Methamphetamine Dependence Is Associated With Neurocognitive Impairment in the Initial Phases of Abstinence
Abstract
This study documented the association between neurocognitive impairment and methamphetamine dependence in a sample of 27 methamphetamine-dependent individuals who achieved 5 to 14 days of continuously monitored abstinence and in 18 control subjects. Methamphetamine-dependent individuals performed significantly worse than control subjects on neurocognitive measures sensitive to attention/psychomotor speed, on measures of verbal learning and memory, and on executive systems measures sensitive to fluency. These findings are the first to demonstrate that methamphetamine dependence is associated with impairments across a range of neurocognitive domains in a sample of users whose abstinence was continuously monitored with the use of urine screening.
Epidemiological studies show that methamphetamine use has increased to the point that it is now a major national public health concern.1 The increased prevalence and legal and social consequences of this problem have been well documented; however, other possible consequences, such as the neurocognitive impairment, have been largely neglected. A rich preclinical literature using animal models of methamphetamine-induced neurotoxicity suggests that neurocognitive consequences can be expected in human subjects. Early studies showed that relatively large doses of methamphetamine produced marked neurophysiological changes, such as altered striatal dopamine function, which were evident even three to four years later.2–5 Recent studies using relatively small doses over shorter periods have reported persistent changes in dopaminergic functioning6–8 and deficits on measures of working memory.9
In studies of human subjects, the psychiatric10–12 and neurological13,14 consequences of methamphetamine use have received attention in the literature; in contrast, limited data are available on whether methamphetamine dependence is a risk factor for neurocognitive impairment. One recent study demonstrated that methamphetamine dependence is associated with memory deficits and motor slowing in individuals who were abstinent for up to one year.15 Another study suggested that methamphetamine-dependent subjects resembled patients with frontal lobe damage in their performance on a novel test of decision making.16 Although the study was informative, it used a single neurocognitive measure. Another study recruited an unusually young sample of methamphetamine-dependent individuals (mean age, 19 years) and used a battery that was relatively insensitive to frontal, temporal, and subcortical impairments.17
Hence, key questions remain unanswered, including the effects of methamphetamine dependence across neurocognitive domains. The purpose of this study was to determine whether methamphetamine dependence is associated with neurocognitive impairment across a range of measures in users whose abstinence was continuously monitored.
METHOD
Participants
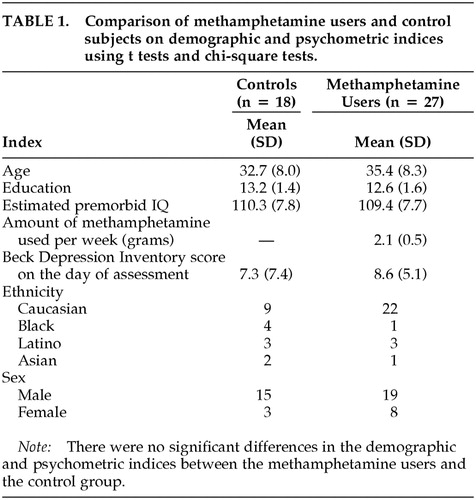
Participants included 27 non-treatment-seeking, methamphetamine-dependent subjects and 18 control subjects recruited from the community through advertisements in local newspapers. Potential participants were excluded if they had a history of stroke, traumatic brain injury with loss of consciousness exceeding 20 minutes, epilepsy, attention deficit disorder, or HIV seropositivity. The Structured Clinical Interview for DSM-IV (SCID-IV)18 was used to rule out the presence of Axis I psychotic or mood disorders. Participants gave written informed consent after being apprised of the study risks and were reimbursed for participation.
The drug-dependent sample met DSM-IV criteria for methamphetamine dependence on the basis of the SCID-IV. Subjects may have used other substances but did not meet DSM-IV criteria for dependence on them, either currently or previously. They reported using at least 0.5 grams of methamphetamine per week for the 6 months preceding the study. The primary route of methamphetamine administration for the drug users was either insufflation (snorting) or smoking. Non-drug-using controls did not meet DSM-IV criteria for abuse or dependence, currently or in the past.
While enrolled in the study, participants were asked to discontinue use of methamphetamine. Participants were included in the study only if 1) on the day of a screening examination that was conducted within 2 weeks of the assessment, their urine tested positive for methamphetamine and tested negative for other drugs, such as cocaine, marijuana, opiates, PCP, and alcohol; 2) they produced a urine sample that was negative for methamphetamine and other drugs on the day in which the neurocognitive measures were administered; and 3) they were not experiencing clinically significant levels of withdrawal symptoms, such as insomnia, reduced appetite, or a mood disorder. The negative urine test indicated that subjects had ceased using methamphetamine at least 5 days before the neurocognitive assessment was conducted, given methamphetamine's elimination half-life of approximately 12 hours. This was considered to be a sufficient length of time for the “crash” phase—which consists of symptoms such as dysphoria, slowing, and agitation—to resolve.19
Procedures
A 2.5-hour battery of neurocognitive measures was administered to each participant, with breaks allowed during that period as needed. The screening measures included the SCID-IV, the Beck Depression Inventory (BDI),20 and the North American Adult Reading Test;21 the latter served as an estimate of premorbid intellectual functioning. The SCID was administered by a master's-level clinician who had completed a 6-week standardized course on the SCID and was certified to administer it.
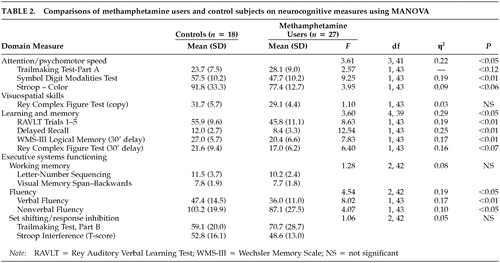
The neurocognitive assessment battery included the following measures: the Controlled Oral Word Association;22 the Letter-Number Sequencing and the Visual Memory Span subtests of the Wechsler Memory Scale–III (WMS-III);23 the Ruff Figural Fluency Test;24 the Rey Auditory Verbal Learning Test;25 the Logical Memory subtest of the WMS-III; the Rey Complex Figure Test;26 the Stroop Color-Word Test;27 the Symbol Digit Modalities Test;28 and the Trailmaking Test, Parts A and B.22 The battery focused on the following domains: attention/psychomotor speed (e.g., Trailmaking Test, Part A; Symbol Digit Modalities Test; Stroop Color), visuospatial skills (Rey Complex Figure Task—copy subtest), learning and memory (Rey Auditory Verbal Learning Test—learning over five trials and delayed recall; WMS-III Logical Memory—delayed recall; Rey Complex Figure Test—delayed recall), and executive systems functioning (untimed working memory, set shifting/response inhibition, fluency). The untimed working memory measures included the Letter-Number Sequencing and the Visual Memory Span—backwards subtests of the WMS-III. The set shifting/response inhibition tasks included the Trailmaking Test, Part B, and a ratio that compared performance on the Color and Word subtests to the Color-Word subtest. Measures of fluency included the Controlled Oral Word Association and the Ruff Figural Fluency Test.
Data Analysis
Multivariate analysis of variance (MANOVA) was used to determine group differences in each of the four neurocognitive domains. For each of the three MANOVAs, the maximum P value was set at 0.05 (two-tailed test), as is recommended when conducting multiple comparisons.29 If the MANOVAs were significant, follow-up univariate ANOVAs were conducted to identify the particular measures that were most sensitive to methamphetamine dependence. The percentage of the variance explained was calculated with eta-squared (η2).
RESULTS
Table 1 shows that the methamphetamine-dependent individuals and the non-drug-using control subjects did not differ in terms of age, education, estimated level of premorbid intellectual functioning, or severity of self-reported depressive symptomatology. Therefore, covariates were not included in subsequent group comparisons.
Table 2 reveals that methamphetamine users showed significantly poorer performance than control subjects on measures of attention/psychomotor speed, on measures of verbal learning and memory, and on fluency-based measures of executive systems functioning (i.e., fluency, set shifting/inhibition). On a measure of nonverbal learning and memory, a trend was observed indicating that methamphetamine-dependent individuals performed poorly relative to non-drug-using controls. The groups did not differ on a measure of visuospatial skills, untimed tests of working memory, or measures of set shifting/response inhibition. The magnitudes of the significant associations ranged from moderate (η2>0.19) to large (η2>0.29). Table 2 also shows consistency in the effects across the neurocognitive tests that constituted each domain.
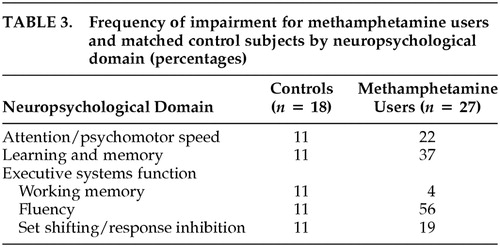
A separate set of analyses determined whether the groups differed in the number of individuals who would be classified as impaired. Participants were considered to be impaired within a particular neurocognitive domain if their score was at least two standard deviations below the mean (according to published norms) on at least one test. For each test, this determination was based on scores adjusted for age, education, and gender. Miller and colleagues have used a similar algorithm.30
Table 3 lists the percentage of individuals in each group who met criteria for impairment in each neurocognitive domain. Although the groups did not differ on untimed measures of working memory or set shifting/response inhibition, moderate group differences in classification rates of impairment were observed on measures of attention/psychomotor speed and learning and memory. Methamphetamine-dependent individuals were much more likely than non-drug-users to be classified as impaired on measures of fluency.
DISCUSSION
To our knowledge, these findings are the first to demonstrate that methamphetamine dependence is associated with impairments across a range of neurocognitive domains, including attention/psychomotor speed, learning and memory, and fluency-based measures of executive systems functioning, in a sample of users whose abstinence was monitored with urine screening. Moreover, methamphetamine-dependent individuals were more likely than non-drug-users to be classified as impaired in the areas of attention/psychomotor speed, learning and memory, and executive systems functioning. Furthermore, the differential performance across the test and control groups was not attributable to demographic profile, estimated premorbid IQ, and level of self-reported depression.
The degree of impairment in this sample of methamphetamine users is substantial, and it is greater than has been observed in neurocognitive studies of cocaine dependence31,32 and even alcohol dependence (without Korsakoff's syndrome).33 Because this study used a cross-sectional design, however, causality cannot be inferred. It is also possible that a single factor that we did not measure produced a liability to develop methamphetamine dependence and neurocognitive impairment.34
Although it is possible that the observed neurocognitive deficits were the result of a transient “crash” phase or the residual symptoms of withdrawal, we used a number of precautions to reduce the likelihood that such factors would affect test performance. For example, urine screening was used to monitor abstinence from drug use. Furthermore, methamphetamine-dependent study participants reported only minimal levels of dysphoric mood, agitation, insomnia, or slowness of movement—the cardinal symptoms of stimulant withdrawal—on the day on which the neurocognitive measures were administered. The latter finding is consistent with a recently published report showing that symptoms of methamphetamine withdrawal resolve to minimal levels by the 5th day of abstinence.19
It is important to note that no controlled studies have documented the time course of methamphetamine withdrawal in human subjects. Although we considered a host of factors that potentially moderate performance on neurocognitive tests, preclinical research has shown that the dopaminergic system is affected for months after cessation of methamphetamine use.6,7 Therefore, undetected factors related to withdrawal might have affected test performance for this sample. Conversely, in a sample of 15 detoxified methamphetamine-dependent individuals, Volkow and colleagues reported that methamphetamine-dependent individuals demonstrate poor test performance even after 12 months of abstinence.15 Their work suggests that the impairments observed during the early phases of abstinence are relatively stable.
While these findings preliminarily show that methamphetamine dependence is associated with neurocognitive impairment, several limitations should be noted. The sample size was not large enough to identify risk factors or protective factors that might distinguish between impaired and nonimpaired methamphetamine-dependent subjects. Likely factors that should be considered in future studies include the amount, frequency, and duration of use of methamphetamine, and demographic factors, such as age, education, and socioeconomic status. The sample size also limits the degree to which these findings are generalizable to methamphetamine-dependent individuals with different demographic and drug use profiles. The period of abstinence for our study subjects, 5 to 14 days, constitutes another potential limitation. Furthermore, although the SCID-IV was used to rule out comorbid psychiatric illness, it is possible that the interview did not detect abuse of other substances. In addition, it is possible that subclinical withdrawal symptoms might have affected test performance. Longitudinal assessment of abstinent methamphetamine-dependent individuals will provide important data on the durability of these deficits.
It is likely that the severity of the neurocognitive impairment observed in this sample is associated with worse functional outcomes, including poorer vocational functioning and an elevated risk of relapse to dependence. Although the association between neurocognition and functional outcomes has not been examined in methamphetamine-dependent individuals, or in users of any drugs, it has been documented in other disorders. For example, in individuals with HIV, neurocognitive deficits have been linked to poor vocational functioning.35,36 Similar findings have been reported for samples of individuals diagnosed as having schizophrenia.37 In the schizophrenia literature, these deficits are associated with an inability to learn skills during the course of rehabilitation or to implement learned skills. Neurocognitive impairment may undermine the effectiveness of psychosocial treatments for methamphetamine dependence as well, suggesting that this concern is an important area of study.
ACKNOWLEDGMENTS
Supported by grants from the National Institute on Drug Abuse (DA50038, DA00388, and DA07272) and from the Universitywide AIDS Research Program of the University of California (CC-96-175). Presented in part at the College on Problems on Drug Dependence, in Scottsdale, Arizona, June 12–17, 1998.
 |
 |
 |
1 Cunningham JK, Thielemeir MA: Amphetamine-Related Emergency Admissions: Trends and Regional Variations in California (1985-1994). Irvine, CA, Public Statistics Institute, 1996Google Scholar
2 Ricaurte GA, Schuster CR, Seiden LS: Long-term effects of repeated methylamphetamine administration on dopamine and serotonin neurons in the rat brain: a regional study. Brain Res 1980; 193:153-163Crossref, Medline, Google Scholar
3 Seiden LS, Fischman MW, Schuster CR: Long-term methamphetamine-induced changes in brain catecholamines in tolerant rhesus monkeys. Drug Alcohol Depend 1976; 1:215-219Crossref, Medline, Google Scholar
4 Seiden LS, Fischman MW, Schuster CR: Changes in brain catecholamines induced by long-term methamphetamine administration in rhesus monkeys, in Cocaine and Other Stimulants. Edited by Ellinwood EH Jr, Kilbey MM. New York, Plenum Press, 1977, pp 179-185Google Scholar
5 Woolverton WL, Ricaurte GA, Forno LS, et al: Long-term effects of chronic methamphetamine administration in rhesus monkeys. Brain Res 1989; 486:73-78Crossref, Medline, Google Scholar
6 Melega WP, Quintana J, Raleigh MJ, et al: 6-[18F]fluoro-L-DOPA-PET studies show partial reversibility of long-term effects of chronic amphetamine in monkeys. Synapse 1996; 22:63-69Crossref, Medline, Google Scholar
7 Melega WP, Raleigh MJ, Stout DB, et al: Recovery of striatal dopamine function after acute amphetamine- and methamphetamine-induced neurotoxicity in the vervet monkey. Brain Res 1997; 766(1-2):113-120Google Scholar
8 Villemagne V, Yuan J, Wong DF, et al: Brain dopamine neurotoxicity in baboons treated with doses of methamphetamine comparable to those recreationally abused by humans: evidence from [11C]WIN-35,428 positron emission tomography studies and direct in vitro determinations. J Neurosci 1998; 18:419-427Crossref, Medline, Google Scholar
9 Castner SA, Goldman PS: Profound cognitive impairments in nonhuman primates exposed to amphetamine. Presented at the annual meeting of the Society for Neuroscience, New Orleans, LA, November 4-9, 2000Google Scholar
10 Buffenstein A, Heaster J, Ko P: Chronic psychotic illness from methamphetamine [letter]. Am J Psychiatry 1999; 156:662Crossref, Medline, Google Scholar
11 Gawin FH, Ellinwood EH Jr.: Cocaine and other stimulants: actions, abuse, and treatment. N Engl J Med 1988; 318:1173-1182Crossref, Medline, Google Scholar
12 Iwanami A, Sugiyama A, Kuroki N, et al: Patients with methamphetamine psychosis admitted to a psychiatric hospital in Japan: a preliminary report. Acta Psychiatr Scand 1994; 89:428-432Crossref, Medline, Google Scholar
13 Caplan LR, Hier DB, Banks G: Current concepts of cerebrovascular disease-stroke: stroke and drug abuse. Stroke 1982; 13:869-872Crossref, Medline, Google Scholar
14 Rothrock JF, Rubenstein R, Lyden PD: Ischemic stroke associated with methamphetamine inhalation. Neurology 1988; 38:589-592Crossref, Medline, Google Scholar
15 Volkow ND, Chang L, Wang GJ, et al: Association of dopamine transporter reduction with psychomotor impairment in methamphetamine abusers. Am J Psychiatry 2001; 158:377-382Crossref, Medline, Google Scholar
16 Rogers RD, Everitt BJ, Baldacchino A, et al: Dissociable deficits in the decision-making cognition of chronic amphetamine abusers, opiate abusers, patients with focal damage to prefrontal cortex, and tryptophan-depleted normal volunteers: evidence for monoaminergic mechanisms. Neuropsychopharmacology 1999; 20:322-339Crossref, Medline, Google Scholar
17 Trites RL, Suh M, Offord D, et al: Neuropsychologic and psychosocial antecedents and chronic effects of prolonged use of solvents and methamphetamine: I. group profiles. Psychiatric Journal of the University of Ottawa 1976; 1(1 suppl 2):14-20Google Scholar
18 Spitzer R, Williams J, Gibbon M: Structured Clinical Interview for DSM-IV (SCID). New York, New York State Psychiatric Institute, Biometrics Research, 1995Google Scholar
19 Srisurapanont M, Jarusuraisin N, Jittiwutikan J: Amphetamine withdrawal: I. reliability, validity, and factor structure of a measure. Aust N Z J Psychiatry 1999; 33:89-93Crossref, Medline, Google Scholar
20 Beck AT, Ward CH, Mendelson J, et al: The Beck Depression Inventory. Arch Gen Psychiatry 1968; 4:561-571Crossref, Google Scholar
21 Nelson HE: National Adult Reading Test (NART): Test Manual. Windsor, England, NFER Nelson, 1982Google Scholar
22 Spreen O, Strauss E: A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary. New York, Oxford University Press, 1998Google Scholar
23 Wechsler D: Wechsler Memory Scale-III. San Antonio, TX, Psychological Corporation, 1997Google Scholar
24 Ruff RM, Light RM, Evans RW: Ruff Figural Fluency Test: A normative study with adults. Developmental Neuropsychology 1987; 3:37-51Crossref, Google Scholar
25 Rey A: L'examen clinique en psychologie. Paris, Presses Universitaires de France, 1964Google Scholar
26 Rey A: L'examen psychologique dans les cas d'encephalopathie traumatique. Archives de Psychologie 1941; 28:286-340Google Scholar
27 Stroop J: Studies of interference in serial verbal reactions. Journal of Experimental Psychology 1935; 18:643-662Crossref, Google Scholar
28 Smith A: Symbol Digit Modalities Test (SDMT). Los Angeles, Western Psychological Services, 1982Google Scholar
29 Keppel G: Design and Analysis: A Researcher's Handbook. Englewood Cliffs, NJ, Prentice-Hall, 1982Google Scholar
30 Miller EN, Selnes OA, McArthur JC, et al: Neuropsychological performance in HIV-1 infected homosexual men: the Multicenter AIDS Cohort Study (MACS). Neurology 1990; 40:197-203Crossref, Medline, Google Scholar
31 Van Gorp WG, Wilkins JN, Hinkin CH, et al: Declarative and procedural memory functioning in abstinent cocaine abusers. Arch Gen Psychiatry 1999; 56:85-89Crossref, Medline, Google Scholar
32 Bolla KI, Rothman R, Cadet JL: Dose-related neurobehavioral effects of chronic cocaine use. J Neuropsychiatry Clin Neurosci 1999; 11:361-369Link, Google Scholar
33 Rourke SR, Loberg T: Neurobehavioral correlates of alcoholism, in Neuropsychological Assessment of Neuropsychiatric Disorders, vol 3. Edited by Adams IGK. New York, Oxford University Press, 1996, pp 423-485Google Scholar
34 Malow RM, Pintard PF, Sutker PB, et al: Psychopathology subtypes: drug use motives and patterns. Psychol Addict Behav 1988; 2:1-13Crossref, Google Scholar
35 Heaton RK, Velin RA, McCutchan JA, et al: Neuropsychological impairment in human immunodeficiency virus infection: implications for employment. HNRC Group, HIV Neurobehavioral Research Center. Psychosom Med 1994; 56:8-17Crossref, Medline, Google Scholar
36 Heaton RK, Marcotte TD, White DA, et al: Nature and vocational significance of neuropsychological impairment associated with HIV infection. Clin Neuropsychol 1996; 10(1):1-14Google Scholar
37 Green MF: Schizophrenia from a neurocognitive perspective: probing the impenetrable darkness. Boston, Allyn and Bacon, 1998Google Scholar



