Somatosensory Investigation of Patients With Orofacial Pain Compared With Controls
Abstract
The objective of this study was to investigate the sensorial characteristics of orofacial pain in patients compared with control subjects. A total of 336 subjects (282 patients and 54 control subjects) were evaluated to identify their thermal (cold and warm), tactile, and pain thresholds. Numbness was reported by 61.7% of the patients (p<0.001). Patients with trigeminal postherpetic neuralgia and burning mouth syndrome showed loss of thermal perception; patients with postherpetic neuralgia, burning mouth syndrome, and posttraumatic painful neuropathy had a decrease in tactile perception compared with the control subjects (p<0.001). In conclusion, other sensorial modalities besides pain are affected by neuropathic orofacial pain; these findings can help in the understanding of the pathophysiological mechanisms in orofacial pain.
The motor findings of several conditions have been extensively studied during the last century. However, it was only in the last few decades that clinical methodologies with precise equipment were developed and used in neurological and nonneurological diseases with the aim of clarification of pathophysiological mechanisms underlying these processes.1–4 Pain is one somatosensory modality, and thus sensory findings are important to be elucidated, and among orofacial conditions, special modalities such as gustation and olfaction have been considered.5–14
Among orofacial syndromes, there is evidence of sensory impairment in patients with idiopathic trigeminal neuralgia,15,16 burning mouth syndrome,17 trigeminal postherpetic neuralgia,18 atypical odontalgia,19 atypical facial pain,20 persistent idiopathic facial pain,21 and temporomandibular disorders22,23 and patients that underwent surgical oral procedures (third molar extractions, orthognatic surgeries),24,25 but these findings are not clearly understood. Thus, the objective of this study was to investigate the sensorial characteristics of orofacial pain in patients compared with control subjects.
Methods
Subjects
We enrolled 282 consecutive patients with orofacial pain who had been diagnosed at the Orofacial Pain Clinic of the Hospital das Clinicas of the School of Medicine, University of Sao Paulo. The patients had been recently diagnosed by the clinic. They were referred to the clinic between March 2003 and March 2009. They were 176 (62.4%) women, and the mean age was 58.20±13.56 years old (mean ± standard deviation). In the control group, we included 54 healthy subjects who had no orofacial pain complaints. There were 30 (55.6%) women, and their mean age was 45.67±15.08 years old (mean ± standard deviation).
Inclusion criterion for the study group was orofacial pain complaint for more than 6 months. The orofacial pain was diagnosed according to the criteria of the International Association for the Study of Pain.26 Eight patients had fibromyalgia associated with their facial pain, and they had been diagnosed according to the criteria of the American College of Rheumatology.27 The healthy subjects of the control group did not had any orofacial or generalized pain complaints before or at the time of the evaluation.
Exclusion criteria for both groups were systemic medical conditions (except fibromyalgia syndrome and diabetes mellitus in the study group) (e.g., other chronic pain syndromes and neurological or other rheumatological conditions). All patients and control subjects were informed about the purposes of the study, and all signed informed consent. The protocol was approved by the local Ethics Committee. Demographic data were compared using Pearson’s chi-square test (SPSS 17.0; SPSS, Chicago). There were no differences in ages or sex between the groups (p>0.50).
The patients that had burning mouth syndrome, posttraumatic neuropathy, cervicogenic headache, temporomandibular disorders, and persistent idiopathic facial pain did not take any medication within the last 6 months before the evaluation. Sporadic use of anti-inflammatory drugs and analgesics was reported by all patients (auto-medication), and the most commonly reported were dipyrone and acetaminophen. The patients with diabetic neuropathy were only taking hypoglycemics (metiformin and glibenclamide), the patients with idiopathic trigeminal neuralgia were taking carbamazepine (400−1200 mg daily), and the patients with postherpetic neuralgia and fibromyalgia were taking amitryptiline (25−150 mg daily). They had been taking the medications for at least 6 months prior to the study with no variation in doses. At the time of the study, none of the control subjects were taking analgesic medications; this is also true for the 6 months before the evaluation.28
Subjective Sensory Investigation
All subjects were interviewed about their perception of sensorial abnormalities, such as numbness and dysesthesia. The frequency of sensorial abnormalities (absent, eventual, frequent, or constant) and the intensity of the sensation (0–10 by the visual analog scale) were evaluated.
Quantitative Sensory Testing
All subjects underwent a standardized protocol of superficial facial perception, which was applied to distinct areas of the face (bilateral trigeminal branches)14,15: 1) thermal perception—cold and warm perception (electrical device designed at the Functional Neurosurgery Division, Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo); 2) mechanical/tactile perception—microfilaments of von Frey; and 3) pain perception—superficial algometry (Micromar, Diadema, São Paulo, Brazil).
Each thermal and mechanical stimulus was applied three times, and the threshold was identified when the subject recognized at least two of the three applications; if not, the next stimulus in a crescent order (starting with the smaller concentration) would be applied to avoid a tolerance effect. The algometry was performed with a superficial device and a disposable needle of 0.7×15 mm.
The somatosensory testing was performed at the three trigeminal branches (ophthalmic: front −2 cm above the pupil; maxillary: cheek −1 cm lateral to the nose wing; mandibular: chin skin −1 cm below the lip angle).15,18 The evaluation was performed in a silent room with acoustic protection and the doors closed. The subjects were sitting with their heads resting on a flat surface in the position with the Frankfurt line parallel to the floor. Only the patient and the researcher were in the room. All patients were evaluated by the same researcher. The patients and control subjects received the same instructions after they were positioned, which were to keep the eyes closed during the exam and that stimulus would be applied at their face and mouth and they should identify and report if they felt it and what they felt (by saying “yes” or “no” and “which was the stimulus”). Only the researcher knew the order of the stimuli. Finally, all findings were tabled and statistically analyzed.
| 1. | Thermal perception. Thermal (cold and warm) tests were performed with the equipment Thermosensi II (Functional Neurosurgery Division, Psychiatry Institute, Hospital das Clinicas, Medical School of the University of São Paulo, Sao Paulo, Brazil). The baseline temperature was 32°C, and the contact area of the thermode was 10×10 mm. The stimuli were applied with an amplitude of 0–45°C; the equipment has a temperature change speed of 1°C/sec. | ||||
| 2. | Mechanical tactile detection. Tactile tests were performed with standardized von Frey filaments with rounded tips of 0.5 mm of diameter (IITC, Woodland Hills, Calif.) (unit: g/mm2). | ||||
| 3. | Pain detection (superficial algometry). Pain thresholds were evaluated at the skin surface with needles of 30×0.7 mm perpendicularly applied with a mechanical algometer (Micromar, Diadema, Brazil). (unit: g/mm2). | ||||
Data Analysis
The mean figures, standard deviations, and frequencies were computed to summarize the distribution of numbers for each variable. After the initial descriptive evaluation, variables were tested in relation to the normal distribution by the Kolmogorov–Smirnoff test and Q-Q plots, and the variables that had normal distribution were analyzed by one-way analysis of variance and Tukey’s post hoc test. Nonparametric tests included Pearson’s chi-square and Fisher’s exact tests. The Z test was used to identify significant categories after the chi-square test. The level of significance was 5%.
Results
The distribution of patients according to the diagnoses is shown in Table 1. The distribution of age, sex, laterality, and branch affected according to the diagnostic is shown in Table 2. In this sample, there were 174 (61.7%) patients and 5 (9.3%) control subjects with numbness complaints (p<0.001). The frequency distribution was 128 (38.1%) constant, 29 (8.6%) frequent, and 22 (6.5%) eventual. The mean numbness intensity according to the visual analog scale was 6.14±2.42, and the mean dysesthesia intensity was 6.81±2.50. The distribution of numbness and dysesthesia according to the diagnosis is shown in Table 3.
| Diagnoses and laterality | N (%) |
|---|---|
| Diagnoses | |
| Idiopathic trigeminal neuralgia | 162 (57.4) |
| Persistent idiopathic facial pain | 30 (10.6) |
| Trigeminal diabetic neuropathy | 30 (10.6) |
| Trigeminal posttraumatic neuropathy | 19 (6.7) |
| Trigeminal postherpetic neuralgia | 18 (6.4) |
| Burning mouth syndrome | 11 (3.9) |
| Temporomandibular disorders | 5 (1.8) |
| Fibromyalgia | 5 (1.8) |
| Cervicogenic headache | 2 (0.7) |
| Laterality | |
| Right | 139 (49.3) |
| Left | 89 (31.6) |
| Bilateral | 54 (19.1) |
TABLE 1. Orofacial Pain Diagnoses and Laterality in the Study Group (N=282)
| Age (years) | Sex (%) | Side affected (%) | Branch affected (%) | |
|---|---|---|---|---|
| ITN (N=162) | 60.51±11.91 (34–85) | 88 (54.3) women | 101 (62.3) right | 6 (3.7) V1 |
| 60 (37) left | 50 (30.9) V2 | |||
| 01 (0.6) bilateral | 44 (27.2) V3 | |||
| 14 (8.6) V1–2 | ||||
| 40 (24.7) V2–3 | ||||
| 7 (4.3) V1–2–3 | ||||
| 1 (0.6) V1–3 | ||||
| TMD (N=5) | 47.50±6.84 (41–56) | 5 (100) women | 3 (60) right | — |
| 2 (40) left | ||||
| BMS (N=11) | 54.25±8.63 (35–61) | 11 (100) women | 11 (100) bilateral | — |
| PIFP (N=30) | 43.80±17.46 (23–76) | 29 (96.7) women | 14 (46.7) right | — |
| 10 (33.3) left | ||||
| 6 (20) bilateral | ||||
| Fibromyalgia (N=5) | 43.50±12.02 (35–52) | 4 (80) women | 5 (100) bilateral | — |
| PHN (N=18) | 71.33±8.16 (55–82) | 12 (66.7) women | 14 (77.8) right | 12 (66.7) V1 |
| 4 (22.2) left | 2 (11.1) V2 | |||
| 1 (5.6) V1–2 | ||||
| 2 (11.1) V2–3 | ||||
| 1 (5.6) V1–2–3 | ||||
| PTN (N=19) | 47.94±14.74 (32–83) | 14 (73.7) women | 5 (26.3) right | — |
| 13 (68.4) left | ||||
| 1 (5.3) bilateral | ||||
| CH (N=2) | 40.00±0.00 (40) | 1 (50) woman | 2 (100) right | — |
| DN (N=30) | 57.67±9.26 (37–77) | 12 (40) women | 30 (100) bilateral | — |
| p valuea | 0.020 | <0.001 | <0.001 | <0.001 |
TABLE 2. Distribution of Age, Sex, Side, and Branch Affected According to the Diagnosis (N=282)
| Numbness [N (%)] | Dysesthesia [N (%)] | |
|---|---|---|
| ITN (N=162) | 106 (65.4) | 29 (17.9) |
| TMD (N=5) | 3 (60) | 3 (60) |
| BMS (N=11) | 5 (45.5) | 5 (45.5) |
| PIFP (N=30) | 15 (50) | 12 (40) |
| Fibromyalgia (N=5) | 3 (60) | 3 (60) |
| PHN (N=18) | 16 (88.9) | 15 (83.3) |
| PTN (N=19) | 17 (89.5) | 16 (84.2) |
| CH (N=2) | 2 (100) | 0 (0) |
| DN (N=30) | 7 (23.3) | 4 (13.3) |
| Controls (N=54) | 5 (9.3) | 5 (9.3) |
| p valuea | <0.001 | <0.001 |
TABLE 3. Numbness and Dysesthesia According to the Diagnosis (N=336)
The evaluation of cold and warm detection showed impairment at the postherpetic neuralgia and burning mouth syndrome groups compared with the control subjects (Figure 1), and postherpetic neuralgia, burning mouth syndrome, and posttraumatic painful neuropathy showed decreased tactile sensorial perception (Figure 2). The mean superficial pain thresholds of patients with postherpetic neuralgia and idiopathic trigeminal neuralgia were higher than in the control subjects (Figure 3).
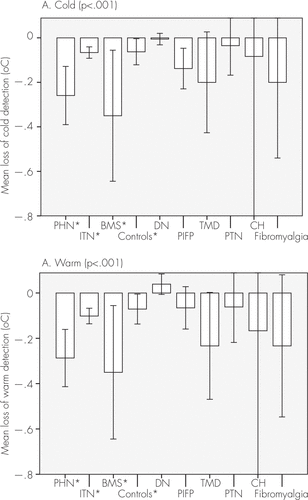
FIGURE 1. Impairment of Thermal Detections
[A]: Cold detection. [B]: Warm detection. In both evaluations, patients with PHN and BMS had sensory losses (N=336). [A]: Cold (p<0.001). [B]: Warm (p<0.001). Chi-square and Z tests: p<0.001. BMS: burning mouth syndrome; CH: cervicogenic headache; DN: diabetic neuropathy; ITN: idiopathic trigeminal neuralgia; PHN: trigeminal postherpetic neuralgia; PIFP: persistent idiopathic facial pain; PTN: posttraumatic neuralgia; TMD: temporomandibular disorder.
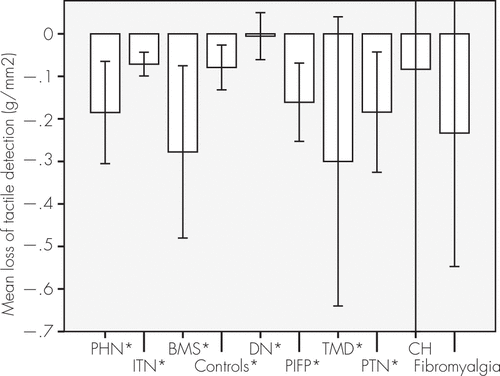
FIGURE 2. Impairment of Tactile Detection
Patients with PHN, BMS, and PTN had low tactile sensation (N=336). *Chi-square and Z tests: p<0.001. BMS: burning mouth syndrome; CH: cervicogenic headache; DN: diabetic neuropathy; ITN: idiopathic trigeminal neuralgia; PHN: trigeminal postherpetic neuralgia; PIFP: persistent idiopathic facial pain; PTN: posttraumatic neuralgia; TMD: temporomandibular disorder.
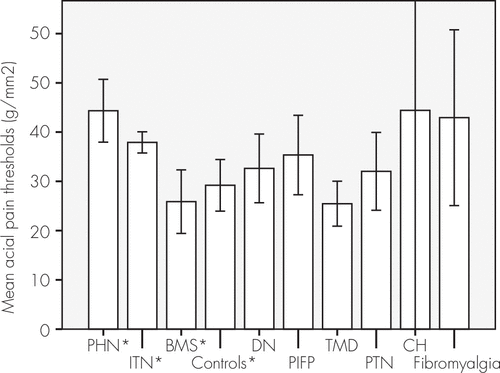
FIGURE 3. Mean Superficial Pain Thresholds
Patients with PHN and ITN had higher thresholds (N=336). *One-way analysis of variance, Tukey test: p=0.003. BMS: burning mouth syndrome; CH: cervicogenic headache; DN: diabetic neuropathy; ITN: idiopathic trigeminal neuralgia; PHN: trigeminal postherpetic neuralgia; PIFP: persistent idiopathic facial pain; PTN: posttraumatic neuralgia; TMD: temporomandibular disorder.
Discussion
Patients with neuropathic orofacial pain were more likely to have impairment of all sensorial modalities (thermal, tactile, and pain); however, patients with musculo-skeletal pain also presented abnormal tactile perception and the sensation of numbness (Table 3). Although sensory impairment is associated with neuropathies, pathophysiological mechanisms of pain chronification generate peripheral and central sensitization, hyperalgesia, hypoesthesia, and neuroplasticity.29 These are general described mechanisms, and in this clinical study, it was observed that the pain chronification may cause an abnormal sensation conducted by large fibers (tactile) that are not dependent on the etiology (neuropathic or not). However, small fiber compromise (pain and thermal) was more common in neuropathic conditions.30 It is possible that the conditions that had more thermal findings are also those more associated with peripheral deafferentation, for example, postherpetic neuralgia.14 The higher sensorial deficit in postherpetic neuralgia compared with others, conversely, possibly means a higher neural compromise.1,30 This has clinical relevance because discrete neurological abnormalities cannot be assessed at the clinic but only with quantitative sensory testing technologies. Conversely, they can be associated with neuropathic or nonneuropathic diagnoses according to the duration of pain and individual aspects that affect chronification besides the nature of etiology; thus, careful clinical examination and anamnesis should be complemented by, but not substituted with, neurological sensory evaluation, at the diagnosis of neuropathic pain in the orofacial region.
Some limitations of this study were the heterogeneity of the sample, resulting in small subgroups. It is possible that these small subgroups limited the power of the sample for statistical differences. However, these patients were not excluded because it was aimed to characterize the patients that are referred to this clinic during a determined period of time. Some patients were taking medications with an effect on the central nervous system, and this could affect the sensorial results. Therefore, this corresponded to a large sample of patients with orofacial pain, mostly with neuropathic pain, that were evaluated and compared according to their somatosensory characteristics.
In conclusion, other sensorial modalities besides pain are affected in neuropathic orofacial pain; these findings can help in the understanding of the pathophysiological mechanisms in orofacial pain. Thermal perception was more affected in patients with neuropathic orofacial conditions with an important peripheral component (burning mouth syndrome and postherpetic neuralgia), but tactile abnormalities were observed even in patients with musculo-skeletal pain, such as temporomandibular disorders. This suggests that central mechanisms of chronification may produce sensory impairment in neuropathic and non-neuropathic orofacial painful conditions, but sensorial abnormalities associated with small nerve fibers are more common in neuropathic facial pain.
1 : Quantitative sensory testing in trigeminal nerve damage assessment. J Orofac Pain 2004; 18:339–344Medline, Google Scholar
2 : Neurophysiologic and quantitative sensory testing in the diagnosis of trigeminal neuropathy and neuropathic pain. Pain 2005; 117:349–357Crossref, Medline, Google Scholar
3 : An up-to-date view on persistent idiopathic facial pain. Minerva Stomatol 2009; 58:289–299Medline, Google Scholar
4 : Reliability of intraoral quantitative sensory testing (QST). Pain 2010; 148:220–226Crossref, Medline, Google Scholar
5 : Interaction of chemosensory, visual, and statocyst pathways in Hermissenda crassicornis. J Gen Physiol 1978; 71:177–194Crossref, Medline, Google Scholar
6 : Thermal taste, PROP responsiveness, and perception of oral sensations. Physiol Behav 2008; 95:581–590Crossref, Medline, Google Scholar
7 : Tactile flow explains haptic counterparts of common visual illusions. Brain Res Bull 2008; 75:737–741Crossref, Medline, Google Scholar
8 : Reciprocal inhibitory visual-vestibular interaction. Visual motion stimulation deactivates the parieto-insular vestibular cortex. Brain 1998; 121:1749–1758Crossref, Medline, Google Scholar
9 : Relationship between dysgeusia and dysesthesia in stroke patients. Int J Neurosci 2008; 118:137–147Crossref, Medline, Google Scholar
10 : Burning mouth disorder (BMD) and taste: a hypothesis. Med Oral Patol Oral Cir Bucal 2008; 13:E470–E474Medline, Google Scholar
11 : Interactions between the chemical senses: trigeminal function in patients with olfactory loss. Int J Psychophysiol 2007; 65:177–181Crossref, Medline, Google Scholar
12 : Burning mouth syndrome and other oral sensory disorders: a unifying hypothesis. Pain Res Manag 2003; 8:133–135Crossref, Medline, Google Scholar
13 : Acoustic and neurophysiologic observations related to lingual nerve impairment. Int J Oral Maxillofac Surg 2009; 38:758–765Crossref, Medline, Google Scholar
14 : Somesthetic, gustatory, olfactory function and salivary flow in patients with trigeminal neuropathic pain. Oral Dis 2010; 16:482–487Crossref, Medline, Google Scholar
15 : Sensitive evaluation of patients with idiopathic trigeminal neuralgia treated with functional neurosurgery. Pain Clin 2006; 18:87–92Crossref, Google Scholar
16 : Olfactory threshold increase in trigeminal neuralgia after balloon compression. Clin Neurol Neurosurg 2006; 108:721–725Crossref, Medline, Google Scholar
17 : Central mechanisms in burning mouth syndrome involving the olfactory nerve: a preliminary study. Clinics (Sao Paulo) 2011; 66:509–512Crossref, Medline, Google Scholar
18 : Evaluation of the sensation in patients with trigeminal post-herpetic neuralgia. J Oral Pathol Med 2007; 36:347–350Crossref, Medline, Google Scholar
19 : Somatosensory abnormalities in atypical odontalgia: A case-control study. Pain 2008; 139:333–341Crossref, Medline, Google Scholar
20 : Differences and similarities between atypical facial pain and trigeminal neuropathic pain. Neurology 2007; 69:1451–1459Crossref, Medline, Google Scholar
21 : Persistent idiopathic facial pain exists independent of somatosensory input from the painful region: findings from quantitative sensory functions and somatotopy of the primary somatosensory cortex. Pain 2005; 118:80–91Crossref, Medline, Google Scholar
22 : Hypersensitivity to mechanical and intra-articular electrical stimuli in persons with painful temporomandibular joints. J Dent Res 2007; 86:1187–1192Crossref, Medline, Google Scholar
23 : Somatosensory profiles in subgroups of patients with myogenic temporomandibular disorders and Fibromyalgia Syndrome. Pain 2009; 147:72–83Crossref, Medline, Google Scholar
24 : Sensory testing of inferior alveolar nerve injuries: a review of methods used in prospective studies. J Oral Maxillofac Surg 2009; 67:292–300Crossref, Medline, Google Scholar
25 : Verification of nerve integrity after surgical intervention using quantitative sensory testing. J Oral Maxillofac Surg 2012; 70:263–271Crossref, Medline, Google Scholar
26 : Classification of Chronic Pain. Seattle, WA, IASP Press, 1994Google Scholar
27 : The American College of Rheumatology 1990 criteria for the classification of fibromyalgia. Arthritis Rheum 1990; 33:160–172Crossref, Medline, Google Scholar
28 : Clinical study of patients with persistent orofacial pain. Arq Neuropsiquiatr 2004; 62:988–996Crossref, Medline, Google Scholar
29 : Acute and chronic craniofacial pain: brainstem mechanisms of nociceptive transmission and neuroplasticity, and their clinical correlates. Crit Rev Oral Biol Med 2000; 11:57–91Crossref, Medline, Google Scholar
30 : Natural history of sensory function after herpes zoster. Pain 2010; 150:83–92Crossref, Medline, Google Scholar



