Verbal Fluency as a Rapid Screening Test for Cognitive Impairment in Early Parkinson’s Disease
Abstract
The phonological verbal fluency test can act as a fast screening test to detect cognitive deficits in neurological conditions. In the present study, its utility in the detection of executive deficits in patients with early Parkinson’s disease is demonstrated.
Early detection and effective management of the nonmotor symptoms of Parkinson’s disease (PD) is integral to the quality of life of both patients and caregivers. Even though cognitive impairment in the early stages of the disease may be subtle, the presence of early cognitive deficits is now well established, with executive function deficits being the most frequently reported.1–7
Even if an extensive evaluation of executive functions is critical to obtain an accurate description of a patient’s neuropsychological profile, such evaluations are not always available to clinicians working with patients with PD. Thus, the existence of a brief screening tool, which is easy to administer and yet shows high sensitivity and specificity in the detection of executive deficits in patients with early PD would be of great value for clinicians working with patients with PD.
Faced with a verbal fluency test, subjects are asked to enumerate either words beginning with a given letter within a limited amount of time (phonological fluency) or words within a given category, such as animals (semantic fluency). These tasks are easy to administer, take no longer than 2 minutes and require no specific materials. The phonological version is thought to reflect more strongly on executive functioning, as it demands that participants list words using a rule that is not natural to the way words are organized in our brain.8 The phonological fluency test has been shown useful in the detection of cognitive deficits in pathologies with frontal involvement.9 Even if verbal fluency represents a well-known test of executive function with established age-based norms, its properties as screening tool in the detection of broader executive dysfunction in PD has not yet been established.
The objective of the present study was, therefore, to investigate the utility of the phonological verbal fluency test in the detection of executive deficits in patients with early PD.
Methods
Permission for the study was initially obtained from the local research ethics committee, and all participants signed an informed consent prior to inclusion.
Thirty-two patients who met the United Kingdom Parkinson’s Disease Society Brain Bank criteria, between Hoehn and Yahr stages I to II,10 were recruited for a broader ongoing study at the Institute of Cognitive Neurology (INECO). Twenty-two control subjects, matched by age, educational level, and premorbid intelligence were also included in the study. Control volunteers were obtained through word of mouth and were only included if they reported no history of neurological or psychiatric disorders. PD patients were grouped as either having (PDED N=15) on not having executive dysfunction (PDNED N=17) based on their performance in gold-standard executive tests (see below). It was considered that a patient had executive dysfunction if performance on at least one tests of the executive battery was more than 1.5 standard deviations under the mean of controls.
Neuropsychological Assessment
Verbal fluency task.
All subjects were assessed with the standard Argentinean phonemic version of the phonological verbal fluency test,11 asking subjects to generate words beginning with the letter P in a 1-minute block. Score was the total number of correct words generated.
Standard executive battery.
Subjects were also assessed with gold-standard executive tests usually used to assess executive functions in patients with PD. Such executive battery included the Trail Making Test Part B,12 Digits Backward Test,13 and the Wisconsin Card Sorting Test.14
Data analysis.
The demographic and neuropsychological data were compared between the groups using analysis of variance (ANOVA) and Tukey honestly significant difference post hoc tests (when appropriate). To control for the influence of age on executive functions tasks, we applied analysis of covariance (ANCOVA) tests adjusted for age. The α value for all statistical tests was set at 0.05.
To compare the usefulness of the IFS (INECO Frontal Screening) in detecting the executive dysfunction of patients with PD, we determined the sensitivity and specificity of this test to discriminate between 1) healthy controls and PD patients (PDED and PDNED), and 2) PDED and PDNED patients. This was done by means of a receiver operating characteristic curve, detecting the optimal cut-off scores. The area under the receiver operating characteristic curve was used as a measure of discriminatory accuracy
Results
Demographic profile and total scores on tests of general cognitive status are summarized in Table 1. No significant difference was found for age (t (52) = 0.98; p=0.33) years of formal education (t (29) =-0.52; p=0.57) nor premorbid IQ (t (52) = -1.66; p=0.10) between PD patients and controls. Significant differences were found for age between PDED patients and controls (F2, 51=5, 54; p=0.028) and between the former and PDNED patients (F2, 51=5, 54; p=0.007).
| Characteristic | PDED | PDNED | Controls | ||||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | p | |
| Age (years) | 68.27 | 7.19 | 56.94 | 9.66 | 59.27 | 11.98 | 0.00 |
| Years of formal education | 13.33 | 4.76 | 14.41 | 4.93 | 14.50 | 2.79 | 0.669 |
| WAT | 35.47 | 5.04 | 38.18 | 3.32 | 38.68 | 2.93 | 0.036 |
| ACE | 89.07 | 7.69 | 93.7 | 4.83 | 95.82 | 4.63 | 0.003 |
| Digits Forward | 5.87 | 0.91 | 6.53 | 0.87 | 7.32 | 1.12 | 0.00 |
| Digits Backward | 4.13 | 0.91 | 4.47 | 0.62 | 4.82 | 1.09 | 0.09 |
| Verbal Fluency | 11.60 | 3.96 | 16.71 | 4.19 | 18.09 | 5.07 | 0.00 |
| Trail Making (A) | 67.33 | 47.39 | 38.76 | 12.54 | 34.86 | 15.12 | 0.00 |
| Trail Making (B) | 183.27 | 122.36 | 77.00 | 20.49 | 79.86 | 40.44 | 0.00 |
| WCST | 3.14 | 1.35 | 5.82 | 0.39 | 5.55 | 0.80 | 0.00 |
TABLE 1. Neuropsychological Performance PDED, PDNED, and Control Groupsa
Significant differences between groups were found on the verbal fluency test (F (2, 50) = 6.87, p<0.01, η2 = 0.21). Although controls showed a mean of 18.09 (SD=5.07), PDNED presented a mean of 16.71 (SD=4.19) and PDED a mean of 11.60 (SD=3.96) words in this test. A post hoc analysis (Tukey honestly significant difference, MS=20.51, df=50) revealed that controls performed significantly better than both PDED (p<0.01) and PDNED (p<0.001). Moreover, both PD groups differed significantly between each other (p<0.01), with PDNED outperforming PDED. As expected, significant differences between groups were also found on most gold-standard executive tests. In this regard, significant differences were found in in the Trail Making Test Part B (F (2, 50) = 6.60; p<0.01, η2 = 0.20). A post hoc analysis (Tukey honestly significant difference, MS=4383.6, df=50) showed that both controls (p<0.01) and PDNED (p<0.01) performed better than PDED, even if a significant effect of age (p<0.01) was observed on Trail Making Test Part B performance. Significant differences between groups were also found on the WCST (Wisconsin Card Sorting Test) (F (2, 50) = 29.46, p<0.01, η2 = 0.50). A post hoc analysis (Tukey honestly significant difference, MS=0.76, df=50) revealed that PDNED (p<0.01) and controls (p<0.01) exhibited a higher performance than PDED. Unexpectedly, no significant differences were found between groups on the Digits Backward Test (F (2, 50) = 2.50, p=0.92, η2 = 0.09). This lack of significant differences can be related to the fact that Digit Backward Test relies on verbal instead of visual working memory, the latter of which is most commonly decreased in early PD patients.15
Significant correlations were found between verbal fluency score, and the total number of categories abstracted on the WCST (r=0.52; p<0.001), and time to complete the TMT-B (r=−0.82; p<0.001). No significant correlations were found between the verbal fluency score and the Digits Backward Test (r=0.23; p<0.01)
A receiver operating characteristic curve analysis on the verbal fluency total score comparing healthy controls and PD patients (PDED and PDNED) generated a cut-off score of 14 points with sensitivity of 73.3% and specificity of 82.1% (Figure 1A).
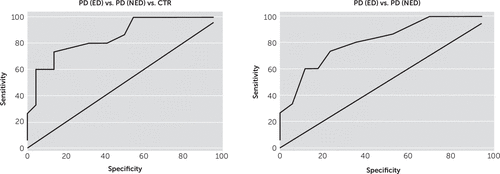
FIGURE 1. Verbal Fluency Receiver Operating Characteristic Curvea
a [A] Receiver operating characteristic curve analysis between PDED, PDNED,, and control groups. [B] Verbal fluency receiver operating characteristic curve analysis between PDED and PDNED.
With a verbal fluency score of 14, 73.3% of the PDED patients, 76.4% of the PDNED patients, and 86.3% of the controls were correctly classified. Area under the receiver operating characteristic curve was 0.85 [confidence interval (CI) 0.72–0.97; p<0.01]. Furthermore, when patients were divided into PDED and PDNED, a receiver operating characteristic curve analysis comparing both groups generated a cut-off score of 14 points, with a sensitivity of 73.3% and a specificity of 76.4% and an area under the curve of 0.81 (CI 0.66–0.96; p<0.01) (Figure 1B).
Discussion
In the present study, we showed that the phonological verbal fluency task may be a good screening test for the detection of executive deficits in PD. Our findings show adequate concurrent validity, as revealed by the significant correlations that the test showed with executive tasks, and adequate discriminant validity, as evidenced by the test’s capability to significantly differentiate PDED patients from healthy controls and from PDNED patients. All the between-group differences remained significant even after controlling for the effect of age save an effect of age observed on the Trail Making Test B performance. This is not unexpected, as several studies16–18 have shown that among adults processing speed is positively correlated with age.
Using a cut-off score of 14 points, the Verbal Fluency Task showed a sensitivity of 73.3% and a specificity of 76.4% in the detection of executive deficits in PD. These results suggest that this brief test is able to correctly classify the majority of PD patients with executive dysfunction and those without it.
Our results have important implications for clinicians working with patients with PD who cannot always access more extensive neuropsychological evaluations. The present investigation indicates that the phonological verbal fluency task can act as a sensitive and specific screening tool in the detection of executive deficits in this pathology. Even if an extensive evaluation of executive functions might be critical to obtain an accurate description of a patient’s neuropsychological profile, this 2-minute screening tool, combined with an adequate interview regarding the impact of cognitive dysfunction in patients’ lives, can be useful in the detection of patients who may benefit from more extensive cognitive evaluations and cognitively targeted interventions.
Although no single test can replace the value of a complete and focused history-taking or that of a comprehensive neuropsychological evaluation, the present study indicates that phonological verbal fluency can act as a reliable, brief, and easy-to administer tool for the screening of executive dysfunction in PD. In this regard, the phonological verbal fluency test could act as a simple tool to be used in everyday clinical practice to detect those patients with PD who will benefit from a more comprehensive neuropsychological battery, including a thorough cognitive history-taking and further test of executive and social functioning.
1 : The cognitive ability of an incident cohort of Parkinson’s patients in the UK. The CamPaIGN study. Brain 2004; 127:550–560Crossref, Medline, Google Scholar
2 : Cognitive profile of patients with newly diagnosed Parkinson disease. Neurology 2005; 65:1239–1245Crossref, Medline, Google Scholar
3 : Heterogeneity of Parkinson’s disease in the early clinical stages using a data driven approach. J Neurol Neurosurg Psychiatry 2005; 76:343–348Crossref, Medline, Google Scholar
4 : Evolution of cognitive dysfunction in an incident Parkinson’s disease cohort. Brain 2007; 130:1787–1798Crossref, Medline, Google Scholar
5 : Executive dysfunction in Parkinson’s disease: a review. J Neuropsychol 2013; 7:193–224Crossref, Medline, Google Scholar
6 : Throwing some light on executive function in Parkinson’s disease. Mov Disord 2013; 28:1052Crossref, Medline, Google Scholar
7 : Pattern of executive impairment in mild to moderate Parkinson’s disease. Dement Geriatr Cogn Disord 2013; 36:50–66Crossref, Medline, Google Scholar
8 : Neuropsychological Assessment. New York, Oxford University Press, 1995Google Scholar
9 : Verbal fluency as a rapid screening test for cognitive impairment in progressive multiple sclerosis. J Neurol Neurosurg Psychiatry 2012; 83:346–347Crossref, Medline, Google Scholar
10 . Parkinsonism: onset, progression, and mortality. Neurology 1967; 17:427–42Crossref, Medline, Google Scholar
11 : Fluencia verbal en español. Datos normativos en Argentina. Medicina (B Aires) 2000; 60:561–564Medline, Google Scholar
12 : Partington’s Pathway Test. Psychological Service Center Bulletin 1949; 1:9–20Google Scholar
13 : WAIS – III. Escala de inteligencia para adultos de Wechsler. Tercera edición. Buenos Aires, Paidós, 2002Google Scholar
14 : A modified card sorting test sensitive to frontal lobe defects. Cortex 1976; 12:313–324Crossref, Medline, Google Scholar
15 : Spatial and non-spatial working memory at different stages of Parkinson’s disease. Neuropsychologia 1997; 35:519–532Crossref, Medline, Google Scholar
16 : [Spanish normative studies in a young adult population (NEURONORMA young adults Project): norms for the verbal span, visuospatial span, Letter-Number Sequencing, Trail Making Test and Symbol Digit Modalities Test]. Neurologia 2012; 27:319–329Crossref, Medline, Google Scholar
17 : Two-factor structure of the Comprehensive Trail-Making Test in adults. Appl Neuropsychol Adult 2013; 20:155–158Crossref, Medline, Google Scholar
18 : Interaction of age, cognitive function, and gait performance in 50-80-year-olds. Age (Dordr) 2014; 36:9693Crossref, Medline, Google Scholar



