Vaping and the Brain: Effects of Electronic Cigarettes and E-Liquid Substances
E-cigarettes (ECs), also known as vape pens, e-cigars, vaping devices, e-hookahs, mods, vapes, tank systems, electronic nicotine delivery systems (ENDS), and puff bars (fourth-generation ECs), are battery-operated electronic devices used to inhale a heat-generated aerosol from an e-liquid source (2, 5, 11). E-liquids contain basic ingredients, including water, propylene glycol, glycerol, optional nicotine (available at various concentrations), and flavoring agents (e.g., chocolate, cotton candy, and green apple) (4). E-liquids in disposable puff bars contain over 120 different substances, including cytotoxic chemicals (2). Refill solutions, e-liquid cartridges, and emission vapors (i.e., aerosol mix of small particles in the air) contain many other substances and hazardous chemicals, including solvents, volatile organic compounds (e.g., benzene and formaldehyde), acrolein, acids, and synthetic coolants (2, 4, 8, 11–14). ECs can also be used to deliver marijuana. Using ECs is commonly known as “vaping” (15) (Figure 1).
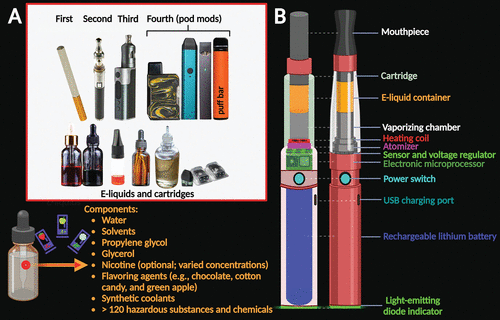
FIGURE 1. Generations of electronic cigarette (EC) designs, e-liquids, and components. A. First-generation disposable ECs were intended for a single use and were not rechargeable or refillable. Second-generation ECs included prefilled or refillable cartridges with rechargeable batteries. Third-generation ECs (referred to as “tanks” or “mods”) are adjustable devices, permitting users to modify the e-liquids (e.g., adding nicotine or marijuana), and are designed to create more abundant vapor emissions, thus delivering a higher dose of the substance. Fourth-generation ECs (commonly called “pod mods”) are prefilled or refillable (pods or cartridges), operate with low-power batteries, may exhibit the same customizable attributes of mods (1), and can deliver higher nicotine doses to users (2, 3). At present, puff bars are the most popular form of ECs (2). Many e-liquids contain the same basic ingredients (2, 4). B. Despite the rapid evolution of EC products, they typically contain the same basic components (second-generation EC pictured here): a cartridge with a reservoir designed for the e-liquid, a manual power switch to prepare and initiate atomization (vapor generation) of the e-liquid, a vaporizing chamber and heating coil to increase the temperature of the e-liquid, an atomizer unit to produce vapors from the heated e-liquid, a voltage regulator and microprocessor to regulate the electricity of the coil and light, a rechargeable lithium battery to power the device, and the light-emitting diode (LED) indicator (red, green, blue) to indicate when the user inhales through the mouthpiece or when the battery is running low. The LED visually emulates a conventional cigarette, and the vapor emissions seem to emulate the appearance of smoke (2, 5, 6).
ECs have several components, including a cartridge or “pod,” where the liquid solution containing nicotine, flavoring substances, and other chemicals is vaporized upon activation of an electronic heating element in the device that is triggered by inhalation (5). The heating element is powered by a rechargeable lithium battery (15). The consumer can choose the nicotine concentration of the e-liquid that is loaded into the EC cartridge (5) (Figure 1). The technology of pod mods allows manufacturers to combine acid mixtures and other dangerous chemicals with nicotine salts or crystals to elevate the nicotine concentrations of their products (2, 3). This design allows delivery of higher drug doses via relatively inconspicuous devices with reduced electrical power that closely resemble electronic USB drives (2, 3, 16, 17). Popular pods contain approximately 59.2–66.7 mg/mL of nicotine, which is comparable to one pack of 20 conventional cigarettes (3, 15, 18). Once the cartridge or pod is installed in the EC device, the user then inhales the vaporized products of the e-liquid into the lungs, and the residues are exhaled into the air (5, 15).
It has been suggested, incorrectly, that ECs are less harmful than conventional cigarettes (19). ECs are promoted as containing fewer chemicals, having lower levels of carcinogens, and producing fewer toxic agents than conventional cigarettes (19, 20). However, they are not emission-free devices, and their aerosol emissions are not harmless (5, 6, 11, 20). Bystanders to exhaled vaping fumes can be exposed to toxic aerosols and carcinogenic agents (e.g., acrolein, benzene, diacetyl, and formaldehyde) through second-hand or third-hand exposure (6, 11, 21). Elements from second-hand EC vapor increase particulate matter in the air, which can be harmful and lead to preclinical forms of cardiovascular, pulmonary, and other diseases (6, 7, 11, 22, 23). Third-hand exposure (through residual chemicals left on surfaces, skin, hair, and clothing following vaping) plays a role in the overall exposure of children to nicotine and a variety of toxicants (24, 25).
Vaping causes serious damage throughout the human body, including organs in the cardiovascular, central nervous, immunological, and respiratory systems. This damage is primarily due to the deleterious effects of toxic compounds that exacerbate oxidative stress responses and inflammatory reactions, causing endothelial dysfunction, cellular senescence, and subsequent disease sequelae (2, 6–10) (Figure 2). The nicotine and nonnicotinic agents in ECs, including hazardous neurotoxic elements (i.e., aluminum, arsenic, chromium, cadmium, copper, lead, manganese, nickel, tin, and zinc) and other by-products, pose serious threats to the brain (8, 13, 26, 27). To date, only a few studies have investigated the effects of these compounds on neurological function, mostly in animal models (8, 28).
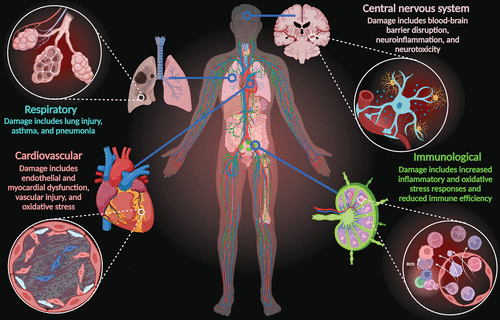
FIGURE 2. Vaping causes serious systemic disturbances throughout the body, including damage to organs of the cardiovascular, nervous, immunological, and respiratory systems. Damage is primarily due to the deleterious effects of toxic compounds contained in the e-liquid, which exacerbate oxidative stress responses and inflammatory reactions, resulting in endothelial dysfunction, cellular senescence, and subsequent disease sequelae (2, 6–10).
COVER. Artistic depiction of the effects of vaping electronic cigarettes in the brain. The gray and smoky outline of the brain represents widespread effects, and the colored arrows represent neural pathways responsible for the psychiatric, neurological, and cognitive effects of nicotine, the e-liquid substances, and their by-products.
All images were created with Canva and BioRender.com, for which the Mid-Atlantic (VISN 6) MIRECC holds renewable registrations.
According to the Centers for Disease Control and Prevention, ECs “have the potential to benefit adults who smoke and who are not pregnant if used as a complete substitute for regular cigarettes and other smoked tobacco products” (29). However, use of ECs may increase the risk of future cigarette smoking among adolescents and young adults (30). The use of any form of tobacco product (smoked, smokeless, or electronic) by youths is considered unsafe (15). The U.S. Surgeon General has concluded that exposure to nicotine during adolescence can cause addiction and impair the developing adolescent brain (11).
The use of ECs has skyrocketed among U.S. adolescents and young adults since 2014 (31, 32). In 2018, usage increased 77.8% over the previous year, the largest surge of youth tobacco consumption ever recorded by the Centers for Disease Control and Prevention (33). Most adolescents and young adult users do not know that ECs may contain higher concentrations of nicotine than conventional cigarettes (15, 34). Similar trends have been reported among youth in countries outside the United States (35–38). However, the United States dominates the global vaping market. By 2022, over 2.6 million young Americans reported current use of ECs (39). Global market research reports indicated that ECs generated $22.45 billion in U.S. dollars in 2022 and a compounded annual growth of 30.6% was projected from 2023 to 2030 (40).
The increased popularity and use of ECs seems to be linked (in part) to novelty and the alluring flavors of various e-liquid options (41). The flavoring chemicals and additives of ECs are largely unregulated (8, 14, 21). In 2020, the Food and Drug Administration enacted a ban on flavored EC pods in an effort to reduce accelerated use among young consumers (42). A technicality in the ban, which did not include “disposable products” (i.e., puffs ECs), has allowed young consumers and suppliers to keep these devices in high demand (2, 43, 44). At present, several thousand flavoring options and hundreds of brands are available (14, 26). However, the explosion in EC use also suggests that users may be experiencing dependency to both smoking and vaping (12, 45).
Studies in Animal Models and Humans
Subchronic exposure to e-liquid (without nicotine) hindered growth and development of nematodes, caused abnormal general neuromotor behavior, and impaired advanced learning and memory (46). A study in mice indicated that chronic exposure (2 months) to vaporized elements from ECs resulted in toxic metal aggregation in the central nervous system, potentially leading to endogenous dysregulation, including metal dyshomeostasis and neurotoxicity (47). Another study in mice compared the neurological effects of short-term exposure to EC vapor and smoke from conventional cigarettes over 14 days. The EC-vapor-exposed group showed neurotoxicity leading to neuroinflammatory responses (confirmed via tumor necrosis factor alpha expression in brain tissue), and comparable cognitive and memory impairments were observed in both groups. In addition, the EC group showed longer latencies to find food rewards, with a lack of improvement in spatial memory learning (48). Taken together, these results link EC-induced neuroinflammation to deleterious alterations in cognitive and memory function (46–48).
It is well established that nicotine exposure during adolescence alters the structure and function of the developing brain, impairing several cognitive processes and increasing rates of mood disorders and addiction (11). Deterioration in cognitive functions (e.g., attention, impulse control, memory, and reasoning) is linked to smoking conventional cigarettes during adolescence and early adulthood, increasing the risk of long-lasting mental health concerns (49, 50). A recent cross-sectional study reported that EC use was associated with subjective cognitive deficits in U.S. adults (51).
The toxicity of several commercially available EC refills across a range of concentrations (0.001%–1%) was analyzed with in vitro experiments using stem cells from mice and humans. Some samples had no cytotoxic effects, and others displayed high levels of cytotoxicity. Interestingly, cytotoxicity was not linked to nicotine concentrations but was linked to the amount and concentration of flavoring toxicants (52). Popular flavors, such as caramel and butterscotch, potentiated levels of cytotoxic effects; thus, commonly known flavoring chemicals should not be assumed to be harmless (52). Similar results have been reported in other studies (16, 53).
Flavoring Additives and Other Toxicants
Menthol was introduced to conventional cigarettes in the 1950s and is also widely found in ECs (8, 54). This popular flavoring agent increases nicotine bioavailability (54), enhances nicotine reward-related behavior, and potentiates nicotine actions in midbrain dopaminergic neurons (55). Menthol also promotes smoking initiation among youths (56). Conventional menthol cigarette smokers are twice as likely to become lifelong smokers compared with conventional nonmenthol cigarette smokers (57). The evidence suggests that menthol is a highly reinforcing sensory cue when paired with nicotine and promotes addiction.
Farnesol, a sesquiterpene alcohol found in many foods, such as peaches and vegetables, is commonly added to ECs as a flavor enhancer (e.g., green apple) (8, 58). This additive promotes reward-related behavior (via nicotinic acetylcholine receptors), amplifying the effects of nicotine on dopaminergic neurons in the ventral tegmental area and altering the function of dopamine and GABA neurons in the midbrain (58, 59). Many EC constituents and flavoring agents chemically decompose during vaporization, which transforms them into different toxicants, and variations in heating temperature contribute to this process. Effects of these substances are unknown (4, 8, 60).
A recent study investigated the long-term impact of maternal exposure to EC vapor on blood-brain barrier integrity and behavioral outcomes in postnatal mice. Vapor exposure led to cellular and molecular alterations, which are potentially associated with long-term neurotoxicity and cerebrovascular diseases (61). These changes resulted in postnatal blood-brain barrier disruptions and neurodevelopmental and behavioral impairments (61). The investigators concluded that maternal exposure to EC vapors was related to motor disabilities and learning and memory dysfunction among the offspring that persist into adolescence and adulthood (61). These findings are relevant because the use of ECs among pregnant women (both smokers and nonsmokers) has increased since the introduction of ECs to the U.S. market (62–64).
Another in vivo study in mice demonstrated that nonnicotinic toxicants (e.g., volatile organic compounds, acetaldehyde, acrolein, and formaldehyde) of EC emissions appeared to abruptly alter the biological and molecular integrity of the blood-brain barrier by reducing gene expression of occludin, a protein essential for stabilizing the blood-brain barrier. Changes were observed within tight junctions of blood vessels in the frontal cortex, and paracellular permeability was increased in thalamic nuclei (27). These alterations contributed to neurovascular dysfunction that promoted neuroinflammation, resulting in cognitive deficits (27, 60).
Endothelial cell-derived microvesicles are both biomarkers and mediators of vascular health and pathogenesis in humans (65). Activation of endothelial cell-derived microvesicles in EC users decreased activation of nitric oxide synthase in microvascular endothelial cells and production of nitric oxide in the brain (9). These changes caused further biomolecular and cellular alterations in the blood-brain barrier, resulting in vascular dysregulation in the brain and increased risk of cerebrovascular diseases (9, 66).
Conclusions
ECs do not seem to be a safer alternative to conventional cigarettes during smoking-cessation therapy. They appear to share similar chemical profiles (nicotine, nonnicotinic toxicants, and other emitted substances). Therefore, users may experience equal or possibly higher pathophysiological alterations, resulting in many health issues and higher risks for certain chronic diseases (e.g., asthma and cancer), as well as neurological conditions (e.g., addiction, stroke, cognitive dysfunction, and neurodevelopmental disorders). Bystanders exposed to second-hand or third-hand EC vapors also appear to be at higher risk for these diseases.
Furthermore, the nonnicotinic constituents in ECs potentiate epigenetic alterations, cellular dysfunction, neuroinflammation, oxidative stress, neurotransmitter dysregulation, and other abnormalities. ECs without nicotine also appear to have substantial detrimental effects on vital central nervous system regulatory components, such as the blood-brain barrier. The added flavorings, solvents, acids, synthetic coolants, volatile organic compounds, and other toxicants, including aerosolized by-products and metabolites of e-liquids, warrant attention, because flavorings seem to have increased the popularity of ECs among adolescents and young adults. Popularity of ECs has also increased among pregnant women, nonsmokers, and former conventional cigarette smokers.
Additional studies are needed to fully understand effects of the chemical constituents (especially nonnicotinic compounds) in e-liquids and their vaporized by-products, which are physiologically and pharmacokinetically uncharacterized. Flavoring substances are largely unregulated based on their clearance for ingestion. However, they are not intended for inhalation, and the exposure of respiratory and nervous tissue to these substances has not been properly studied. The popularity and increasing usage of e-liquids raise major concerns for public health.
1. E-Cigarette, or Vaping, Products Visual Dictionary. Atlanta, Centers for Disease Control and Prevention. https://www.cdc.gov/tobacco/basic_information/e-cigarettes/pdfs/ecigarette-or-vaping-products-visual-dictionary-508.pdf Google Scholar
2. : Disposable puff bar electronic cigarettes: chemical composition and toxicity of e-liquids and a synthetic coolant. Chem Res Toxicol 2022; 35:1344–1358Crossref, Medline, Google Scholar
3. : Toxicological assessment of electronic cigarette vaping: an emerging threat to force health, readiness and resilience in the US Army. Drug Chem Toxicol 2022; 45:2049–2085Crossref, Medline, Google Scholar
4. : Toxicology of e-cigarette constituents; in Public Health Consequences of E-Cigarettes. Washington, DC, National Academies Press, 2018. https://www.ncbi.nlm.nih.gov/books/NBK507184/ Google Scholar
5. : E-cigarettes: a scientific review. Circulation 2014; 129:1972–1986Crossref, Medline, Google Scholar
6. : Impact of electronic cigarettes on the cardiovascular system. J Am Heart Assoc 2017; 6:e006353Crossref, Medline, Google Scholar
7. : Update: interim guidance for health care providers evaluating and caring for patients with suspected e-cigarette, or vaping, product use associated lung injury: United States, October 2019. MMWR Morb Mortal Wkly Rep 2019; 68:919–927Crossref, Medline, Google Scholar
8. : Neurotoxicity of e-cigarettes. Food Chem Toxicol 2020; 138:111245Crossref, Medline, Google Scholar
9. : Endothelial-derived extracellular vesicles associated with electronic cigarette use impair cerebral microvascular cell function. J Appl Physiol 2023; 135:271–278Crossref, Medline, Google Scholar
10. : The impact of e-cigarette exposure on different organ systems: a review of recent evidence and future perspectives. J Hazard Mater 2023; 457:131828Crossref, Medline, Google Scholar
11. E-Cigarette Use Among Youth and Young Adults: A Report of the Surgeon General. Atlanta, Centers for Disease Control and Prevention. 2017. https://www.cdc.gov/tobacco/sgr/e-cigarettes/index.htm Google Scholar
12. : E-cigarette effects on vascular function in animals and humans. Pflugers Arch 2023; 475:783–796Crossref, Medline, Google Scholar
13. Know the Risks: E-Cigarettes and Young People. Washington, DC, Office of the US Surgeon General, US Department of Health and Human Services. https://e-cigarettes.surgeongeneral.gov/knowtherisks.html Google Scholar
14. : Use patterns, flavors, brands, and ingredients of nonnicotine e-cigarettes among adolescents, young adults, and adults in the United States. JAMA Netw Open 2022; 5:e2216194Crossref, Medline, Google Scholar
15. Quick Facts on the Risks of E-Cigarettes for Young People. Atlanta, Centers for Disease Control and Prevention. 2023. https://www.cdc.gov/tobacco/basic_information/e-cigarettes/Quick-Facts-on-the-Risks-of-E-cigarettes-for-Kids-Teens-and-Young-Adults.html Google Scholar
16. : High concentrations of flavor chemicals are present in electronic cigarette refill fluids. Sci Rep 2019; 9:2468Crossref, Medline, Google Scholar
17. : Adolescents’ use of “pod mod” e-cigarettes: urgent concerns. N Engl J Med 2018; 379:1099–1102Crossref, Medline, Google Scholar
18. : High-nicotine electronic cigarette products: toxicity of JUUL fluids and aerosols correlates strongly with nicotine and some flavor chemical concentrations. Chem Res Toxicol 2019; 32:1058–1069Crossref, Medline, Google Scholar
19. : Electronic cigarettes as a harm reduction strategy for tobacco control: a step forward or a repeat of past mistakes? J Public Health Policy 2011; 32:16–31Crossref, Medline, Google Scholar
20. : Generally recognized as safe: uncertainty surrounding e-cigarette flavoring safety. Int J Environ Res Public Health 2017; 14:1274Crossref, Medline, Google Scholar
21. : Emissions from electronic cigarettes: assessing vapers’ intake of toxic compounds, secondhand exposures, and the associated health impacts. Environ Sci Technol 2017; 51:9271–9279Crossref, Medline, Google Scholar
22. : Increased cardiac sympathetic activity and oxidative stress in habitual electronic cigarette users: implications for cardiovascular risk. JAMA Cardiol 2017; 2:278–284Crossref, Medline, Google Scholar
23. General Information About Secondhand Smoke. Atlanta, Centers for Disease Control and Prevention, 2022. https://www.cdc.gov/tobacco/secondhand-smoke/about.html Google Scholar
24. : Should we worry about children’s exposure to third-hand by-products generated from electronic nicotine delivery systems? ERJ Open Res 2020; 6:00194-02020Crossref, Medline, Google Scholar
25. : Preliminary evidence that high levels of nicotine on children’s hands may contribute to overall tobacco smoke exposure. Tob Control 2018; 27:217–219Crossref, Medline, Google Scholar
26. : Effects of electronic nicotine delivery systems and cigarettes on systemic circulation and blood-brain barrier: implications for cognitive decline. Am J Pathol 2021; 191:243–255Crossref, Medline, Google Scholar
27. : Electronic cigarette exposure disrupts blood-brain barrier integrity and promotes neuroinflammation. Brain Behav Immun 2020; 88:363–380Crossref, Medline, Google Scholar
28. : Impact of electronic cigarette vaping on cerebral ischemia: what we know so far. Transl Stroke Res 2022; 13:923–938Crossref, Medline, Google Scholar
29. About Electronic Cigarettes (E-Cigarettes). Atlanta, Centers for Disease Control and Prevention. 2023. https://www.cdc.gov/tobacco/basic_information/e-cigarettes/about-e-cigarettes.html#:∼:text=E%2Dcigarettes%20have%20the%20potential,not%20currently%20use%20tobacco%20products Google Scholar
30. : Association between initial use of e-cigarettes and subsequent cigarette smoking among adolescents and young adults: a systematic review and meta-analysis. JAMA Pediatr 2017; 171:788–797Crossref, Medline, Google Scholar
31. : Prevalence of e-cigarette use among adults in the United States, 2014-2018. JAMA 2019; 322:1824–1827Crossref, Medline, Google Scholar
32. : Tobacco product use among middle and high school students: United States, 2011–2017. MMWR Morb Mortal Wkly Rep 2018; 67:629–633Crossref, Medline, Google Scholar
33. : Vital signs: tobacco product use among middle and high school students: United States, 2011–2018. Morb Mortal Wkly Rep 2019; 68:157–164Crossref, Medline, Google Scholar
34. : Synthetic nicotine descriptors: awareness and impact on perceptions of e-cigarettes among US youth. Tob Control 2023:tc-2023-057928Crossref, Medline, Google Scholar
35. : Tripling use of electronic cigarettes among New Zealand adolescents between 2012 and 2014. J Adolesc Health 2015; 56:522–528Crossref, Medline, Google Scholar
36. : Electronic cigarettes in Canada: prevalence of use and perceptions among youth and young adults. Can J Public Health 2014; 105:e97–e102Crossref, Medline, Google Scholar
37. : Cross-sectional study examining the prevalence, correlates and sequencing of electronic cigarette and tobacco use among 11–16-year olds in schools in Wales. BMJ Open 2017; 7:e012784Crossref, Medline, Google Scholar
38. : Protecting youth from the risks of electronic cigarettes. J Adolesc Health 2020; 66:127–131Crossref, Medline, Google Scholar
39. : Notes from the field: e-cigarette use among middle and high school students: United States, 2022. MMWR Morb Mortal Wkly Rep 2022; 71:1283–1285Crossref, Medline, Google Scholar
40. E-Cigarette and Vape Market Size and Share Report, 2030. San Francisco, Grand View Research. https://www.grandviewresearch.com/industry-analysis/e-cigarette-vaping-market Google Scholar
41. : Reasons for current e-cigarette use among US adults. Prev Med 2016; 93:14–20Crossref, Medline, Google Scholar
42. Enforcement Priorities for Electronic Nicotine Delivery Systems (ENDS) and Other Deemed Products on the Market Without Premarket Authorization (Revised): Guidance for Industry. Food and Drug Administration, US Department of Health and Human Services, 2020. https://www.fda.gov/media/133880/download Google Scholar
43. : Flavoured pod attachments score big as FDA fails to enforce premarket review. Tob Control 2020; 29:e129Medline, Google Scholar
44. : Shortcomings of the Food and Drug Administration guidance addressed by Congress 2020 HR 2339. Am J Public Health 2020; 110:776–777Crossref, Medline, Google Scholar
45. : E-liquid flavor preferences and individual factors related to vaping: a survey among Dutch never-users, smokers, dual users, and exclusive vapers. Int J Environ Res Public Health 2019; 16:4661Crossref, Medline, Google Scholar
46. : Preliminary study on the e-liquid and aerosol on the neurobehavior of C elegans. Environ Int 2023; 179:108180Crossref, Medline, Google Scholar
47. : Exposure to e-cigarette aerosol over two months induces accumulation of neurotoxic metals and alteration of essential metals in mouse brain. Environ Res 2021; 202:111557Crossref, Medline, Google Scholar
48. : Short-term e-cigarette toxicity effects on brain cognitive memory functions and inflammatory responses in mice. Toxicol Res 2020; 36:267–273Crossref, Medline, Google Scholar
49. : Lasting synaptic changes underlie attention deficits caused by nicotine exposure during adolescence. Nat Neurosci 2011; 14:417–419Crossref, Medline, Google Scholar
50. : Smoking and cognition. Curr Drug Abuse Rev 2016; 9:76–79Crossref, Medline, Google Scholar
51. : Electronic cigarette use and subjective cognitive complaints in adults. PLoS One 2020; 15:e0241599Crossref, Medline, Google Scholar
52. : Comparison of electronic cigarette refill fluid cytotoxicity using embryonic and adult models. Reprod Toxicol 2012; 34:529–537Crossref, Medline, Google Scholar
53. : Comparing the cytotoxicity of electronic cigarette fluids, aerosols and solvents. Tob Control 2018; 27:325–333Crossref, Medline, Google Scholar
54. : How menthol alters tobacco-smoking behavior: a biological perspective. Yale J Biol Med 2015; 88:279–287Medline, Google Scholar
55. : Menthol enhances nicotine reward-related behavior by potentiating nicotine-induced changes in nAChR function, nAChR upregulation, and DA neuron excitability. Neuropsychopharmacology 2017; 42:2285–2291Crossref, Medline, Google Scholar
56. : Menthol cigarettes: moving toward a broader definition of harm. Nicotine Tob Res 2010; 12 Suppl 2:S85–S93Crossref, Medline, Google Scholar
57. : Cessation outcomes among treatment-seeking menthol and nonmenthol smokers. Am J Prev Med 2012; 43:S242–S248Crossref, Medline, Google Scholar
58. : Green apple e-cigarette flavorant farnesene triggers reward-related behavior by promoting high-sensitivity nAChRs in the ventral tegmental area. eNeuro 2020; 7:ENEURO.0172–20.2020Crossref, Google Scholar
59. : Why flavored vape products may be attractive: green apple tobacco flavor elicits reward-related behavior, upregulates nAChRs on VTA dopamine neurons, and alters midbrain dopamine and GABA neuron function. Neuropharmacology 2019; 158:107729Crossref, Medline, Google Scholar
60. : The vape has gone to your head. Brain Behav Immun 2020; 89:5–6Crossref, Medline, Google Scholar
61. : Maternal e-cigarette use can disrupt postnatal blood-brain barrier (BBB) integrity and deteriorates motor, learning and memory function: influence of sex and age. Fluids Barriers CNS 2023; 20:17Crossref, Medline, Google Scholar
62. : Prevalence and distribution of electronic cigarette use before and during pregnancy among women in 38 states of the United States. Nicotine Tob Res 2021; 23:1459–1467Crossref, Medline, Google Scholar
63. : The use of electronic cigarettes in pregnancy: a review of the literature. Obstet Gynecol Surv 2018; 73:544–549Crossref, Medline, Google Scholar
64. : E-cigarette use patterns and high-risk behaviors in pregnancy: behavioral risk factor surveillance system, 2016–2018. Am J Prev Med 2020; 59:187–195Crossref, Medline, Google Scholar
65. : Microparticles: from biogenesis to biomarkers and diagnostic tools in cardiovascular disease. Curr Stem Cell Res Ther 2017; 12:89–102Crossref, Medline, Google Scholar
66. : Plasma endothelial microvesicles and their carrying miRNA-155 serve as biomarkers for ischemic stroke. J Neurosci Res 2020; 98:2290–2301Crossref, Medline, Google Scholar



