187–196Crossref, Medline, Google Scholar
28 : Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 2003; 301:805–809Crossref, Medline, Google Scholar
29 : Preventing depression in age-related macular degeneration. Arch Gen Psychiatry 2007; 64:886–892Crossref, Medline, Google Scholar
30 : Problem-solving therapy and supportive therapy in older adults with major depression and executive dysfunction. Am J Psychiatry 2010; 167:1391–1398Crossref, Medline, Google Scholar
31 : Problem-solving treatment and coping styles in primary care for minor depression. J Consult Clin Psychol 2008; 76:933–943Crossref, Medline, Google Scholar
32 : Stepped-care prevention of anxiety and depression in late life: a randomized, controlled trial. Arch Gen Psychiatry 2009; 66:297–304Google Scholar



