Neuropsychiatric Symptoms and Interleukin-6 Serum Levels in Acute Stroke
Abstract
The role of interleukin−6 (IL−6) as a risk factor for developing depressive symptoms, neuropsychological impairment, and related functional and neurological symptom severity during the acute phase of ischemic stroke is still underexplored. Here, the authors assessed this issue, in 48 patients without significant clinical history for major medical illnesses or other factors that promote inflammation, 72 hours after a first-ever acute ischemic stroke. In the acute phase of ischemic stroke, increased IL−6 plays a key role in the onset of depressive disorders, apathy/amotivation, somatic symptoms of depression, and neurological/functional symptoms, resulting in higher disability and poor outcome of stroke patients.
Ischemic stroke is associated with high rate of cognitive1,2 and neuropsychiatric3–6 disorders that can adversely affect disease outcome.7,8 Most authors agree with the hypothesis that a biological dysfunction in brain networks plays a role in the post-stroke neuropsychiatric symptoms.9
There is evidence that ischemic stroke is intimately linked to inflammatory processes, and proinflammatory cytokines appear to be involved in the pathogenesis of brain ischemia.7,10–12 Moreover, cytokines have been extensively proposed as key factors in the modulation of mood and other psychiatric disorders.13–21 Accordingly, very recent data have demonstrated that cytokine-dependent T-cell dysfunction might have a role in depressive disorders and that polymorphisms in inflammation-related genes are associated with susceptibility to major depression and antidepressant response.22 Also, the hypothesis that proinflammatory cytokines may play a central role as mediators of mood and mood-related disorders even in patients with stroke has been previously formulated.23,24 However, only a limited number of reports suggest a role of neuroinflammation on post-stroke affective phenomenology.13,25–27
The attention of the literature has been recently focused on interleukin 6 (IL−6), a pleiotropic cytokine involved in inflammatory processes and able to induce acute inflammatory responses, as does interleukin 1 (IL−1).28 Although a clear picture of the role of IL−6 in stroke has not yet been elucidated, increased IL−6 production after cerebral ischemia has been described in several clinical studies,10,26 and elevated levels of IL−6 have been correlated with severity and clinical outcomes of stroke.24,26 All the above studies show heterogeneous findings due to different stroke populations and the inclusion and exclusion criteria adopted. Moreover, patient and control groups were often not matched for all demographic variables, and patients with depression were under varying exposure to antidepressant therapy in terms of dosage and duration. Another important limitation is the presence of confounding factors able to influence cytokine production, including immunologically-mediated disorders, chronic inflammatory diseases, cancer, etc. Finally, IL−6 levels were mostly investigated during different phases of the stroke (acute, subacute, or chronic).24,29
The relevance of IL−6 as a prognostic marker of post-stroke clinical outcome and the potential effect of depression on stroke recovery support the need to investigate the hypothesis that IL−6 production, associated with brain ischemic lesion, contributes to the pathogenesis of stroke-related mood, cognitive, and functional disorders, starting from the very acute phase.
Thus, the main aim of our study was to investigate the relationships between IL−6 serum level and severity of neuropsychiatric and functional symptoms, impaired neuropsychological functions, and clinical neurological outcomes in patients with acute ischemic stroke. We have also examined which individual symptoms of depression are associated with IL−6 levels.
Methods
All data were collected in the Stroke Unit of the Sant’Andrea Hospital in Rome, Italy; 48 consecutive patients admitted to the hospital for a first-ever stroke were enrolled in the study. Only patients without significant clinical factors that promote inflammation were selected. Particular attention was given to exclude patients suffering from those diseases that are known to be associated to increased IL−6 production that may be very common in elderly people, such as autoimmune diseases (e.g., Crohn’s disease, rheumatoid arthritis, systemic lupus erythematosus), infections, history of previous stroke, pre-stroke psychiatric and neurological disorders (e.g., pre-stroke depression, Alzheimer’s disease), and cancer. Patients who underwent surgical procedures determining chronic inflammatory reaction (see exclusion criteria) were also excluded. In particular, the inclusion criteria were the following: 1) first acute ischemic stroke diagnosis according to National Institute of Neurological and Communicative Disorders and Stroke (NINCDS) criteria;30 2) vision and hearing sufficient for compliance with testing procedures. Exclusion criteria included the following: 1) severe or moderate cognitive deficit as evaluated by Mini-Mental State Exam (MMSE)31 score <17; 2) aphasia with moderate or severe impairment of language comprehension precluding both the interview and the administration of the questionnaires; 3) previous history of head trauma or other brain diseases; 4) major medical illnesses (e.g., diabetes; obstructive pulmonary disease or asthma; hematologic disorders; clinically significant and unstable active gastrointestinal, renal, hepatic, endocrine, or cardiovascular disorders); 5) history or evidence of any clinically important autoimmune disease or disorder of the immune system; 6) history of cancer within the last 5 years; 7) clinically important infection within the last 30 days (e.g., chronic persistent or acute infection, such as bronchitis or urinary tract infection); 8) implant of carotid or coronary stent or other major surgical interventions; 9) known or suspected lifetime history of alcoholism or drug dependence and abuse; 10) use of anti-inflammatory drugs within the last 30 days (e.g., corticosteroids or nonsteroidal anti-inflammatory drugs); 11) history of psychiatric treatment (e.g., antidepressants, antipsychotics, mood stabilizers, benzodiazepines).
At the admission into the Stroke Unit, an expert neurologist performed a full neurological evaluation of the patients. The ischemic lesion was assessed and confirmed by both the clinical examination and computed tomography (CT) or magnetic resonance imaging (MRI).
In order to evaluate biological markers during the acute phase, one blood sample was obtained about 72 (±12) hours after the acute stroke onset, in the early morning, with the patient fasting. We selected this time-point because several studies have reported that IL−6 levels peak at 3 days after an acute event.32–34 Moreover, in all acute patients admitted to the Stroke Unit at this time-point, it was possible to perform the blood sampling for measurement of the IL−6 levels. Thus, the blood sample collection at 72 hours after the neurological stroke symptom onset may be a good clinical outcome (both from a biological and practical point of view) for this inflammatory marker.
All included patients underwent neuropsychiatric, neuropsychological, functional, and neurological assessments, carried out 3 days (about 72 hours) after the onset of the neurological symptoms of the acute stroke, when the patient was stabilized and we were able to perform the evaluations. Trained specialists (one psychiatrist for the psychiatric diagnoses, three neurologists for the neurological assessment, and two neuropsychologists for the neuropsychological and psychiatric rating scales) who were blind to the aims of the study performed the testing. Evaluation of interrater reliability in this study was in the excellent-to-good range for all scales used, with intraclass correlations ranging from 0.80 to 0.93.
The study was approved by the ethical committees of the IRCCS “Santa Lucia” Foundation and “Sant’Andrea” Hospital, and, in accordance with the Helsinki Declaration, each subject signed an informed consent form before enrollment.
Neuropsychiatric Assessment
All patients underwent a structured psychiatric interview (SCID-P)35 for the identification of mental disorders, according to the Diagnostic and Statistical Manual of Mental Disorders 4th Edition–Text Revision (DSM–IV-TR);36 in particular, the presence of Major Depressive Disorder (MDD) and Minor Depressive Disorder (MiDD) was investigated. The DSM–IV-TR diagnostic criteria both for symptoms (criterion A) and functioning (criterion C) were observed to determine the diagnosis of major and minor depression, although the part of criterion A based on symptoms present during the same 2-week period could not be assessed because of the scheduling of evaluations. In particular, for this part of the criterion A, we initially assessed the first 3 days after the stroke onset, and eventually we confirmed the presence of symptoms for the same 6-day period, from admission to the stroke unit to the last clinical evaluation before discharge from the hospital. Thus, the symptom-duration part of the A criterion was modified, in this study on acute stroke inpatients, from the same 2-week period to the same 6-day period. The SCID–P was also used to measure the frequency of the nine individual symptoms of MDD, as indicated by the symptom criteria (in criterion A) of the DSM–IV-TR, in our stroke patients with and without depression.
Depressive symptom severity was evaluated with the Hamilton Rating Scale for Depression (Ham-D),37 a 17-item inventory composed of psychological (PSY) and somatic (SOM) subscores that together contribute to the total score. The PSY subscale consisted of 6 questions about depressed mood, guilt, suicide, work, loss of interest, anxiety, and insight; the SOM subscale consisted of 11 questions regarding insomnia (initial, middle, and delayed), retardation and agitation, somatic anxiety, somatic gastrointestinal symptoms, general somatic symptoms, genital somatic symptoms, loss of weight, and hypochondriasis.
Severity of anxiety symptoms was measured with the Hamilton Rating Scale for Anxiety (Ham-A).38
Neuropsychological Assessment
Global cognitive functioning was evaluated with the Mini-Mental State Exam (MMSE).30 To assess performance in specific cognitive domains, the patients were administered the Mental Deterioration Battery (MDB),39 a standardized and validated neuropsychological battery including the Rey's 15-word test: Immediate Recall (RIR), Delayed Recall (RDR), and Word Recognition (RWR), to evaluate short- and long-term verbal memory; the Copy Drawings (CD) and Copy Drawings with Landmarks (CDL), to evaluate simple and constructional praxis, and the Stroop Word–Color Test (SWCT)40 for the evaluation of executive functions of attention-shifting and control.
Functional and Neurological Assessment
We used the Barthel Index (BI)41 to evaluate functional abilities and the National Institutes of Health Stroke Scale (NIHSS)42 for the quantitative evaluation of stroke-induced neurological deficit during the acute phase. In particular, the NIHSS is a 15-item neurologic examination stroke scale useful for measuring stroke severity and predicting both short- and long-term outcomes of stroke patients by evaluating the effect of acute cerebral infarction on level of consciousness, language, neglect, visual-field loss, extraocular movement, motor strength, ataxia, dysarthria, and sensory loss. Ratings for each item are scored with 3-to-5 measures, with 0 as normal, and there is an allowance for untestable items.
IL−6 Measurement in Serum
Serum was obtained from all patients at 72 hours after the onset of the acute ischemic stroke symptoms by centrifugation of clotted blood samples; aliquots were stored at −80°C until cytokine assay. IL−6 levels were determined with Quantikine HS High Sensitivity ELISA for Human IL−6 (R&D Systems, Minneapolis, MN). The sensitivity of the assay was 0.039 pg/ml.
Statistical Analysis
Univariate analyses of variance (ANOVAs) followed by Fisher’s protected least significant deviance (PLSD) were performed to assess differences in IL−6 mean values and other continuous variables (the dependent variables) among patients with MDD, MiDD, and no depression (NODEP); diagnostic groups were considered as the independent variable. Fisher’s PLSD test was used for pairwise comparisons in post-hoc analyses.
A series of nine t-tests was performed in order to compare IL−6 values between the two groups of people with and without each of the nine depressive symptoms identified by the SCID-P. In these analyses the level of statistical significance was set at p <0.05 after Bonferroni correction for multiple comparisons (α <0.05/9 number of comparisons − α <0.006).
The predictors of IL−6 levels were determined by means of a series of stepwise multiple-regression analyses, using a forward procedure and an F to enter at 4. Pre-selection of variables to include in the stepwise regression models was done by using correlation analyses and Fisher’s r-to-z transformation in order to determine the significance of correlations for continuous variables. In the stepwise multivariate models, only variables with p<0.05 in the pre-selection analyses were included. The level of statistical significance in these multivariate analyses was set at p <0.05.
Results
Sociodemographic and clinical characteristics of 48 patients with first-ever stroke are summarized in Table 1. The occurrence of depressive disorders was 41.7%. In particular, 6 patients (12.5%) were diagnosed as having MDD; 14 patients (29.2%) had MiDD; and 28 patients (58.3%) did not show mood disorders (NODEP).
| Variable | Mean (SD); range |
|---|---|
| Age, years | 68.2 (14.3); 29–93 |
| Women, N (%) | 22 (46) |
| Educational level, years | 8.6 (4.7); 1–21 |
| IL–6 high sensitivity (pg/ml) | 5.0 (4.8); 0–24 |
| Mini-Mental State Exam score | 26.2 (3.7); 17–30 |
| Ham-D Total score | 7.8 (5.4); 0–22 |
| Ham-D Psychological score | 3.2 (2.2); 0–8 |
| Ham-D Somatic score | 4.7 (3.7); 0–15 |
| Ham-A score | 8.3 (5.3); 1–23 |
| Barthel Index | 72.6 (28.4); 10–100 |
| National Institute of Health Stroke Scale | 2.7 (2.7); 1–11 |
| Rey’s (15-word) Immediate Recall test | 29.6 (12.0); 0–57 |
| Rey’s Delayed Recall | 5.6 (3.5); 0–12 |
| Rey’s Word Recognition, true recognition | 11.9 (3.5); 0–15 |
| Rey’s Word Recognition, false recognition | 7.7 (6.8); 0–27 |
| Stroop Word–Color Test; word reading time, seconds | 23.4 (14.2); 11–88 |
| Stroop Word–Color Test; color naming time, seconds | 31.4 (13.9); 16–88 |
| Stroop Word–Color Test; interference time, seconds | 65.8 (29.1); 23–149 |
| Copying Drawings | 7.7 (3.2); 0–12 |
| Copying Drawings with Landmarks | 50.2 (19.5); 0–70 |
The analysis of variance (ANOVA) showed an increased progression of IL−6 levels (F[2, 45]=5.318; p=0.008) from NODEP (mean [standard deviation {SD}]: 3.39 [2.69]) to MiDD (6.55 [6.94]), to MDD (9.05 [3.01]). In particular, post-hoc analyses revealed that MDD (p=0.006) and MiDD (p=0.033) patients had significantly higher IL−6 levels than NODEP patients (Figure 1).
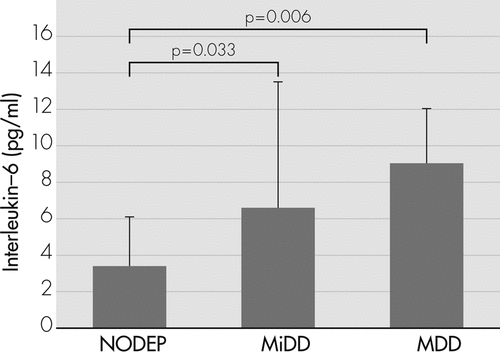
Table 2 shows differences on IL−6 values among patients with or without each depressive symptom identified by the SCID−P; only patients with at least one depressive symptom among the following: loss of interest, loss of weight or appetite, and insomnia, had significantly higher IL−6 values than those without that depressive symptom.
| Symptom | Patients With Symptom | Patients Without Symptom | T | p | ||
|---|---|---|---|---|---|---|
| N | IL–6 (pg/ml) | N | IL–6 (pg/ml) | |||
| Depressed mood | 23 | 6.075 (4.55) | 25 | 4.046 (4.86) | 1.490 | NS |
| Loss of interest | 7 | 9.690 (6.93) | 41 | 4.221 (3.87) | 3.037 | 0.004* |
| Loss of weight/appetite | 5 | 11.3 (7.72) | 43 | 4.288 (3.82) | 3.451 | 0.001* |
| Insomnia | 15 | 8.571 (6.09) | 33 | 3.404 (2.93) | 3.990 | <0.001* |
| Agitation and retardation | 3 | 2.99 (1.53) | 45 | 5.154 (4.89) | 0.757 | NS |
| Fatigability | 18 | 5.386 (5.24) | 30 | 4.798 (4.55) | 0.409 | NS |
| Feelings of guilt | 2 | 6.305 (2.93) | 46 | 4.962 (4.85) | 0.386 | NS |
| Concentration and decision-making problems | 16 | 6.027 (4.67) | 32 | 4.514 (4.82) | 1.036 | NS |
| Suicide | 1 | 2.76 | 47 | 5.066 (4.81) | — | — |
Table 3 shows raw correlations between IL−6 values and cognitive, neuropsychiatric, and clinical variables; IL−6 levels were positively correlated with Ham-D (PSY, SOM, and Total score) and NIHSS scores, and negatively correlated with RWR Correct Recognitions and BI score.
| Variable | IL–6 | |
|---|---|---|
| Pearson r | p | |
| Ham-D Psychological score | 0.39 | 0.0057* |
| Ham-D Somatic score | 0.512 | 0.0001* |
| Ham-D Total score | 0.509 | 0.0002* |
| Ham-A | 0.172 | NS |
| Barthel Index | –0.449 | 0.0012* |
| National Institutes of Health Stroke Scale | 0.602 | <0.0001* |
| Mini-Mental State Exam | –0.066 | NS |
| Rey’s (15-word) Immediate Recall test | –0.170 | NS |
| Rey’s Delayed Recall | –0.195 | NS |
| Rey’s Word Recognition, correct recognitions | –0.288 | 0.0467* |
| Rey’s Word Recognition, false recognitions | 0.224 | NS |
| Stroop Word–Color Test; word reading time, seconds | 0.091 | NS |
| Stroop Word–Color Test; color naming time, seconds | 0.175 | NS |
| Stroop Word–Color Test; interference time, seconds | 0.28 | 0.0536 |
| Copying Drawings | –0.203 | NS |
| Copying Drawings with Landmarks | –0.251 | 0.0854 |
To clarify which variables were independently correlated with the levels of IL−6, we performed multiple stepwise regression analyses. The preselected variables related (p <0.05) to the dependent variable in the univariate correlation analyses (see Table 3) were considered as independent variables. Results of stepwise multiple regression analyses are shown in Table 4: significant predictors of the IL−6 values were Ham-D, SOM, and NIHSS scores. The resulting equation was highly significant (F[2, 45]=22.095; p <0.0001) and explained 49.5% (r2) of the overall variance in IL−6 values. In particular, more severe somatic symptoms of depression and higher degree of stroke severity predicted higher IL−6 values in patients with acute ischemic stroke.
| Variable | Step 1a | Step 2a |
|---|---|---|
| National Institutes of Health Stroke Scale | 0.602 (26.178) | 0.501 (20.802) |
| Ham-D Somatic | 0.456 (11.842) | 0.378 (11.842) |
| Ham-D Psychological | 0.266 (3.418) | –0.030 (0.041) |
| Barthel Index | 0.012 (0.007) | 0.226 (2.371) |
| Rey’s Word Recognition, correct recognitions | –0.220 (2.293) | –0.167 (1.266) |
| Stroop Word–Color Test; interference time, seconds | 0.201 (1.901) | 0.151 (1.027) |
| r2 | 0.363 | 0.495 |
| p | <0.0001 | <0.0001 |
Discussion
In the present study, we investigated the relationship between IL−6 serum levels and psychiatric, neurological, cognitive, and functional features during the acute phase of ischemic stroke.
We found that IL−6 levels were increased in patients with depressive disorders, particularly in those with MDD. Furthermore, IL−6 levels were associated with severity of the apathetic−amotivational and somatic symptoms of depression and the neurological symptoms of stroke. Noteworthy, unlike other studies focused on IL−6, in our sample we included only patients without history of possible confounders; thus it is unlikely that our results were affected by different external factors able to promote interleukin production independently of stroke and stroke-related phenomenology.
Despite the fact that pathophysiological consequences of acute ischemic stroke are still not very well understood, several findings suggest that neuroinflammatory mechanisms play a crucial role in ischemic injury, and the imbalance between pro-inflammatory and anti-inflammatory agents can result in worsened neurological outcomes43,44 and cognitive and neuropsychiatric disorders.2,9,24,26,27 Among pro-inflammatory agents, IL−6 has been proposed as an important mediator of stroke-induced immunological/inflammatory reaction. Indeed, elevated IL−6 values after cerebral ischemia seem to be associated with higher stroke severity and worse stroke outcomes.8,10,44
The relationship between IL−6 and stroke severity also appeared in our data. In particular, our study showed that higher IL−6 values are independently associated with higher degree of clinical severity during the acute phase of ischemic stroke. These findings are consistent with other studies showing that plasma and cerebrospinal cytokine fluid levels are related to clinical worsening of patients with acute ischemic stroke.43 For instance, in the study of Basic Kes and colleagues, an association between inflammatory parameters (IL−6, IL−10, and TNF-α), greater neurological deficit (evaluated by the NIHSS), and greater degree of patient disability (assessed with BI and modified Rankin Scale) was found in stroke patients at admission to the hospital compared with controls.10 Also, Yan and colleagues, in a study focused on the levels of cytokines up to 3 weeks after stroke, found a relationship between IL−6 levels and stroke severity, especially up to 1 week after the ischemic event.28 Worthy of note is that whereas most of these studies exploring IL−6 production after ischemic stroke did not exclude potential confounders that often appear after stroke and are associated with both higher levels of inflammatory markers and poor stroke outcomes, independently of other factors,8,44 we carefully selected our sample of patients in order to avoid all potential factors that affect inflammatory processes. Thus, we can assume that, in our work, the pro-inflammatory cytokine IL−6 is strongly associated with the ischemic brain damage occurring in the acute phase of stroke.
In several studies, changes in cytokine levels have been hypothesized to be associated with the etiology of post-stroke depression (PSD). Our research group suggested that pro-inflammatory cytokines may play a central role as mediators of mood and mood-related disorders, specifically in patients with stroke.24 We based our hypothesis on the evidence that pro-inflammatory cytokines and ischemic stroke were strongly related and interleukins were correlated with the existence of PSD. Most recently, Yang et al. found that, on Day 1 after stroke, patients diagnosed with PSD have increased serum levels of IL−6 and TNF-α.29 Finally, Su et al., evaluating cytokine changes over 1 year in patients with ischemic stroke, found a significant increase of cytokines in patients with PSD, confirming the hypothesis that PSD is associated with increased inflammation and that immune dysregulation leading to changes in cytokine levels may be involved in the etiology of PSD.27 Unfortunately, these previous studies do not clarify the relationship between the complex clinical picture of stroke phenomenology and inflammatory markers such us IL−6. In our study, we clearly identified that, in acute stroke patients, IL−6 production is most closely related to specific symptoms of depression and specific neurological symptoms only, and this is the real innovation of the present findings.
In the literature, only one study described an association between high IL−6 levels and somatic symptoms of depression. In this study, Stewart et al. investigated the association between depressive symptoms and inflammatory markers in 263 healthy adults for up to 6 years; the authors found that the somatic/vegetative symptoms of depression, assessed in the Beck Depression Inventory−II, were predictors of IL−6 changes over 6 years.45 Thus, to our knowledge, we are the first to describe the relationship between IL−6 and somatic symptoms of depression in the acute phase of stroke. Interestingly, IL−6 was higher in patients with loss of interest, loss of weight or appetite, and insomnia. Although loss of interest (the nuclear symptoms of the apathetic−amotivation syndrome) is one of the two core symptoms of depression along with sad mood, and its association with IL−6 peripheral levels can be part of the strong relationship between inflammation and PSD, loss of weight or appetite and insomnia are somatic symptoms of depression that deserve further explanation. Although IL−6 is mainly identified as an immune-modulatory cytokine with important pro-inflammatory effects,46 several studies in animal models have found that IL−6 also plays an important role in metabolic pathways. Indeed, mice treated with peripheral IL−6 show loss of weight, increased energy expenditure, and reduced food intake.47 Moreover, in humans, IL−6 was demonstrated to be associated with weight loss and cachexia in patients with cancer.48 In sleep disorders, the relationship between IL−6 and sleep is documented both in people with sleep disturbances and in healthy subjects.49 In particular, people with obstructive sleep apnea who experience fatigue, excessive daytime sleepiness, and narcolepsy have higher levels of IL−6 than normal subjects.50 Similarly, in healthy people, morning IL−6 levels are positively related to fragmented sleep and wakefulness and inversely correlated with deep sleep and sleep efficiency.51 Some authors have postulated that IL−6, by stimulating the activity of the hypothalamic-pituitary-adrenal axis, could be involved in the onset of depressive symptoms, insomnia, and alteration of sleep architecture in aging people.50 Thus, considering the central role played by IL−6 in the regulation of appetite, energy expenditure, body composition, and quality of sleep, we can assume that the overproduction of IL−6 during the early phase of cerebral ischemia might lead to the development of these important somatic symptoms, such as loss of appetite and sleep disturbances, which affect the quality of life of patients and their outcomes after stroke.
The present study has some limitations: 1) the sample of stroke patients is not very large; indeed, 48 patients with acute ischemic stroke were recruited and assessed. However, this is also a strength of the study, because we aimed to recruit patients without any possible confounding factors that might influence the inflammatory process, therefore including a very homogeneous sample of stroke patients; 2) the assessment of all patients might have been influenced by the environment or external stimuli because, during the hospitalization, the patients were treated in a stroke unit that cares for patients during the acute phase. In reality, this problem is common for all patients assessed during the very acute phase of the illness, thereby reflecting the real clinical world of this issue; 3) for the mood-disorder diagnosis, we could not respect the DSM-IV−TR clinical criterion based on duration of symptoms (i.e., at least 14 days) because the mean hospitalization duration in our stroke unit is 6 days. Thus, we opted to reduce the symptom-duration criterion of minor and major depression diagnosis from 2 weeks to 6 days; 4) the blood sample was performed only one time, at 72 hours after the onset of ischemic stroke, and we may have missed possible fluctuations in IL−6 levels and their relationships with the severity of the clinical phenomenology of stroke. Several studies in the literature, however, described IL−6 peak levels at 3 days after the acute event;32–34 5) finally, during the acute phase, both the extent of brain damage52 and the social and psychological stressors associated with stroke53 may be causal factors for PSD, and the weight of these two individual factors should be best clarified in future studies. Furthermore, we did not investigate the relationships among IL−6 secretion, somatic symptoms of depression, and stroke severity during the subacute and chronic stroke phases, when the inflammatory process may be less evident, and residual depressive symptoms may be less dependent on cytokine release.
The strengths of our study include a thorough evaluation of the complex picture of depressive disorders and symptoms and other neuropsychiatric and cognitive features by means of specific and validated scales and diagnostic criteria. Furthermore, in our sample we recruited only first-ever acute ischemic stroke patients without significant clinical history of medical conditions able to influence IL−6 production or promote inflammatory processes.
Conclusions
In conclusion, our results highlight the evidence of a possible biological network underlying the relationship between inflammatory parameters, in particular IL−6, stroke severity, and PSD in the acute phase of ischemic stroke. We suggest that IL−6, at least in the acute phase of stroke, could play an important role in the onset of apathetic−amotivational and somatic symptoms of depression, such as loss of appetite and sleep disorders, resulting in higher disability and poorer outcome of stroke patients.
Further studies are needed to confirm our findings and to explore the role of cytokines during the entire longitudinal phase of acute stroke, in order to develop adequate strategies aimed at improving clinical outcome and quality of life of patients with acute ischemic stroke.
1 : National Institute of Neurological Disorders and Stroke–Canadian Stroke Network vascular cognitive impairment harmonization standards. Stroke 2006; 37:2220–2241Crossref, Medline, Google Scholar
2 : Predictors of cognitive level and depression severity are different in patients with left and right hemispheric stroke within the first year of illness. J Neurol 2002; 249:1541–1551Crossref, Medline, Google Scholar
3 : Management of depression after stroke: a systematic review of pharmacological therapies. Stroke 2005a; 36:1098–1103Crossref, Medline, Google Scholar
4 : Citalopram reduced depressive symptoms in coronary artery disease with depression. ACP J Club 2007; 146:68Medline, Google Scholar
5 : Poststroke depression: prevalence, diagnosis, treatment, and disease progression. Biol Psychiatry 2003; 54:376–387Crossref, Medline, Google Scholar
6 : Alexithymic features in stroke: effects of laterality and gender. Psychosom Med 2001; 63:944–950Crossref, Medline, Google Scholar
7 : Levels of anti-inflammatory cytokines and neurological worsening in acute ischemic stroke. Stroke 2003; 34:671–675Crossref, Medline, Google Scholar
8 : Inflammatory markers and poor outcome after stroke: a prospective cohort study and systematic review of interleukin–6. PLoS Med 2009; 6:e1000145Crossref, Medline, Google Scholar
9 : Poststroke depression: a review. Can J Psychiatry 2010; 55:341–349Crossref, Medline, Google Scholar
10 : Pro-inflammatory and anti-inflammatory cytokines in acute ischemic stroke and their relation to early neurological deficit and stroke outcome. Clin Biochem 2008; 41:1330–1334Crossref, Medline, Google Scholar
11 : Clinical outcome following acute ischaemic stroke relates to both activation and autoregulatory inhibition of cytokine production. BMC Neurol 2007; 7:5Crossref, Medline, Google Scholar
12 : The relationship between inflammatory markers and poststroke cognitive impairment. J Geriatr Psychiatry Neurol 2010; 23:199–205Crossref, Medline, Google Scholar
13 : Disease outcome, alexithymia, and depression are differently associated with serum IL-18 levels in acute stroke. Curr Neurovasc Res 2009; 6:163–170Crossref, Medline, Google Scholar
14 : A meta-analysis of cytokines in major depression. Biol Psychiatry 2010; 67:446–457Crossref, Medline, Google Scholar
15 : A meta-analysis of differences in IL–6 and IL–10 between people with and without depression: exploring the causes of heterogeneity. Brain Behav Immun 2012; 26:1180–1188Crossref, Medline, Google Scholar
16 : Affective symptoms are associated with markers of inflammation and immune activation in bipolar disorders but not in schizophrenia. J Psychiatr Res 2011; 45:1608–1616Crossref, Medline, Google Scholar
17 : A neuroimmunological perspective on anxiety disorders. Hum Psychopharmacol 2012; 27:6–14Crossref, Medline, Google Scholar
18 : Associations of depression with C-reactive protein, IL–1, and IL–6: a meta-analysis. Psychosom Med 2009; 71:171–186Crossref, Medline, Google Scholar
19 : Inflammatory mechanisms in major depressive disorder. Curr Opin Psychiatry 2011; 24:519–525Crossref, Medline, Google Scholar
20 : Cytokines and major depression. Prog Neuropsychopharmacol Biol Psychiatry 2005; 29:201–217Crossref, Medline, Google Scholar
21 : Polymorphisms in inflammation-related genes are associated with susceptibility to major depression and antidepressant response. Mol Psychiatry 2008; 13:800–812Crossref, Medline, Google Scholar
22 : Plasma cytokine profiles in depressed patients who fail to respond to selective serotonin reuptake inhibitor therapy. J Psychiatr Res 2007; 41:326–331Crossref, Medline, Google Scholar
23 : Low serum IL–10 concentrations and loss of regulatory association between IL–6 and IL–10 in adults with major depression. J Psychiatr Res 2009; 43:962–969Crossref, Medline, Google Scholar
24 : The etiology of poststroke depression: a review of the literature and a new hypothesis involving inflammatory cytokines. Mol Psychiatry 2006; 11:984–991Crossref, Medline, Google Scholar
25 : Role of inflammatory markers in brain ischemia. Curr Opin Neurol 2008; 21:353–357Crossref, Medline, Google Scholar
26 : Interleukin–6 as an early predictor for one-year survival following an ischaemic stroke/transient ischaemic attack. Int J Stroke 2010; 5:16–20Crossref, Medline, Google Scholar
27 : Cytokine changes in the pathophysiology of poststroke depression. Gen Hosp Psychiatry 2012; 34:35–39Crossref, Medline, Google Scholar
28 : Prolonged elevation of cytokine levels after human acute ischaemic stroke with evidence of individual variability. J Neuroimmunol 2012; 246:78–84Crossref, Medline, Google Scholar
29 : The serum interleukin–18 is a potential marker for development of post-stroke depression. Neurol Res 2010; 32:340–346Crossref, Medline, Google Scholar
30 : The Stroke Data Bank: design, methods, and baseline characteristics. Stroke 1988; 19:547–554Crossref, Medline, Google Scholar
31 : “Mini-Mental State:” a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975; 12:189–198Crossref, Medline, Google Scholar
32 : Inflammatory cytokines in experimental and human stroke. J Cereb Blood Flow Metab 2012; 32:1677–1698Google Scholar
33 : Temporal profile of serum anti-inflammatory and pro-inflammatory interleukins in acute ischemic stroke patients. Neurol Sci 2001; 22:289–296Google Scholar
34 : IL–6: an early marker for outcome in acute ischemic stroke. Acta Neurol Scand 2005; 111:360–365Crossref, Medline, Google Scholar
35 : Structured Clinical Interview for DSM-IV–TR Axis I Disorders, Research Version, Patient Edition. (SCID–I/P). New York, New York State Psychiatric Institute, 2002Google Scholar
36
37 : A rating scale for depression. J Neurol Neurosurg Psychiatry 1960; 23:56–62Crossref, Medline, Google Scholar
38 : The clinical distinction between anxiety and depression. Br J Clin Pharmacol 1983; 15(Suppl 2):165S–169SCrossref, Medline, Google Scholar
39 ;
40 : Studies of interferences in serial verbal reactions. J Exp Psychol 1935; 18:643–662Crossref, Google Scholar
41 : Functional Evaluation: The Barthel Index. MD State Med J 1965; 14:61–65Medline, Google Scholar
42 ;
43 : Inflammatory cytokines in acute ischemic stroke. Curr Pharm Des 2008; 14:3574–3589Crossref, Medline, Google Scholar
44 : The use of blood biomarkers to predict poor outcome after acute transient ischemic attack or ischemic stroke. Stroke 2012; 43:86–91Crossref, Medline, Google Scholar
45 : A prospective evaluation of the directionality of the depression-inflammation relationship. Brain Behav Immun 2009; 23:936–944Crossref, Medline, Google Scholar
46 : IL–6 and IL–10 anti-inflammatory activity links exercise to hypothalamic insulin and leptin sensitivity through IKKbeta and ER stress inhibition. PLoS Biol 2010; 8Crossref, Medline, Google Scholar
47 : Interleukin–6-deficient mice develop mature-onset obesity. Nat Med 2002; 8:75–79Crossref, Medline, Google Scholar
48 : Pathophysiological role of hormones and cytokines in cancer cachexia. J Korean Med Sci 2012; 27:128–134Crossref, Medline, Google Scholar
49 : Cytokine-induced depression during IFN-alpha treatment: the role of IL–6 and sleep quality. Brain Behav Immun 2009; 23:1109–1116Crossref, Medline, Google Scholar
50 : Cytokines and pathological sleep. Sleep Med 2008; 9:603–614Crossref, Medline, Google Scholar
51 : The association between interleukin–6, sleep, and demographic characteristics. Brain Behav Immun 2005; 19:165–172Crossref, Medline, Google Scholar
52 : Plasmatic level of neuroinflammatory markers predict the extent of diffusion-weighted image lesions in hyperacute stroke. J Cereb Blood Flow Metab 2003; 23:1403–1407Crossref, Medline, Google Scholar
53 : Etiological mechanisms of post-stroke depression: a review. Neurol Res 2009; 31:904–909Crossref, Medline, Google Scholar



