Associations Between Neuroanatomic Patterns of Cerebral Infarctions and Vascular Dementia
Abstract
Objective:
A history of multiple cerebral infarctions is generally regarded as an important risk factor for vascular dementia. The authors examined the risk of vascular dementia in patients with multiple acute ischemic lesions.
Methods:
The authors conducted a hospital-based prospective study of 11,200 patients with first-time stroke who underwent 1.5 or 3-T MRI and a global cognitive assessment. Univariate and multivariate logistic regression analyses estimated the risk of dementia associated with multiple lesions versus a single lesion.
Results:
Having multiple lesions, compared with having a single lesion, was significantly associated with dementia in patients with stroke (odds ratio=5.83, 95% CI=5.08, 6.70; p<0.001). The apoliproprotein ε4 allele was more frequent in patients with multiple lesions than in those with a single lesion (odds ratio=1.70, 95% CI=1.39, 2.07; p<0.001). Severe leukoaraiosis (odds ratio=15.77, 95% CI=8.38, 29.68; p<0.001) and microbleedings (odds ratio=1.31, 95% CI=1.06, 1.63; p<0.01) were strong confounders for dementia in the multivariate analysis. Multiple logistic regression analysis showed that multiple lesions in one hemisphere versus a single lesion (odds ratio=2.14, 95% CI=1.83, 2.51; p<0.001), involvement of strategic regions (odds ratio=4.73, 95% CI=4.07, 5.49; p<0.001), and stroke lesion volume (odds ratio=1.31, 95% CI=1.03, 1.66; p=0.03) were significantly associated with dementia. There was a preponderance of lesions on the left side in patients with dementia (odds ratio=2.56, 95% CI=2.11, 3.11; p<0.001).
Conclusions:
Multiple spontaneous anterior or posterior circulation lesions after stroke increase a patient’s risk of developing dementia. Recognition of multiple ischemic lesions after stroke may allow targeted rapid therapeutic interventions to prevent subsequent cognitive deterioration.
Vascular dementia is recognized as one of the most common causes of major cognitive impairment after Alzheimer’s disease (1–3). Currently, the lesional markers of vascular dementia and their relative contributions are better known. Several brain MRI markers, such as stroke subtype (ischemic or hemorrhagic), stroke volume and gross location (4, 5), recurrent strokes (2, 6), the presence and extent of white matter hyperintensities (7, 8), brain microbleeds (9, 10), and medial temporal lobar atrophy (11), may produce complex neurocognitive symptoms and dementia. Multiple brain infarcts have also been suggested to be an important risk factor for dementia.
The term “multiple brain infarcts” has been used to define a variety of multiple ischemic lesions that appear in one or both hemispheres or in the anterior and posterior circulation territories (multilevel) (12). Multiple ischemic lesions in different parts of the brain may present specific clinical patterns as described in previous studies (13, 14). A large body of evidence suggests that simultaneous multiple cortical and subcortical ischemic lesions in the anterior and posterior circulation are linked to the incidence of cognitive decline among healthy people (15, 16). Despite these new insights, to our knowledge, there are no systematic studies of stroke cohorts investigating associations between simultaneous multiple infarcts and cognitive decline among dementia-free individuals.
In this study, we aimed to clarify the risk of dementia after first stroke in the anterior and posterior circulation territories in a large, prospective stroke cohort.
Methods
Between September 2004 and December 2017, 11,200 patients with ischemic stroke were prospectively included from the Ege University Medical Center. Details of the study cohort have been described previously (17). All patients who were hospitalized (<30 days) for ischemic stroke, with initial positive imaging, a reliable informant from among caregivers or family members, and no previously diagnosed disorders affecting cognition, were included. Clinical, neuropsychological, and MRI examinations were performed during the first month of participant enrollment in the study. Baseline examinations and definitions of vascular disease (18) and risk factors are described briefly below. The prior cognitive status of each patient was requested from his or her primary care physician or from Turkey’s National Health Data system.
Prospectively recorded variables included age, gender, risk factors, blood pressure, ischemic stroke characteristics, location, pathogenesis, disability (determined using the National Institutes of Health (NIH) Stroke Scale [19] and the modified Rankin Scale [20]). clinical subtypes, topography of infarcts on diffusion-weighted imaging or fluid attenuation inversion recovery (FLAIR) MRI studies, and neurologic and systemic complications. The combination of computerized tomography (CT) scan and angiography was the routine of our stroke unit for all patients with stroke. MRI was performed within 24 hours of admission by using 1.5- and 3-T scanners (Siemens Sonata, Siemens Medical Solutions, Erlangen, Germany). To differentiate acute from chronic lesions, T1 and T2, diffusion-weighted imaging and FLAIR MRI sequences were used concomitantly. The diffusion-weighted imaging sequence was obtained using echoplanar imaging (field of view=230 mm; slice thickness=5 mm; repetition time [TR] 3,200 ms; echo time [TE]=94 ms). All CT and MRI scans were reviewed by two neuroradiologists with extensive CT and MRI experience who were blind to clinical status. The kappa coefficients (interexaminer reliabilities) for lesions including infarct, leukoaraiosis, brain microbleeds, and dilated perivascular spaces were 0.98, 0.97, 0.96, and 0.98, respectively, for FLAIR and diffusion-weighted imaging.
The study was approved by the ethics committee of the Ege University Medical Center, and written informed consent was obtained from all participants.
Assessment of Poststroke Cognitive Status
The evaluation of cognitive function was performed by a neurologist (E.K.) and a clinical neuropsychologist (H.A.) 3 months after stroke, because mental status may be affected by pathophysiological factors during the acute hospitalization, and thus patients may be falsely classified as having vascular dementia. Dementia (referred to as a neurocognitive disorder diagnosis in DSM-5 after 2015) was determined in accordance with DSM-IV and DSM-5 criteria (21). Therefore, the diagnosis of dementia or major neurocognitive disorder was made if cognitive deficits interfered with the patient’s independence in everyday activities and if significant cognitive decline from a previous level of performance in one or more cognitive domains was present. Normal cognitive status was determined using evidence of normal cognitive performance in cognitive domains without interference with the capacity for independence in everyday activities, which was measured by the Bristol Activities of Daily Living Scale (22). Global mental function was evaluated in each patient using the Mini-Mental State Examination (MMSE) or Montreal Cognitive Assessment (MoCA) batteries (23).
Lesion Patterns
We established diagnostic MRI criteria to identify multiple cerebral infarcts in different arterial territories, defined as at least two diffusion-weighted imaging or FLAIR lesions in at least two different arterial territories (right or left anterior or posterior cerebral circulation) (18, 19). Multiple acute infarcts in one cerebral hemisphere was considered in patients who had infarcts with combinations involving the territories of the middle cerebral artery, the anterior cerebral artery, and the anterior choroidal artery. Multiple acute bihemispheric infarcts in the anterior circulation were presumed in patients with acute multiple infarcts involving both hemispheres with lesions either in superficial or deep arterial territories. Multiple acute infarcts in the posterior circulation were determined in patients with concomitant infarcts in the territory of the vertebral arteries, the basilar artery, the superior cerebellar artery, the anterior inferior cerebellar artery, the posterior inferior cerebellar artery, and the posterior cerebral artery. Multiple acute infarcts in the anterior and posterior circulation were considered in patients with combined supratentorial and infratentorial infarcts.
The locations of strategic regions as defined previously (3, 15, 16) were determined by two neuroradiologists with extensive CT and MRI experience who were blind to clinical status. These strategic sites included large foci (>1.5 cm) within the following regions: the middle and inferior frontal gyri, parietal region, middle temporal gyrus, the putamen and pallidum, cingulate gyrus, corpus callosum, arcuate fasciculus, middle occipital region and hippocampus, thalamus, midbrain, and postero-inferior cerebellum. The stroke lesion volume was determined using STandards for ReportIng Vascular changes on nEuroimaging criteria and a previously validated method (16). White matter hyperintensity burden (leukoaraiosis) was assessed using the Fazekas scale score (24) on the basis of FLAIR sequence. Brain microbleeds were defined as small (diameter, <10 mm) areas of signal void with associated blooming seen on T2*-weighted MRI, according to the Brain Observer MicroBleed Scale criteria (25). T2 sequence was used to assess the presence of dilated perivascular spaces.
Stroke Mechanisms
Etiology was determined by using the Trial of Org 10172 in Acute Stroke Treatment classification (18) and classified as large-artery atherosclerosis, cardioembolism, small-vessel disease, or other and unknown causes. Large-artery disease was presumed in patients with carotid artery stenosis >50% and diffusion-weighted imaging demonstrating multiple infarcts distal to the stenosed vessel in the territory of the relevant artery. Small-artery disease or local branch occlusion was defined in patients with infarcts <15 mm in diameter localized in the deep regions of the brain or in the brainstem without large-artery disease and cardioembolism. Cardiac embolism was determined in patients having a source of embolism. Strokes of other determined or undetermined causes were also recorded.
Concomitant Risk Factors
Hypertension, diabetes mellitus, hyperlipidemia, and current cigarette smoking, were recorded. Apolipoprotein E (ApoE) genotype was determined by polymerase chain reaction amplification of leukocyte DNA. Participants were classified by the presence versus absence of at least one ApoE ε4 allele.
Statistical Analysis
The prevalence of dementia was calculated as the ratio of the number of patients with dementia to the total number of testable patients with first-ever stroke. Proportions, odds ratios, and means of baseline characteristics were compared between patients with and without dementia using the chi-square test, logistic regression, and analysis of variance, respectively. Univariate associations among age, sex, vascular risk factors, ApoE ε4 allele, stroke subtypes, single versus multiple lesions in the anterior and posterior circulation, white matter lesions, microbleedings, and dementia were analyzed using logistic regression to estimate unadjusted odds ratios. In the second analysis, we introduced into the model all potential confounders with a probability value <0.20 in univariate analysis, and multiple logistic regression analysis by forward stepwise method (likelihood ratio) was used to find the association of dementia with single versus multiple lesions in different territories. Association between a single lesion and multiple lesions in different territories with dementia was sought to estimate models using multiple logistic regression analysis by including all lesions (model 1) and by excluding (model 2) Fazekas scale scores and microbleedings. Probability values <0.05 were considered statistically significant. All statistical analyses were performed using SPSS, version 18.0.
Results
Of the 11,200 patients with ischemic stroke, 8,700 patients were eligible for neuroimaging evaluation and cognitive examination for the final analysis. Patients with old infarcts or old hemorrhagic lesions on imaging were excluded. None of the eligible patients had previously diagnosed diseases affecting cognition. Among 8,700 patients (mean age: 66 years [SD=12]; women, N=4,219 [48%]), 3,117 (36%) had multiple lesions involving anterior circulation, posterior circulation, or both on brain imaging. Vascular dementia developed in 1,048 (12%) patients; those with multiple lesions had more frequent dementia (27%) compared with patients with a single lesion (6%) (odds ratio=5.83, 95% CI=5.08, 6.70; p<0.001). Multiple acute infarcts in one cerebral hemisphere were present in 1,466 patients (17%), multiple acute bihemispheric infarcts in the anterior circulation in 612 patients (7%), multiple lesions involving posterior circulation in 663 patients (8%), and multiple lesions involving both anterior and posterior circulation in 376 patients (4%) (Table 1, Figure 1).
| With vascular dementia (N=1,048) | Without vascular dementia (N=7,652) | ||||
|---|---|---|---|---|---|
| Characteristic | N | % | N | % | p |
| Male sex | 543 | 52 | 3,938 | 51 | 0.43 |
| History of transient ischemic attack | 114 | 11 | 334 | 4 | 0.0001 |
| Hypertension | 829 | 79 | 5,562 | 73 | 0.0001 |
| Diabetes mellitus | 333 | 32 | 2,186 | 29 | 0.02 |
| Cigarette smoking | 180 | 17 | 1,401 | 18 | 0.19 |
| Atrial fibrillation | 256 | 24 | 1,312 | 17 | 0.01 |
| Hyperlipidemia | 493 | 47 | 3,463 | 45 | 0.15 |
| ApoE ε4 allele | 284 | 27 | 1,478 | 19 | 0.0001 |
| Leukoaraiosis (Fazekas scale score 1–3) | 613 | 59 | 4,112 | 54 | 0.002 |
| Microbleedings | 138 | 13 | 718 | 9 | 0.0001 |
| Strategic lesions | 551 | 53 | 1,127 | 15 | 0.0001 |
| Cause of stroke | |||||
| Large-artery disease | 295 | 28 | 1,918 | 25 | 0.02 |
| Cardioembolic stroke | 296 | 28 | 1,748 | 23 | 0.0001 |
| Small-artery disease | 207 | 20 | 1,576 | 21 | 0.23 |
| Other causes | 137 | 13 | 1,197 | 16 | 0.002 |
| Unknown cause | 125 | 12 | 1,203 | 16 | 0.001 |
| All single infarcts | 235 | 22 | 5,348 | 70 | 0.0001 |
| All multiple infarcts | 813 | 78 | 2,304 | 30 | 0.0001 |
| MAIOCH | 417 | 40 | 1,049 | 14 | 0.0001 |
| MABIAC | 141 | 14 | 471 | 6 | 0.0001 |
| MAIPC | 147 | 14 | 516 | 7 | 0.0001 |
| MAIAPC | 108 | 10 | 268 | 4 | 0.0001 |
TABLE 1. Demographic and clinical characteristics of patients with and without vascular dementiaa
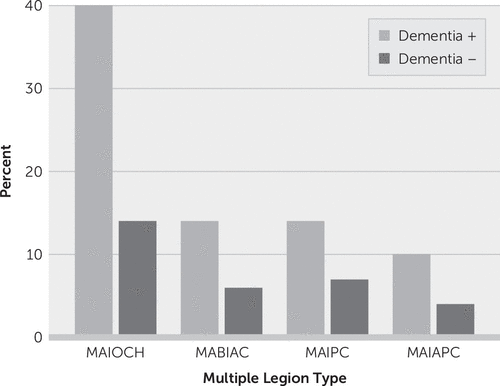
FIGURE 1. Frequency of dementia in patients with multiple infarcts at different locationsa
a MABIAC=multiple acute bihemispheric infarcts in the anterior circulation, MAIAPC=multiple acute infarcts in the anterior and posterior circulation, MAIOCH=multiple acute infarcts in one cerebral hemisphere, MAIPC=multiple acute infarcts in the posterior circulation.
Normative cognitive data from healthy volunteers included a mean MMSE score of 27.7 (SD=1.9) and a mean MoCA score of 24.6 (SD=3.9). For patients with dementia, the mean MMSE score was 16.8 (SD=4.0), and for those without dementia, it was 26 (SD=1.9; p<0.001). For patients with multiple lesions, the mean MMSE score was significantly lower than the mean score for those with a single lesion (23.2 [SD=5.1] versus 26.1 [SD=2.7]; p=0.001).
In the univariate analysis, factors related to dementia with a p value <0.2 were age, sex, hypertension, diabetes mellitus, ApoE ε4 allele, NIH Stroke Scale score at stroke onset, causes of stroke, white matter hyperintensity burden, the presence of microbleeds, and types of multiple infarcts versus a single infarct. These factors were entered into the regression analysis (Table 2). Stepwise linear regression was used to select all types of multiple infarcts that were independently associated with dementia compared with those not associated with dementia (Figure 2). Large-artery disease (odds ratio=1.32, 95% CI=1.01, 1.35; p=0.002) and cardioembolism (odds ratio=1.64, 95% CI=1.37, 1.96; p<0.001) were significantly associated with multiple infarcts in patients with dementia. Assessment of a possible threshold effect for leukoaraiosis burden and the presence of microbleeds did not change the final multivariate analysis findings. The ApoE ε4 allele in patients with dementia was more frequent in those with multiple lesions (17%) than in those with a single lesion (13%) (odds ratio=1.63, 95% CI=1.38, 1.93; p<0.001).
| Analysis | ||||||
|---|---|---|---|---|---|---|
| Univariate | Multivariate | |||||
| Characteristic | Crude odds ratio | 95% CI | p | Adjusted odds ratio | 95% CI | p |
| Age (years) | 1.04 | 1.03, 1.04 | 0.0001 | 1.03 | 1.02, 1.03 | <0.001 |
| Male sex | 1.01 | 0.89, 1.15 | 0.83 | |||
| Hypertension | 1.42 | 1.22, 1.67 | 0.0001 | |||
| Diabetes mellitus | 1.17 | 1.01, 1.34 | 0.03 | |||
| Hyperlipidemia | 1.08 | 0.95, 1.22 | 0.24 | |||
| Atrial fibrillation | 1.56 | 1.34, 1.82 | 0.0001 | 1.32 | 1.11, 1.58 | 0.002 |
| NIHSS score | ||||||
| 1–4 | 0.62 | 0.39, 0.99 | 0.05 | 0.72 | 0.42, 1.17 | 0.18 |
| 5–14 | 0.42 | 0.92, 2.17 | 0.11 | 1.47 | 0.92, 2.35 | 0.11 |
| ≥15 | 1.95 | 1.25, 3.02 | 0.003 | 1.79 | 1.11, 2.90 | 0.02 |
| Leukoaraiosis (Fazekas scale score) | ||||||
| 1 | 1.23 | 1.06, 1.43 | 0.006 | 0.94 | 0.80, 1.12 | 0.48 |
| 2 | 5.39 | 4.45, 6.52 | 0.0001 | 3.43 | 2.75, 4.27 | 0.0001 |
| 3 | 20.94 | 11.94, 36.72 | 0.0001 | 15.77 | 8.38, 29.68 | 0.0001 |
| Presence of microbleedings | 1.47 | 1.21, 1.78 | 0.0001 | 1.31 | 1.06, 1.63 | 0.01 |
| Strategic lesions | 6.42 | 5.59, 7.38 | 0.0001 | 3.27 | 2.76, 3.88 | 0.0001 |
| Stroke lesion volume | 1.04 | 1.034, 1.038 | 0.0001 | 1.044 | 1.035, 1.054 | 0.0001 |
| Cause of stroke | ||||||
| Large-artery disease | 1.17 | 1.01, 1.35 | 0.03 | 1.32 | 1.11, 1.58 | 0.002 |
| Small-artery disease | 0.94 | 0.80, 1.10 | 0.44 | |||
| Cardioembolic stroke | 1.33 | 1.15, 1.54 | 0.0001 | 1.64 | 1.37, 1.96 | 0.0001 |
| Other causes | 0.81 | 0.67, 0.98 | 0.03 | |||
| Unknown cause | 0.73 | 0.59-0.88 | 0.001 | |||
| MAIOCH | 4.16 | 3.62, 4.78 | 0.0001 | 2.59 | 1.40, 4.80 | 0.002 |
| MABIAC | 2.37 | 1.94, 2.90 | 0.0001 | 2.33 | 1.31, 4.15 | 0.001 |
| MAIPC | 2.26 | 1.86, 2.75 | 0.0001 | 2.41 | 1.89, 3.07 | |
| MAIAPC | 3.17 | 2.51, 3.99 | 0.0001 | |||
TABLE 2. Univariate and multivariate logistic regression analysis of clinical stroke subtypes and lesion characteristics of patients with and without vascular dementiaa
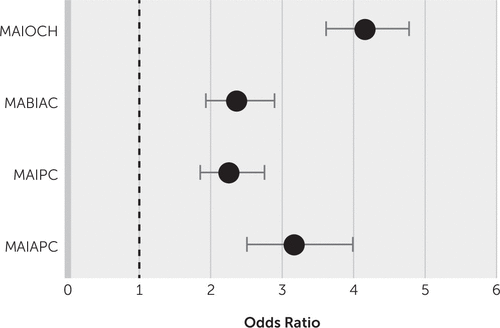
FIGURE 2. Univariate analysis of different multiple lesion locations in patients with dementia versus patients without dementiaa
a MABIAC=multiple acute bihemispheric infarcts in the anterior circulation, MAIAPC=multiple acute infarcts in the anterior and posterior circulation, MAIOCH=multiple acute infarcts in one cerebral hemisphere, MAIPC=multiple acute infarcts in the posterior circulation.
Multiple regression analysis for different location type after the exclusion of confounder factors, such as brain microbleeds and white matter hyperintensity burden, showed that multiple lesions in one hemisphere versus a single lesion (p<0.001), strategic region involvement (p<0.001), and stroke lesion volume (p=0.03) were significantly associated with dementia (Table 3, Figure 3). Left hemispheric lesions were significantly associated with dementia (odds ratio=2.56, 95% CI=2.11, 3.11; p<0.001).
| Variable | Adjusted odds ratio | 95% CI | p |
|---|---|---|---|
| Modelb | |||
| Single hemispheric lesion | Reference | ||
| Multiple hemispheric lesions (MAIOCH) | 1.31 | 1.08, 1.57 | 0.005 |
| Strategic lesions | |||
| 0 | Reference | ||
| ≥1 | 2.09 | 1.73, 2.53 | 0.003 |
| Leukoaraiosis (Fazekas scale) | |||
| 0 | Reference | ||
| 1 | 1.19 | 0.98, 146 | 0.08 |
| 2 | 4.77 | 3.71, 6.29 | 0.0001 |
| 3 | 19.24 | 7.84, 47.23 | 0.0001 |
| Microbleedings | |||
| 0 | Reference | ||
| ≥ 1 | 1.31 | 1.05, 1.62 | 0.01 |
| Stroke lesion volume | |||
| <90 cm3 | Reference | ||
| >91 cm3 | 2.21 | 1.76, 2.77 | 0.0001 |
| Model 2c | |||
| Single hemispheric lesion | Reference | ||
| Multiple hemispheric lesions (MAIOCH) | 2.14 | 1.83, 2.51 | 0.001 |
| Strategic lesions | |||
| 0 | Reference | ||
| ≥1 | 4.73 | 4.07, 5.49 | 0.0001 |
| Stroke lesion volume | |||
| <90 cm3 | Reference | ||
| >91 cm3 | 1.31 | 1.03, 1.66 | 0.03 |
TABLE 3. Variables independently associated with vascular dementia in multiple logistic regression in the study cohorta
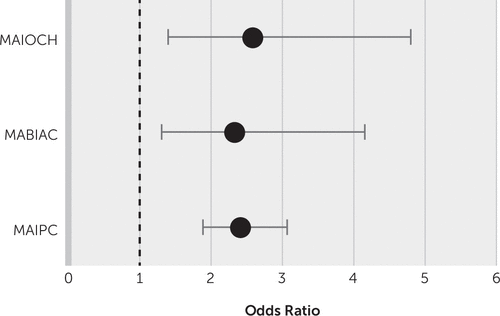
FIGURE 3. Multivariable analysis of different location types and other risk factors in patients with dementia compared with patients without dementiaa
a MABIAC=multiple acute bihemispheric infarcts in the anterior circulation, MAIAPC=multiple acute infarcts in the anterior and posterior circulation, MAIOCH=multiple acute infarcts in one cerebral hemisphere, MAIPC=multiple acute infarcts in the posterior circulation.
Discussion
The clinical syndrome of vascular dementia developed in 12% of patients in this study cohort, whereas dementia was four times more frequent in patients with multiple lesions compared with those with a single lesion. The main findings of this hospital-based study were that dementia developed through many different locations of multiple lesions involving anterior and posterior circulation. Minor neurocognitive disorder due to multiple lesions is the topic of another study conducted by our group.
The presence of brain microbleeds and the leukoaraiosis burden (especially grades 2 and 3) were found to have a strong association with dementia. This prominent effect of microbleeds and the leukoaraiosis burden in univariate and multivariate analyses may indicate their contribution to dementia in some multiple lesion subtypes. Contrary to our finding, a recent prospective study did not find any evidence of a threshold effect for the leukoaraiosis burden or the number of microbleeds (26). Our results and previous findings suggest that leukoaraiosis and brain microbleeds make a contribution, to a certain extent, to dementia in the stroke population (9, 10, 27).
Overall, our data indicate that large-artery disease and cardioembolism are the most prominent factors for poststroke cognitive disorders. This finding emphasizes the fact that these pathologies play a key role in the occurrence of dementia by causing multifocal emboli involving either bilateral or multilevel structures. Small-artery disease was not a significant factor for poststroke dementia in the multivariate analysis. However, limited and strategic lesions in small subcortical structures (e.g., the bilateral paramedian thalamus, caudate nucleus, left arcuate fasciculus, left middle frontal gyrus, and left postero-inferior cerebellum) have been reported as the main determinants of poststroke cognitive impairment, explained by disruption of cortico-subcortical pathways (28–31).
There are some limitations to this study. First, in a recruitment time period of approximately 15 years, there was noteworthy progress in the MRI techniques and software to investigate brain structures and lesion volume (32, 33). We found that stroke lesion volume was likely larger in the multiple infarcts, but we did not determine cerebral atrophy, which could be assessed by using three-dimensional T1-weighted images with fully automated software, which is one of the most controversial MRI markers with regard to the influence on poststroke cognitive impairment (26, 34, 35). The mechanism of poststroke brain atrophy has not been well understood, but postischemic secondary neurodegeneration in the subcortical region is probably one of the main contributors (36). Second, multivariate analysis showed that small lesions (<15 mm) were not associated with dementia either in patients with bilateral infarcts or in those with multilevel infarcts. However, small lesions were visually determined by an experienced neuroradiologist, and improper measurements would have decreased the strength of the relationship with dementia. Moreover, a risk of uncertainty in the measurement of small lesions using specifically automated software methods has been reported (32). Third, we did not assess cortical microinfarcts, which may be an independent risk factor for cognitive disorder in older populations (37). Fourth, we used a global mental assessment test to explore cognitive performance. Global mental scores are significantly lower among individuals with multiple lesions compared with those with a single lesion. However, the present study was restricted to the presence of dementia, and we did not analyze subcognitive domains, such as language, executive function, and memory functions, as continuous variables. The association between multiple ischemic lesions and cognitive domains requires further investigation. In addition to the traditional vascular risk factors examined in this cohort, other contributing factors, such as obstructive sleep apnea (38), hyperhomocysteinemia from common cobalamin deficiency in the elderly, and genetic (MTHFR and CTH gene polymorphisms) and dietary factors, are potential causes of multiple infarcts associated with dementia.
This study has many strengths. First, the large sample size enabled us to define many different multiple infarct patterns and feed many factors into the regression model. Our study provides an original and reliable approach using detailed clinical mental status examination and neuroimaging examination to assess multiple and multilevel involvements of brain structures in a large stroke population. Second, we analyzed most of the likely radiological determinants of poststroke cognitive performance, and we delineated the importance of multiple lesions in the anterior and posterior circulation. Third, the study showed a strong association between strategic sites (i.e., the thalamus; angular gyrus; basal ganglia, including the caudate nucleus and globus pallidus; middle and inferior frontal gyri; parietal region; and middle temporal gyrus or postero-inferior cerebellum) and dementia, which may be an independent risk factor for cognitive impairment. Specific clinical pictures have been defined previously in patients with multiple ischemic lesions involving strategic regions either in one hemisphere or in both (12, 13). A large body of evidence suggests that lesions in strategic areas play a key role in the occurrence of neurocognitive disorder, regardless of their volume (26), but the associations between strategic site and cognitive domains warrant further investigation that are outside the topic of our study. Fourth, we chose to study only ischemic lesions so that our study cohort was representative of the homogeneous pathology of the clinical population. The study design prevented us from specifically analyzing stroke subtypes, such as subarachnoid hemorrhage and spontaneous cerebral hemorrhage-related lesions. Furthermore, this design has the advantage of reducing multiple linearity and proving the factors associated with ischemic stroke subtypes. Fifth, we found a strong association between the ApoE ε4 allele and dementia, supporting epidemiological association studies showing that the ApoE ε4 allele may increase the risk of dementia, particularly in patients with Alzheimer’s disease or mixed dementia (39). Last, the present cohort was characterized by an older age and optimal severity compared with individuals from population-based stroke studies (40). It is well known that demographic characteristics may affect the prevalence and mechanisms of cognitive deficits, with a higher prevalence of Alzheimer’s disease in the oldest patients. Additionally, acute multiple lesions in older patients may cause dementia, depending on the depletion or deterioration of the cognitive reserve.
Finally, multiple acute spontaneous ischemic lesions in the anterior circulation, posterior circulation, or both may be independent factors for vascular dementia. Global cognitive assessment and determination of multiple and multilevel structural involvements related to specific causes are important factors that will determine the cognitive state after stroke.
1 : Prevalence and outcomes of vascular cognitive impairment. Neurology 2000; 54:447–451Crossref, Medline, Google Scholar
2 : Prevalence, incidence, and factors associated with pre-stroke and post-stroke dementia: a systematic review and meta-analysis. Lancet Neurol 2009; 8:1006–1018Crossref, Medline, Google Scholar
3 : Prevalence of poststroke neurocognitive disorders using National Institute of Neurological Disorders and Stroke-Canadian Stroke Network, VASCOG criteria (Vascular Behavioral and Cognitive Disorders), and optimized criteria of cognitive deficit. Stroke 2018; 49:1141–1147Crossref, Medline, Google Scholar
4 : Clinical determinants of dementia related to stroke. Ann Neurol 1993; 33:568–575Crossref, Medline, Google Scholar
5 : Clinical determinants of poststroke dementia. Stroke 1998; 29:75–81Crossref, Medline, Google Scholar
6 : Cerebral infarcts and cognitive performance: importance of location and number of infarcts. Stroke 2009; 40:677–682Crossref, Medline, Google Scholar
7 : Evolution of cognitive impairment after stroke and risk factors for delayed progression. Stroke 2005; 36:2670–2675Crossref, Medline, Google Scholar
8 : Association of leukoaraiosis with stroke recurrence within 5 years after initial stroke. J Stroke Cerebrovasc Dis 2015; 24:573–582Crossref, Medline, Google Scholar
9 : Association of cerebral microbleeds with cognitive decline and dementia. JAMA Neurol 2016; 73:934–943Crossref, Medline, Google Scholar
10 : Cognitive dysfunction in patients with cerebral microbleeds on T2*-weighted gradient-echo MRI. Brain 2004; 127:2265–2275Crossref, Medline, Google Scholar
11 : Medial temporal lobe atrophy on MRI differentiates Alzheimer’s disease from dementia with Lewy bodies and vascular cognitive impairment: a prospective study with pathological verification of diagnosis. Brain 2009; 132:195–203Crossref, Medline, Google Scholar
12 : Multiple, multilevel and bihemispheric infarcts, in Stroke Syndromes, 2nd ed: Edited by Bogousslavsky J, Caplan L. Cambridge, United Kingdom, Cambridge University Press, 2001, pp 499–511Crossref, Google Scholar
13 : Acute multiple infarction involving the anterior circulation. Arch Neurol 1996; 53:50–57Crossref, Medline, Google Scholar
14 : Significance of acute multiple brain infarction on diffusion-weighted imaging. Stroke 2000; 31:688–694Crossref, Medline, Google Scholar
15 : Vascular dementia, in Behavioral and Cognitive Neurology of Stroke, 2nd ed: Edited by Godefroy O. Cambridge, United Kingdom, Cambridge University Press, 2013, pp 387–409Crossref, Google Scholar
16 : Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol 2013; 12:822–838Crossref, Medline, Google Scholar
17 : The Ege Stroke Registry: a hospital-based study in the Aegean region, Izmir, Turkey. Analysis of 2,000 stroke patients. Cerebrovasc Dis 1998; 8:278–288Crossref, Medline, Google Scholar
18 : Baseline NIH Stroke Scale score strongly predicts outcome after stroke: a report of the Trial of Org 10172 in Acute Stroke Treatment (TOAST). Neurology 1999; 53:126–131Crossref, Medline, Google Scholar
19 : Interrater reliability of the NIH Stroke Scale. Arch Neurol 1989; 46:660–662Crossref, Medline, Google Scholar
20 : Improving the assessment of outcomes in stroke: use of a structured interview to assign grades on the modified Rankin Scale. Stroke 2002; 33:2243–2246Crossref, Medline, Google Scholar
21 : Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Washington, DC, American Psychiatric Publishing, 2013, pp 621–624Crossref, Google Scholar
22 : Assessment of activities of daily living in dementia: development of the Bristol Activities of Daily Living Scale. Age Ageing 1996; 25:113–120Crossref, Medline, Google Scholar
23 : Validation of the Turkish version of the Montreal Cognitive Assessment Scale (MoCA-TR) in patients with Parkinson’s disease. Clin Neuropsychol 2014; 28:333–343Crossref, Medline, Google Scholar
24 : White matter signal abnormalities in normal individuals: correlation with carotid ultrasonography, cerebral blood flow measurements, and cerebrovascular risk factors. Stroke 1988; 19:1285–1288Crossref, Medline, Google Scholar
25 : Improving interrater agreement about brain microbleeds: development of the Brain Observer MicroBleed Scale (BOMBS). Stroke 2009; 40:94–99Crossref, Medline, Google Scholar
26 : Neuroimaging determinants of poststroke cognitive performance. Stroke 2018; 49:2666–2673Crossref, Medline, Google Scholar
27 : Cerebral microbleeds are associated with worse cognitive function: the Rotterdam Scan Study. Neurology 2012; 78:326–333Crossref, Medline, Google Scholar
28 : Deficits of memory, executive functioning and attention following infarction in the thalamus; a study of 22 cases with localised lesions. Neuropsychologia 2003; 41:1330–1344Crossref, Medline, Google Scholar
29 : Anatomy of aphasia revisited. Brain 2018; 141:848–862Crossref, Medline, Google Scholar
30 : Stroke and action slowing: mechanisms, determinants and prognosis value. Cerebrovasc Dis 2010; 29:508–514Crossref, Medline, Google Scholar
31 : Neuropsychological outcome following thalamic stroke in adolescence: an identical twin comparison. Clin Neuropsychol 2019; 33:905–927Crossref, Medline, Google Scholar
32 : Age-specific CT and MRI templates for spatial normalization. Neuroimage 2012; 61:957–965Crossref, Medline, Google Scholar
33 : Evaluation of brain atrophy estimation algorithms using simulated ground-truth data. Med Image Anal 2010; 14:373–389Crossref, Medline, Google Scholar
34 : Cerebral small vessel disease, medial temporal lobe atrophy and cognitive status in patients with ischaemic stroke and transient ischaemic attack. Eur J Neurol 2017; 24:276–282Crossref, Medline, Google Scholar
35 : Progression of cognitive impairment in stroke/TIA patients over 3 years. J Neurol Neurosurg Psychiatry 2014; 85:1324–1330Crossref, Medline, Google Scholar
36 : Secondary neurodegeneration in remote regions after focal cerebral infarction: a new target for stroke management? Stroke 2012; 43:1700–1705Crossref, Medline, Google Scholar
37 : Microinfarct pathology, dementia, and cognitive systems. Stroke 2011; 42:722–727Crossref, Medline, Google Scholar
38 : Cerebral cortical microinfarcts on magnetic resonance imaging and their association with cognition in cerebral amyloid angiopathy. Stroke 2018; 49: 2330–2336Crossref, Medline, Google Scholar
39 : Epidemiological studies of the effect of stroke on incident dementia: a systematic review. Stroke 2010; 41:e41–e46Crossref, Medline, Google Scholar
40 : Stroke in the very old: incidence, risk factors, clinical features, outcomes and access to resources: a 22-year population-based study. Cerebrovasc Dis 2010; 29:111–121Crossref, Medline, Google Scholar



