Apathy and Huntington’s Disease: A Literature Review Based on PRISMA
Abstract
Objective:
Although apathy is commonly reported among patients with Huntington’s disease (HD), this psychiatric symptom has not yet been clearly defined or extensively studied in HD. Most researchers have adopted descriptive approaches, showing that apathy is a multidimensional entity but leaving the processes underlying its different dimensions relatively unexplored.
Methods:
A systematic review of the literature on apathy in HD, focusing on current approaches and measurement tools, was conducted.
Results:
Searches in PubMed and PubMed Central yielded 368 articles, 25 of which were included in the present review.
Conclusions:
This systematic review suggests that more comprehensive research is needed to help shed light on apathy in HD, especially regarding its multidimensional aspect and underlying mechanisms.
Huntington’s disease (HD) is an autosomal-dominant, genetic neurodegenerative disease caused by an unstable expansion of the CAG triplet in the gene coding for Huntingtin. Onset can be at any age but generally occurs between the ages of 30 and 50.
In neuropathological terms, HD is essentially characterized by loss of medium spiny neurons in the caudate and putamen (1). Although atrophy starts in the striatum (2), imaging data show that cortical regions are also affected at an early stage (3, 4).
HD is characterized by motor, cognitive, psychiatric, and behavioral symptoms. The order of appearance of these symptoms varies between patients, but they inevitably lead to dementia. Nonmotor symptoms include executive dysfunction, memory impairments, attentional difficulties, disinhibition, depression, irritability, anxiety, and apathy (5).
Apathy seems to be the most common psychiatric symptom of HD. However, it is also present in many other pathological profiles. For example, it is highly prevalent in neurodegenerative disorders, such as Alzheimer’s disease (19%−88% of patients [6]) and Parkinson’s disease (7%−70% of patients [7]). In addition, apathy is associated with other neurological pathologies, such as stroke (8) and vascular dementia (9), as well as traumatic brain injury (10, 11). It can also be observed in psychiatric illnesses, most notably schizophrenia with negative symptoms (12). Some investigators have suggested that apathy is a behavior that can be related to normal aging (13).
Several studies (14–16) have shown that apathy is associated with poor quality of life among both patients and caregivers. However, these studies explored apathy by indicating only its presence/absence without a real description of the mechanisms underlying its appearance and its evolution as the disease progresses. While some definitions of apathy acknowledge that the symptom has cognitive, affective, and behavioral dimensions, the precise nature of the psychological processes underlying each dimension remains unknown. In our opinion, this lack of knowledge about the multidimensional aspect of apathy has negative consequences for clinical practice. In the present systematic review, we focus on different theoretical aspects of apathy. After highlighting current definitions, the tools used to assess apathy in several pathologies, and the relationship between apathy and other behavioral disorders, we examine the prevalence and characteristics of apathy in HD. Finally, we provide a critical review of the existing approaches to apathy. This critical review opens up new perspectives for improving our understanding of the multidimensional aspect of apathy and its considerable impact on the quality of life of patients with HD.
Current Definitions of Apathy
Apathy was first conceptualized by Marin in 1990 (17). According to Marin, apathy is characterized by a reduction in goal-directed cognition, associated with a lack of motivation in the following three dimensions: emotional (flat affect and emotional indifference), cognitive (decreased interest and lack of plans and goals), and behavioral (lack of effort, initiative, and productivity). This definition makes clear that apathy is a multifactorial concept with a variety of underlying mechanisms (10). Marin distinguished between apathy as a syndrome and apathy as a symptom of another syndrome. By doing so, he established a set of criteria for diagnosing apathy syndrome.
In 1994, Cummings et al. (18) suggested that apathy should be conceived as a loss of interest and motivation, with loss of initiative and flat affect. However, there is still no consensus on the conception of apathy. For example, Stuss et al. (19) stated that apathy cannot simply be attributed to a lack of motivation, because this is complicated to assess and requires observation and quantification of behaviors and affects. The authors therefore defined apathy as “an absence of responsiveness to stimuli internal or external as demonstrated by a lack of self-initiated action” (19). Here, apathy revolves around one key notion: initiation. For these authors, apathy is a set of related but separate states, and the clinical manifestations depend on the neural substrate and functional processes involved (cognitive, behavioral, and affective).
Based on Marin’s definition, Starkstein et al. (20) developed a set of diagnostic criteria for apathy in Alzheimer’s disease in order to distinguish clearly between depression and apathy, with a special emphasis on lack of motivation. The criteria were as follows: lack of motivation relative to the patient’s previous level of functioning or the standards of his or her age and culture as indicated by subjective account or observation by others; presence for at least 4 weeks during most of the day of at least one symptom belonging to each of the following three domains: diminished goal-directed behavior, diminished goal-directed cognition, and diminished concomitants of goal-directed behavior; the symptoms cause clinically significant distress or impairment in social, occupational, or other important areas of functioning; and the symptoms are not due to diminished level of consciousness or the direct physiological effects of a substance (20).
Levy and Dubois (21) put forward an alternative conception of apathy based on a cognitive model of behavior. Given that clinical manifestations of apathy are specific to lesions in the prefrontal cortex and basal ganglia, they deemed that apathy is a “behavioral observable syndrome” that manifests itself in a “quantitative reduction of self-generated, voluntary and purposeful behaviors.” They suggested that there are three underlying processes of apathy: emotional/affective (inability to associate affective/emotional signals with ongoing and forthcoming behaviors), caused by lesions in the orbital and medial prefrontal cortex or basal ganglia; cognitive (inability to elaborate a plan of action needed for ongoing and forthcoming behaviors), caused by lesions in the dorsolateral prefrontal cortex and caudate nucleus; and auto-activation (inability to activate thoughts or initiate actions). These authors suggested that there are many forms of apathy syndrome, the most severe corresponding to disturbance of the auto-activation process, which frequently results from lesions in the limbic and associative areas of the internal globus pallidus.
In line with Marin’s conception, Starkstein and Leentjens (22) focused on lack of motivation, linked to a reduction in goal-directed behaviors, and they included a temporal element in their diagnostic criteria. In order to establish a consensus in research and clinical practice, a new set of diagnostic criteria of apathy was developed (23, 24) (Box 1). These criteria had the same general structure as those of Starkstein et al. (25) based on Marin’s conception (26), and not only allowed apathy to be clearly distinguished from other concepts, but also considered all its dimensions (24).
| Robert et al. (27) | Mulin et al. (23); Robert et al. (24) |
|---|---|
| Criterion A: Quantitative reduction in goal-directed activity in behavioral, cognitive, emotional, or social areas, compared with the patient’s previous levels of functioning. These changes may be reported by the patients themselves or observed by others. | Criterion A: Reduction in motivation compared with the patient’s previous level of functioning |
| Criterion B | Criterion B: Two of the three following domains of apathy, which must have been present for at least 4 weeks. |
| Subcriterion B1: Behavior and cognition | |
| Loss of or diminished, goal-directed behavior or cognitive activity, as evidenced by at least one of the following: | |
| General level of activity | Subcriterion B1: Loss of or diminished goal-directed behavior |
| Persistence of activity | Subcriterion B2: Loss of or diminished goal-directed cognitive activity |
| Making choices | Subcriterion B3: Loss of or diminished emotions |
| Interest in external issues | |
| Personal well-being | |
| Subcriterion B2: Emotion: loss of, or diminished, emotion, as evidenced by at least one the following: | |
| Spontaneous emotions | |
| Emotional reactions to environment | |
| Impact on others | |
| Empathy | |
| Verbal or physical expressions | |
| Subcriterion B3: Social Interactions: Loss of, or diminished engagement in social interaction, as evidenced by at least one of the following: | |
| Spontaneous social initiative | |
| Environmentally stimulated social interaction | |
| Relationship with family | |
| Verbal interaction | |
| Homebound | |
| Criterion C: These symptoms (A and B) cause clinically significant impairment in personal, social, occupational, or other important areas of functioning. | Criterion C: Symptoms should cause clinically significant impairment in various functional domains. |
| Criterion D: The symptoms (A and B) are not exclusively explained or due to physical disabilities (e.g., blindness and loss of hearing), motor disabilities, a diminished level of consciousness, the direct physiological effects of a substance (e.g., drug of abuse, medication), or major changes in the patient’s environment. | Criterion D: Exclusion criteria specify symptoms that mimic apathy, such as the direct physiological effects of a substance |
BOX 1. Summary of main changes in diagnostic criteria for apathy
More recently, a group of 23 experts (researchers, healthcare professionals, etc.) met to revise the diagnostic criteria for apathy in brain disorder. Using the Standard Delphi Methodology, they revised and developed the criteria in a two-step process: web surveys, followed by a consensus meeting (27) (Box 1). The modifications mainly concerned criteria A and B (subcriteria B1 and B3). More specifically, in criterion A, the term motivation was replaced with the term goal-directed behavior. According to Robert et al. (27), goal-directed behavior is a quantifiable term, unlike motivation. The second major modification concerned criterion B. Experts wanted to keep the same three-dimensional structure (behavior, cognition, and emotion). However, it was important to add a social aspect. Behavior and cognition have different underlying brain processes, but in clinical practice, apathy manifests itself in both cases as diminished observable activity (27). Thus, the expert group placed behavior and cognition in the same subcriterion (B1) and added a separate dimension: social interaction (subcriterion B3). According to the authors, this modification improved the assessment of apathy in both clinical research and everyday clinical practice. Another key change concerned criterion D, with the addition of the sentence “the symptoms (criteria A and B) are not exclusively explained or due to major changes in the patient’s environment.” This took account of the fact that some tragic events (e.g., terrorist attack) can lead to changes that may mimic apathy (27). By including a separate social interaction dimension covering spontaneous or stimulated social interaction, this new diagnostic criterion for apathy reflects an awareness of apathy’s multidimensional aspect. It provides as objective an assessment as possible, as it is based on aspects whose impairment can easily be described and observed.
The concept of apathy is also found in DSM, but it is only mentioned as a symptom of other pathologies (e.g., depression). The International Classification of Diseases makes no reference to apathy.
Defining specific criteria is very important. It avoids potential confusion between apathy and other clinical terms like abulia, anhedonia, depression, and learned helplessness. For example, the Oxford English Dictionary defines apathy as a lack of interest (cognitive aspect) or a lack of enthusiasm (affective aspect). However, abulia is defined as an absence of willpower or an inability to act decisively. This confusion has led to diagnostic errors. For example, in psychiatry, abulia is regarded as the severe form of apathy (24).
Apathy Assessment Scales
Many different instruments have been used to examine the presence and severity of apathy in psychiatric, neurological and neurodegenerative pathologies. Box 2 cites the apathy measures most commonly used in HD (5).
| Apathy assessment tool | Characteristic |
|---|---|
| Unified Huntington’s Disease Rating Scale (UHDRS) | This rating scale covers four dimensions of HD: motor, cognitive, functional, and behavioral. The latter comprises 10 items, only one of which refers to apathy (“inability to enjoy anything”). This apathy item is included in the general mood category. |
| Problem Behaviors Assessment for Huntington’s Disease (PBA-HD) | This semistructured interview features 40 items that cover a range of psychological symptoms, including apathy. It is widely used in studies of HD. It measures the frequency and severity of apathy over the previous 4 weeks (scores between 0 and 5). There is a short version of the PBA. |
| Neuropsychiatric Inventory | This is based on an interview with a caregiver. The apathy subscale consists of a yes/no screening question followed by seven subquestions, depending on the answer to the first question. It covers loss of interest, lack of motivation, decreased spontaneity and enthusiasm, loss of emotion, flat affect, and disinterest in carrying out new activities. |
| Structured Clinical Interview for Apathy | This interview was designed to detect apathy in Alzheimer’s disease. It covers “lack of motivation relative to the individual’s previous level of functioning, lack of effort to perform everyday activities, lack of interest in learning new things or in new experiences, lack of self-concern, flat affect and lack of emotional response.” It was used, together with AES and UHDRS, as a measure in the first treatment trial for apathy in HD. |
| Apathy Evaluation Scale (AES) | The AES was the very first apathy scale (18 items). It is now available in three versions: caregiver (AES-I), clinician (AES-C), and self-report (AES-S). It covers the cognitive, emotional, and behavioral aspects of apathy. Items are rated on a 4-point Likert scale. A high score indicates severe apathy. |
| 14-Item Apathy Scale | This simplified version of the AES covers the three dimensions of apathy identified by Marin (26). The psychometric properties of this tool have not been fully investigated. |
| Frontal Systems Behavior Scale | This scale is designed to evaluate disorders after frontal lesions: apathy (14 items), disinhibition (15 items), and executive dysfunction (17 items). There are self, informant, and clinician versions. The apathy subscale covers loss of interest and self-care, and difficulties in initiation and spontaneity. |
BOX 2. Summary of characteristics of the most commonly used apathy tools in Huntington’s disease (HD)
As can be seen, there is still only a limited choice of tests. In the majority of studies, apathy is evaluated with nonspecific tools. For example, the short version of the Problem Behaviors Assessment (PBA-s), the Frontal Systems Behavior Scale, and the Neuropsychiatric Inventory (NPI) are general tests for behavioral disorders, with only one or two items devoted to apathy. Other measures can assess apathy, but they are one-dimensional, simply providing an overall score to confirm or refute the presence of apathy (e.g., Apathy Evaluation Scale [AES] and 14-item Apathy Scale) (28). Another tool that is supposed to assess the different substructures of apathy is the Apathy Inventory, which was developed to collect comprehensive information about apathy’s presence in several brain pathologies (29). It takes several components of apathy into account: flat affect, loss of initiative, and loss of interest. This scale comes in three versions: clinician, caregiver, and patient. Its aim is to detect behavioral changes since disease onset. The clinician version takes the form of a semistructured interview (or even observation, in the case of prolonged hospitalization). The Apathy Inventory has consistent psychometric properties (30).
The Lille Apathy Rating Scale (LARS) also assesses different aspects of apathy. It was developed by Sockeel et al. (31), based on Marin’s conception of apathy. This scale includes 33 items covering nine domains: reduction in everyday productivity, lack of interest, lack of initiative, extinction of novelty seeking, motivation, blunting of emotional responses, lack of concern, poor social life, and self-awareness. It is administered in the form of a structured interview. It has a four-factor structure: intellectual curiosity, emotion, action initiation, and self-awareness. However, there has been no confirmatory analysis to validate these four factors (10). Internal consistency (α=0.80) and test–retest reliability (r=0.95) are satisfactory (30). A shortened version was developed and validated by Dujardin et al. (32). This version includes seven of the original nine domains (reduction in everyday productivity, lack of interest, lack of initiative, extinction of novelty seeking, motivation, blunting of emotional responses, and poor social life). The other two (lack of concern and self-awareness) were deleted because of their lack of sensitivity (33). This shortened version has acceptable internal consistency (α=0.73), good interrater reliability (r=0.95), and good test–retest reliability at 4 months (r=0.92) (11).
Despite its psychometric limitation, the LARS is one of the rare assessment tools to consider the multifactorial and multidimensional aspect of apathy. However, according to Radakovic et al. (34), the Apathy Inventory and LARS are good assessment tools for unidimensional apathy but not for multidimensional apathy. In order to remedy the lack of multidimensional scales, Radakovic and Abrahams (35) created a tool that is well adapted to the multidimensional aspect of apathy. The Dimensional Apathy Scale (DAS) was designed for persons with neurodegenerative diseases who have motor difficulties. To avoid the impact of motor deficits, none of the items rely on physical performances to assess the motor aspect of apathy. The structure of this scale was based on Levy and Dubois’s (21) conception, and it contains items taken from other scales (11). The authors conducted an exploratory factorial analysis based on 311 responses from healthy individuals. There were initially 45 items, but after deleting some items with a low saturation index, they came up with a version featuring 24 items evaluating the executive, emotional, and cognitive/behavioral dimensions of apathy. These items have good internal consistency (α=0.80). No study has yet been conducted with the scale’s target population (11). Although the exclusion of the motor aspect avoids confusion between pure motor difficulties and apathetic behaviors, the total exclusion of the motor dimension deprives users of information about the possible contribution of physical illness to the emergence of apathy (11). In several studies, researchers have argued that motor difficulties can contribute to the development of apathy and that there is a significant link between the two (36, 37). These authors suggested that physical disorders lead to a lack of interest in activities requiring physical performances. In this context, it is important to use scales that take account of the different factors that may contribute to the emergence of apathy (38).
However, the DAS remains one of the few multidimensional scales of apathy. For this reason, it was adapted and translated into French by M’Barek et al. (28). The French version (f-DAS) was validated with 223 healthy participants. Analyses indicated psychometric properties similar to those of the original version (35). The f-DAS has good validity and reliability, good internal consistency, and good discriminant validity against depression. A confirmatory factor analysis corroborated the three-factor model (28) of Radakovic and Abrahams (35).
In essence, the absence of a consensus on the nature of aspects of apathy and the different conceptions of apathy are reflected in several inconsistencies in the psychometric properties of all these assessment tools. To remedy these, we must have a single theoretical and conceptual basis on which to develop reliable and valid tools for measuring apathy (11).
Apathy and Other Neuropsychiatric Disorders
Psychiatric and behavioral disorders are not yet well documented in HD, even though researchers have known about them for a long time. These disorders are known to be responsible, at least in part, for the occupational and social disintegration that accompanies HD (39). They are also known to have a more harmful impact than the cognitive and motor symptoms on functional skills (40) and quality of life (41). In their literature review, van Duijn et al. (42) revealed a high prevalence of depression, anxiety, irritability, and apathy in HD. The frequency of these manifestations varied across studies from 33% to 76%, probably owing to disease progression. Obsessive-compulsive disorder (10%−52%) and psychotic disorder (3%−11%) were less frequent. Other psychobehavioral disorders are also reported in HD, including sexual behavior disorders (25%), which can give rise to everyday conflict (39). Eating disorders have also been reported, such as bulimia, snacking, or refusing food on account of delusions such as fear of poison (39). Furthermore, Snowden et al. (43) identified social behavior disorders such as egocentricity and lack of sympathy or empathy combined with mental toughness in patients with HD. Respect for shared social conventions can be disturbed in this population. For example, people with HD may make crude or obscene comments or engage in offensive behavior (44).
Apathy has always been addressed as a symptom of other pathologies (e.g., depression). Some authors consider apathy to be a symptom of depression. For example, Paoli et al. (45) suggested that depression is the most frequent symptom in HD, and that apathy is one of its clinical manifestations or somatic expressions. Authors sometimes include apathy in the same category as inactivity and weight loss. The boundaries between apathy and depression remained blurred for a long time, because they share common symptoms such as loss of interest and hypersomnia (35, 46). Over time, researchers realized that they are actually two completely different psychological entities: Apathy is a lack of motivation, whereas depression is a mood disorder. Marin (17, 26) noticed that the relation between apathy and depression varies according to the disease profile but also according to the individual’s profile. It should be noted that these two entities can frequently coexist. This confusion can lead to bias when estimating their frequency in different clinical pictures (47).
Apathy and Huntington’s Disease
As discussed above, apathy is one of the most common symptoms of HD. It has been investigated in several studies using different scales (e.g., NPI, PBA for HD [PBA-HD], and AES), as shown in Table 1.
| Study | Sample size | Disease stage | Neuropsychiatric measures | Major findings related to apathy |
|---|---|---|---|---|
| Craufurd et al. (48) | 134 | Confirmed HD | PBA-HD for apathy, anxiety, depression, and irritability, etc. | Presence of three factors—apathy, irritability, and depression—were found. Apathy was the only factor related to disease duration. |
| Paulsen et al. (49) | 52 | Confirmed HD | NPI for apathy, anxiety, agitation, dysphoria, irritability, and anxiety, etc. | Prevalence of apathy was 55.8%. Apathy prevalence increased with disease duration in HD. Apathy was linked to disruption of the medial prefrontal circuit. |
| Thompson et al. (50) | 82 | Confirmed HD | PBA-HD for apathy, depression, and irritability | Apathy was correlated with cognition and motor functions. The apathy subscale was a valid measure of apathy severity in HD. The correlation between apathy score and cognitive impairment in HD was explained by disruption of striatofrontal circuits. |
| Baudic et al. (51) | 36 | Early HD | Semistructured interview with patient and close relative (presence and severity of apathy); MADRS for depression | Twenty out of 36 patients were apathetic. Apathy was associated with cognitive impairment in early HD: attention, executive functions, and episodic memory. In apathetic patients, cognitive impairments were linked to motor disturbance. |
| van Duijn et al. (52) | 152 | HD mutation carriers | AS for apathy | Apathy had a prevalence of 32% in mutation carriers (0% in noncarriers). |
| 56 | Noncarriers | CIDI for depression | Apathy was associated with depression, poorer overall functioning, motor and cognitive symptoms, and increased use of psychotropic medication. | |
| Naarding et al. (53) | 34 | Confirmed HD | AES for apathy | Prevalence of apathy was 52%. Apathy was far more present in HD than depression, and these two entities were largely distinct dimensions. Apathy was related to cognitive impairments and functional decline. |
| Duff et al. (54) | 745 | Expansion positive (premanifest) | FrSBe for apathy, disinhibition, and executive functions | Pre-HD expansion positive patients had significantly higher total FrSBe scores than participants with negative expansion. |
| 163 | Expansion negative | |||
| Reedeker et al. (55) | 122 | Premotor and motor HD | AS for apathy and CIDI for depression | At baseline, 34 out of 122 patients exhibited apathy. Apathy was correlated with lower clinical and neuropsychiatric scores. Thirteen (14%) of 88 patients without apathy at baseline developed apathy by the 2-year follow-up. Apathy was reversible: 14 (41%) of 34 patients who had apathy at baseline no longer manifested apathy 2 years later, while 20 (59%) patients remained apathetic. Cognitive dysfunctions contributed to apathy onset in HD. |
| Thompson et al. (56) | 111 | Confirmed HD; stages I–II; stages II–III; and stages III–IV | PBA-HD for apathy, depression, and irritability | Prevalence of apathy was 68% at stages I–II, 73% at stages II–III, and 85% at stages III–IV. Apathy scores were associated with HD progression; apathy was intrinsic to disease progression. Apathy, depression, and irritability were each associated with a distinct longitudinal profile, suggesting that each had a different neurobiological substrate. |
| Tabrizi et al. (57) | N=366 at baseline; N=298 at 36-month follow-up | Pre- and early HD | PBA for apathy | Apathy scores increased with disease progression. |
| van Duijn et al. (58) | 1,993 | Premanifest and manifest HD | UHDRS-b for depression, apathy, psychosis, irritability/aggression, and obsessive-compulsive behaviors | Prevalence of apathy was 47.4% (19.3% mild apathy and 28.1% moderate to severe apathy). Apathy was the main symptom, occurring most often in advanced HD. Apathy was perhaps the only nonmotor feature that was linearly associated with the neurodegenerative progression. |
| Mason and Barker (59) | N=108 at baseline; N=73 at 18.7 months follow-up | Premanifest and advanced HD | AES-P (participant version) for apathy AES-I (companion version for caregivers) for apathy | Prevalence of apathy was 26.9% (N=29/108) of patients were apathetic according to self-rated AES scores, and 37.9% (N=30/79) of patients were apathetic according to AES (companion version) scores. Statistically, apathy could be equally assessed by either patients or their proxies. Apathy did not progress during an 18-month follow-up, and it was distinct from depression. |
| Martinèz-Horta et al. (60) | 230 | Premanifest far from motor-based HD onset (pre-HD, criterion A); premanifest patients close to motor-based HD onset (pre-HD, criterion B); and early manifest HD | PBA-s for apathy, depression, irritability, psychosis, and dysexecutive behavior | Prevalence of apathy was 23% in pre-HD criterion A, 64% in pre-HD criterion B, and 63% in early HD. Apathy could occur up to 10 years before disease onset. Apathy was closely linked to the neurodegenerative process of HD. |
| Kempnich et al. (61) | 32 | Premanifest and manifest HD | TASIT for emotion recognition and FrSBe for apathy and disinhibition | Emotion recognition skills were associated with apathy measures. This association was independent of disease severity (motor dysfunction). Emotion recognition and apathy had an overlapping neural circuit (anterior cingulate circuit). |
| Sousa et al. (7) | 29 | Confirmed HD | AES-C for apathy and BDI-II for depression | Apathetic patients displayed more cognitive disorders than nonapathetic patients. Apathy was strongly related to motor symptoms, disease duration, and the presence of cognitive disorders. |
| Fritz et al. (14) | 487 | N=193 pre-HD; N=186 early HD; N=108 late-stage HD | PBA-s HD for 11 items; one of which was apathy | Apathy could appear up to 3 years before disease onset and increased as the disease progressed. Apathy was correlated with disease duration and motor and cognitive disorders, strongly linked to invalidity, and had a negative impact on quality of life. |
| Jacobs et al. (15) | 220 | Pre- and manifest HD; N=114 employed; N=106 unemployed | PBA-s HD for apathy, depression, and irritability | Apathy was the main predictive factor (in association with executive functions) for unemployment and had a negative impact on quality of life. |
| Baake et al. (62) | 31 | Premotor manifest | AES for apathy | Apathy severity increased with disease progression. Premotor manifest patients experienced a higher level of apathy that went unnoticed by their caregivers. |
| 49 | HD, | |||
| Early motor manifest | ||||
| 29 | HD | |||
| Late motor manifest | ||||
| HD | ||||
| Baake et al. (63) | 91 | Premotor manifest | PBA-s for 11 items; one of which was apathy | Prevalence at baseline was 12% for pre-HD, 24% for stage 1, and 50% for stage 2 of motor manifestations. The percentage of apathetic participants increased over 2 years. Apathy was present in the early stages of HD and was associated with thalamic atrophy in HD gene carriers. Apathy had an underlying neural mechanism. |
| 80 | Motor manifest | |||
| Aldaz et al. (64) | 53 | Mild, moderate, and severe stages of HD | PD NMS Quest covering nine domains (apathy, depression, and digestion, etc.) | Prevalence of apathy was 64%. Up to 60% of patients in slight-mild stages complained of apathy, and therefore apathy can appear early in the disease course. |
| Misiura et al. (65) | 797 | Prodromal HD | FrSBe for apathy and depression | Greater severity of apathy was associated with striatal atrophy, and increased apathy was related to reduced cognitive functions. |
| McLauchlan et al. (66) | 51 | Pre-HD to moderate HD | AES for apathy and PBA-s for the 11 symptomsb and BIS-BAS for reward value, reward effort, and response to reward and loss | Apathy was related to a deficit in response to loss. Altered response to reward or altered reward effort valuation was not necessary to develop apathetic behavior. |
| De Paepe et al. (67) | 46 | N=22 pre-HD; N=24 manifest HD | LARS-s for apathy and PBA-s for apathy and affective components (depressed mood, suicidal ideations, and anxiety) | Manifest patients displayed higher levels of overall apathy and auto-activation deficit. Pre-HD patients only had higher levels of auto-activation deficit. Increased apathy was correlated with HD progression. |
| Gunn et al. (68) | 7,914 | N=974 FM; N=1,041 GN; N=1,790 pre-HD; and N=4,109 manifest HD | PBA-s for apathy, affect, and irritability | Patients with HD underestimated their apathetic symptoms. Informants made major contributions to apathy assessments. |
| Andrews et al. (69) | 229 | N=118 pre-HD; and N=111 early HD | PBA-s HD for apathy BDI-II for depression | In premanifest HD, severe apathy symptoms predicted a more rapid cognitive decline. By contrast, in early manifest HD, motor signs were better predictors of cognitive decline than apathy. |
TABLE 1. Studies of Apathy in Huntington’s Disease (HD)a
Methods
We undertook a systematic review, based on Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. This approach was chosen because of the lack of research and the heterogeneous nature of existing studies, as well as the lack of a comprehensive review of the literature on apathy and HD. This systematic review was conducted with two major databases; PubMed and PubMed Central. We manually searched reviews and checked reference lists to identify further relevant citations. We used the following key terms to search the databases: PubMed Central: (huntington[Abstract] AND apathy[Abstract]) and (huntington[Body - Key Terms]) AND apathy[Body - Key Terms]); PubMed: (huntington[Title/Abstract]) AND (apathy[Title/Abstract]) and (huntington[Text Word]) AND (apathy[Text Word]).
Inclusion and Exclusion Criteria
To be included in the present review, studies had to fulfill the following criteria: describing research among individuals with confirmed HD; involving patients aged 18 years and above; measuring apathy and other neuropsychiatric symptoms; drafted in the form of an article; and written in the English language. We excluded studies in the form of poster and paper presentations, editorials, and letters.
Data Extraction
In our systematic review, we extracted data on sample size, disease stage, apathy measure, other measures of neuropsychiatric problems, and major findings related to apathy.
Quality Appraisal
Owing to the nature of systematic reviews, we did not perform a quality appraisal. We did, however, highlight and discuss the methodological and clinical limitations of the studies we included wherever possible.
Results
The database searches yielded a total of 368 articles, which were reduced to 230 articles after duplicate suppression; removal of posters, letters, and other forms of study apart from original articles; and filtering for English language. Following the screening of titles and abstracts, 65 articles were eligible for review. After screening of the full-text articles, 25 were included in this systematic review (Figure 1).
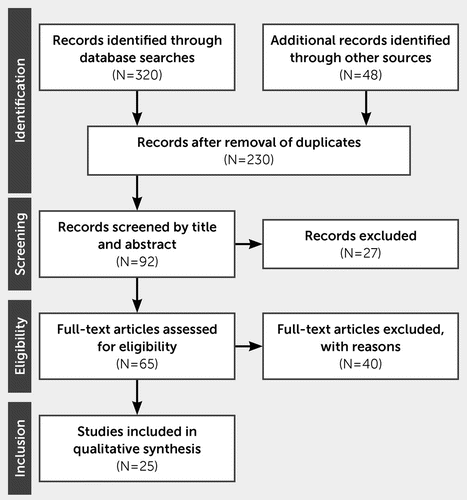
FIGURE 1. Flow diagram of the study selection in a systematic review of the literature on apathy in Huntington’s disease
Our review indicates that apathy is present at every stage of the disease. Studies of apathy in HD that were included are summarized in Table 1 (48–69). It is also the only symptom that gradually progresses, making it an excellent marker of disease progression (48, 57, 70). In their clinical review, McColgan and Tabrizi (71) confirmed that apathy is the most prevalent symptom in HD. According to these authors, this may be due to the absence of treatment for apathy, as other neuropsychiatric symptoms, such as depression and irritability, can be treated medically (antidepressants and antipsychotics).
First of all, as we can see in Table 1, the prevalence of apathy varied from study to study. This may have been due to several factors, such as place of recruitment, inclusion and exclusion criteria, disease severity (stage), and type of assessment tool. Nevertheless, all the authors agreed that apathy is a frequent neuropsychiatric manifestation in HD.
Second, it should be noted that patients rarely complained of apathy, although this does not imply that apathy is not disabling and does not have a negative effect on patients’ quality of life (16). Apathy is considered to be one of the most impactful disorders in terms of the quality of life of patients with HD and their caregivers (67). Patients have fewer social interactions and are less engaged in their activities and hobbies, while caregivers have a sense of isolation and the feeling of missing out on their lives (5). Apathy therefore has a substantial direct negative effect on the quality of life of both patients and caregivers. It also has a negative effect on family relations, as shown in the review by Good et al. (70), although it should not be forgotten that every patient is unique, in terms of the association of symptoms and the implications of the disease at family, social, and occupational levels. By the same token, the assessment of individual patients must take into account the social, cultural, and environmental dimensions of their psychological disorders. Patients each have their own life story, which obviously has an impact on the manifestation and maintenance of their disorders. By considering these different aspects, researchers and clinicians can avoid the neurobiologization of psychological functioning.
The findings of this systematic review show that research on apathy in HD is limited in terms of methods, approaches, and clinical findings. These are discussed below, along with suggestions for a new approach allowing apathy in HD and its underlying mechanisms to be better understood.
Limitations of the Current Concept of Apathy
As indicated earlier, despite its frequent occurrence in HD and its significant impact on quality of life, apathy is rarely studied and interpreted by taking its multidimensional nature and various underlying mechanisms into account.
Most investigators agree that apathy is a multidimensional entity. However, current conceptions tend to be applied in practice and research without any heed to the variety of apathy’s mechanisms. These conceptions are based on a descriptive and categorical approach, such as the one described in DSM, where patients are categorized according to the presence of symptoms and their severity and frequency. Several studies have adopted this kind of approach, particularly when it comes to tool choice and the interpretation of results. For example, in recent studies, the most common test was the PBA-HD or AES. However, the PBA was not specifically designed to evaluate apathy; it actually allows users to assess 11 different behavioral and psychiatric symptoms (depression, suicidal ideation, anxiety, anger/aggression, irritability, apathy, obsessive-compulsive behavior, perseverative thinking, delusions, hallucinations, and disoriented behavior). The AES only assesses severity and frequency.
Another limitation related to the categorical approach is the ambiguous relation between apathy and other psychological disorders like anhedonia, abulia, and, in particular, depression. Some authors consider apathy to be a symptom of depression. For Paoli et al. (45), depression is the most common symptom in HD, and apathy is one of the somatic expressions of depressive mood, belonging to the same category as inaction and weight loss. This confusion between apathy and depression is observed in neurological pathologies (10) and neurodegenerative diseases like Alzheimer’s disease (72). It should be noted that apathy can occur independently of depression in several pathologies, but also in the absence of depression. However, comorbidity is frequently observed (11). This dilemma surrounding diagnosis stems mainly from the existence of shared characteristics. According to diagnostic criteria, characteristics of apathy include lack or loss of interest; while according to DSM criteria, depression involves a loss of interest or pleasure. There is clearly an overlap between apathy and depression. It should be noted that these two psychological entities appear independently in 10% of patients with mood and personality disorders after closed head injury, and in combination in 60% of these patients (73). In their confirmatory factor analysis among patients with Parkinson’s disease, Kirsch-Darrow et al. (74) confirmed that apathy and depression share common signs. Their study revealed four factors: apathy, dysphoric mood, loss of interest or pleasure (reflecting the characteristic common to apathy and depression), and somatic. This goes against the idea that apathy is a sign of depression and highlights that apathy is a distinct entity.
In HD, the relation between apathy and depression is unclear. Naarding et al. (53) conducted a study among patients with HD to explore the relationship between these two behavioral dimensions. The main finding was that apathy and depression are two distinct entities and there is no clear correlation between them. The same results were reported by other authors such as Levy et al. (75). Contradictory results concerning the relation between apathy and depression probably stem from ambiguous definitions of their criteria and from the use of different approaches and diagnostic tools to assess apathy. Regarding this last point, most studies use tools based on categorical and descriptive approaches. There is no consideration of environmental, social, individual, and biological factors. Overall, these heterogeneous data findings raise a serious question about the validity of some study results.
According to Haslam et al. (76), depression is not a separate category but rather a dimensional entity. This raises several issues, the main one being the comorbidity of depression with other types of psychological disorders (77) such as apathy, and more notably loss of interest. Several authors have suggested conceiving of psychopathological disorders as patterns of dynamic interactions between symptoms (78–80). Put another way, we can conceive of psychopathological difficulties as a network of causally connected symptoms, such that if symptoms A and B are linked, then if symptom A is activated, symptom B will have a higher probability of being activated. These networks of symptoms are transdiagnostic, meaning that symptoms may be shared by other psychopathological problems. For example, Borsboom et al. (81) deemed that insomnia is common to both major depression and anxiety disorder and is induced by two different factors: feelings of guilt (for depression) or chronic anxiety (for generalized anxiety). Once insomnia is activated, it may in turn affect depressive mood (symptom of depression) or irritability (symptom of generalized anxiety).
Taken together, these circumstances suggest that apathy is not a latent entity that causes several symptoms, but rather a dimension where symptoms dynamically interact, and overlap too with other symptoms of other dimensional entities, notably depression. This highlights the importance of not focusing on a purely descriptive categorical approach, and not being limited to the assessment of pre-established symptoms. Rather, it involves assessing a wide array of symptoms and taking account of the heterogeneity of the mechanisms involved at different levels (biological, psychological, environmental, and sociocultural) in order to better understand comorbidities. To overcome categorical diagnostics such as DSM, the individuality and globality of the person should be considered, by attempting to gain more detailed and rigorous descriptions of his or her experiences, problems, difficulties, symptoms or specific complaints (82, 83).
Another typical limitation of the categorical approach is that it does not consider the existence of different and heterogeneous apathy profiles in HD. Apathy is not simply the sum of various specific and isolated symptoms whose clinical manifestations are limited to a specified brain area. Rather, it has several dimensions that combine to create different profiles, and its manifestations depend on interactions between symptoms and the nature of the mechanisms or factors involved. In their study, for instance, Strauss et al. (84) examined a sample of outpatients with schizophrenia to test the possibility that subgroups could be identified according to their negative symptoms. The authors ran cluster analyses of scores on the Scale for the Assessment of Negative Symptoms. Results revealed two different apathy profiles: one dominated by asociality, avolition, and anhedonia (apathy-avolition), and one that included alogia and blunted affect (diminished expressions). Authors also found that participants in the apathy-avolition subgroup were less likely to be in paid employment and to provide finished, high-quality work than the diminished expressions subgroup. They were also more likely to manifest symptoms of formal thought disorder and more likely to have poorer premorbid social adaptation in childhood. These data revealed the heterogeneity of apathy’s clinical manifestation in schizophrenia and the mechanisms and factors involved, as well as the various occupational and socioeconomic repercussions.
These findings point to the existence of different apathy profiles in HD, although to our knowledge, no study has yet explicitly investigated the complex and heterogeneous dynamics of apathy in HD, especially in the presymptomatic stage. It is important to identify the heterogeneous manifestations of apathy and the variety of underlying mechanisms, to ensure a better understanding of deficits and provide appropriate care and treatment.
All these circumstances suggest that it may be necessary to change the current thinking about psychopathological disorders in HD, notably apathy. It is important to make the assessment of apathy more systematic until the premanifest stage of the disease, then use a tool that assesses the different facets of apathy, instead of focusing solely on patients’ overall score and number of symptoms. Thereafter, a stringent method should be applied that details the complaints and factors involved in apathy manifestations without omitting to consider the different possible comorbidities and the overlapping of apathy symptoms with those of other disorders, particularly depression. This will allow for an accurate assessment of apathy and a precise identification of patients’ apathy profile to ensure better psychological care. This will be the next step in our research.
Conclusions
Apathy is common to many pathologies (psychiatric, neurological, and neurodegenerative), but regrettably its underlying mechanisms have been relatively unexplored. This has affected the development of apathy assessment scales and their psychometric properties. Apathy clearly has a multidimensional aspect, but its study continues to be based on a categorical and descriptive approach, even if a new multidimensional framework has been developed and applied in some research. For example, Arnould’s (11) study of apathy in patients with severe head trauma highlighted the diversity and heterogeneity of apathy profiles in these patients. Arnould also found that each profile was associated with different psychological processes. For example, the high apathy profile was significantly related to poor psychosocial functioning and a high level of subjective burden reported by caregivers, while the loss of initiative and interest profile was related exclusively to poor psychosocial functioning. This study assumed that apathy has four possible dimensions (cognitive, affective, motivational, and identity) and that each dimension is sustained by a variety of mechanisms (11).
Research needs to adopt this new model based on an integrative and multifactorial approach, thereafter generalizing it to other pathologies, which is what we are planning to do in our next study. At the theoretical level, we will aim to understand apathy, its various mechanisms, and its interrelations with other psychological difficulties in order to redefine and reconsider this symptom in HD. At the clinical level, this research will lead to a better understanding of apathetic behaviors related to HD and thus to more reliable and more sensitive assessments aimed at developing more effective psychological interventions targeting the relevant mechanisms.
1 : Reduced basal ganglia volume associated with the gene for Huntington’s disease in asymptomatic at-risk persons. Neurology 1994; 44:823–828Crossref, Medline, Google Scholar
2 : Magnetic resonance imaging as an approach towards identifying neuropathological biomarkers for Huntington’s disease. Brain Res Brain Res Rev 2008; 58:209–225Crossref, Google Scholar
3 : Defective emotion recognition in early HD is neuropsychologically and anatomically generic. Neuropsychologia 2008; 46:2152–2160Crossref, Medline, Google Scholar
4 : Topography of cerebral atrophy in early Huntington’s disease: a voxel based morphometric MRI study. J Neurol Neurosurg Psychiatry 2004; 75:213–220Medline, Google Scholar
5 : Apathy in Huntington’s disease: a review of the current conceptualization. J Alzheimers Dis Parkinsonism 2018; 8:1000431Crossref, Google Scholar
6 : Apathy in Alzheimer’s disease. Curr Opin Behav Sci 2018; 22:7–13Crossref, Medline, Google Scholar
7 : Apathy profile in Parkinson’s and Huntington’s disease: a comparative cross-sectional study. Eur Neurol 2018; 79:13–20Crossref, Medline, Google Scholar
8 : Are there common core features of apathy in different neuropsychiatric samples as assessed by the Apathy Evaluation Scale? Nord J Psychiatry 2010; 64:49–57Crossref, Medline, Google Scholar
9 : Hypertension and white matter lesions are independently associated with apathetic behavior in healthy elderly subjects: the Sefuri brain MRI study. Hypertens Res 2009; 32:586–590Crossref, Medline, Google Scholar
10 : A multidimensional approach to apathy after traumatic brain injury. Neuropsychol Rev 2013; 23:210–233Crossref, Medline, Google Scholar
11 : Apathie et traumatisme crânio-cérébral: Une approche plurifactorielle (doctoral dissertation). Geneva, Switzerland, University of Geneva, 2017Google Scholar
12 : Negative symptoms: a review of schizophrenia, melancholic depression and Parkinson’s disease. Brain Res Bull 2006; 70:312–321Crossref, Medline, Google Scholar
13 : Do people become more apathetic as they grow older? a longitudinal study in healthy individuals. Int Psychogeriatr 2010; 22:426–436Crossref, Medline, Google Scholar
14 : Relationships among apathy, health-related quality of life, and function in Huntington’s disease. J Neuropsychiatry Clin Neurosci 2018; 30:194–201Link, Google Scholar
15 : Cognitive performance and apathy predict unemployment in Huntington’s disease mutation carriers. J Neuropsychiatry Clin Neurosci 2018; 30:188–193Link, Google Scholar
16 : The neuropsychology of Huntington’s disease. Arch Clin Neuropsychol 2017; 32:876–887Crossref, Medline, Google Scholar
17 : Differential diagnosis and classification of apathy. Am J Psychiatry 1990; 147:22–30Crossref, Medline, Google Scholar
18 : The Neuropsychiatric Inventory: comprehensive assessment of psychopathology in dementia. Neurology 1994; 44:2308–2314Crossref, Medline, Google Scholar
19 :
20 : Syndromic validity of apathy in Alzheimer’s disease. Am J Psychiatry 2001; 158:872–877Crossref, Medline, Google Scholar
21 : Apathy and the functional anatomy of the prefrontal cortex-basal ganglia circuits. Cereb Cortex 2006; 16:916–928Crossref, Medline, Google Scholar
22 : The nosological position of apathy in clinical practice. J Neurol Neurosurg Psychiatry 2008; 79:1088–1092Crossref, Medline, Google Scholar
23 : Diagnostic criteria for apathy in clinical practice. Int J Geriatr Psychiatry 2011; 26:158–165Crossref, Medline, Google Scholar
24 : Proposed diagnostic criteria for apathy in Alzheimer’s disease and other neuropsychiatric disorders. Eur Psychiatry 2009; 24:98–104Crossref, Medline, Google Scholar
25 : Prevalence and clinical correlates of disinhibition in dementia. Cogn Behav Neurol 2004; 17:139–147Crossref, Medline, Google Scholar
26 : Apathy: a neuropsychiatric syndrome. J Neuropsychiatry Clin Neurosci 1991; 3:243–254Link, Google Scholar
27 : Is it time to revise the diagnostic criteria for apathy in brain disorders? the 2018 International Consensus Group. Eur Psychiatry 2018; 54:71–76Crossref, Medline, Google Scholar
28 : Different aspects of emotional processes in apathy: application of the French translated Dimensional Apathy Scale. Curr Psychol 2018; 39/564-570Crossref, Google Scholar
29 : The Apathy Inventory: assessment of apathy and awareness in Alzheimer’s disease, Parkinson’s disease and mild cognitive impairment. Int J Geriatr Psychiatry 2002; 17:1099–1105Crossref, Medline, Google Scholar
30 : Are the available apathy measures reliable and valid? a review of the psychometric evidence. J Psychosom Res 2011; 70:73–97Crossref, Medline, Google Scholar
31 : The Lille Apathy Rating Scale (LARS), a new instrument for detecting and quantifying apathy: validation in Parkinson’s disease. J Neurol Neurosurg Psychiatry 2006; 77:579–584Crossref, Medline, Google Scholar
32 : Assessing apathy in everyday clinical practice with the short-form Lille Apathy Rating Scale. Mov Disord 2013; 28:2014–2019Crossref, Medline, Google Scholar
33 : Validation of diagnostic criteria for apathy in Parkinson’s disease. Parkinsonism Relat Disord 2010; 16:656–660Crossref, Medline, Google Scholar
34 : A systematic review of the validity and reliability of apathy scales in neurodegenerative conditions: CORRIGENDUM. Int Psychogeriatr 2015; 27:925Crossref, Medline, Google Scholar
35 : Developing a new apathy measurement scale: Dimensional Apathy Scale. Psychiatry Res 2014; 219:658–663Crossref, Medline, Google Scholar
36 : Prevalence of neuropsychiatric symptoms and their association with functional limitations in older adults in the United States: the Aging, Demographics, and Memory Study. J Am Geriatr Soc 2010; 58:330–337Crossref, Medline, Google Scholar
37 : Multidimensional model of apathy in older adults using partial least squares–path modeling. Age (Dordr) 2016; 38:55Crossref, Medline, Google Scholar
38 : Assessment of neurobehavioural disability: a review of existing measures and recommendations for a comprehensive assessment tool. Brain Inj 2008; 22:905–918Crossref, Medline, Google Scholar
39 :
40 : Behavioural abnormalities contribute to functional decline in Huntington’s disease. J Neurol Neurosurg Psychiatry 2003; 74:120–122Crossref, Medline, Google Scholar
41 : Health-related quality of life in Huntington’s disease: which factors matter most? Mov Disord 2009; 24:574–578Crossref, Medline, Google Scholar
42 : Psychopathology in verified Huntington’s disease gene carriers. J Neuropsychiatry Clin Neurosci 2007; 19:441–448Link, Google Scholar
43 : Social cognition in frontotemporal dementia and Huntington’s disease. Neuropsychologia 2003; 41:688–701Crossref, Medline, Google Scholar
44 : Crime in Huntington’s disease: a study of registered offences among patients, relatives, and controls. J Neurol Neurosurg Psychiatry 1998; 65:467–471Crossref, Medline, Google Scholar
45 : Neuropsychiatric burden in Huntington’s disease. Brain Sci 2017; 7(6)Medline, Google Scholar
46 : Treating apathy in Alzheimer’s disease. Dement Geriatr Cogn Disord 2004; 17:91–99Crossref, Medline, Google Scholar
47 : Are legal measures for the protection of patients with Alzheimer disease useful for the caregivers? Psychol Neuropsychiatr Vieil 2004; 2:141–146Medline, Google Scholar
48 : Behavioral changes in Huntington Disease. Neuropsychiatry Neuropsychol Behav Neurol 2001; 14:219–226Medline, Google Scholar
49 : Neuropsychiatric aspects of Huntington’s disease. J Neurol Neurosurg Psychiatry 2001; 71:310–314Crossref, Medline, Google Scholar
50 : Behavior in Huntington’s disease: dissociating cognition-based and mood-based changes. J Neuropsychiatry Clin Neurosci 2002; 14:37–43Link, Google Scholar
51 : Cognitive impairment related to apathy in early Huntington’s disease. Dement Geriatr Cogn Disord 2006; 21:316–321Crossref, Medline, Google Scholar
52 : Correlates of apathy in Huntington’s disease. J Neuropsychiatry Clin Neurosci 2010; 22:287–294Link, Google Scholar
53 : Apathy is not depression in Huntington’s disease. J Neuropsychiatry Clin Neurosci 2009; 21:266–270Link, Google Scholar
54 : “Frontal” behaviors before the diagnosis of Huntington’s disease and their relationship to markers of disease progression: evidence of early lack of awareness. J Neuropsychiatry Clin Neurosci 2010; 22:196–207Link, Google Scholar
55 : Incidence, course, and predictors of apathy in Huntington’s disease: a two-year prospective study. J Neuropsychiatry Clin Neurosci 2011; 23:434–441Link, Google Scholar
56 : Longitudinal evaluation of neuropsychiatric symptoms in Huntington’s disease. J Neuropsychiatry Clin Neurosci 2012; 24:53–60Link, Google Scholar
57 : Predictors of phenotypic progression and disease onset in premanifest and early-stage Huntington’s disease in the TRACK-HD study: analysis of 36-month observational data. Lancet Neurol 2013; 12:637–649Crossref, Medline, Google Scholar
58 : Neuropsychiatric symptoms in a European Huntington’s disease cohort (REGISTRY). J Neurol Neurosurg Psychiatry 2014; 85:1411–1418Crossref, Medline, Google Scholar
59 : Rating apathy in Huntington’s disease: patients and companions agree. J Huntingtons Dis 2015; 4:49–59Crossref, Medline, Google Scholar
60 : Neuropsychiatric symptoms are very common in premanifest and early stage Huntington’s disease. (published correction appears in Parkinsonism Relat Disord 2016 Oct;31:161) Parkinsonism Relat Disord 2016; 25:58–64Crossref, Medline, Google Scholar
61 : Emotion recognition correlates with social-neuropsychiatric dysfunction in Huntington’s disease. J Int Neuropsychol Soc 2018; 24:417–423Crossref, Medline, Google Scholar
62 : Huntington’s disease gene expansion carriers are aware of their degree of apathy. J Neuropsychiatry Clin Neurosci 2018; 30:183–187Link, Google Scholar
63 : Apathy and atrophy of subcortical brain structures in Huntington's disease: a two-year follow-up study. Neuroimage Clin 2018;19:66–70Crossref, Medline, Google Scholar
64 : Non-motor symptoms in Huntington’s disease: a comparative study with Parkinson’s disease. J Neurol 2019; 266:1340–1350Crossref, Medline, Google Scholar
65 : Apathy is related to cognitive control and striatum volumes in prodromal Huntington’s disease. J Int Neuropsychol Soc 2019; 25:462–469Crossref, Medline, Google Scholar
66 : Insensitivity to loss predicts apathy in Huntington’s disease. Mov Disord 2019; 34:1381–1391Crossref, Medline, Google Scholar
67 : White matter cortico-striatal tracts predict apathy subtypes in Huntington’s disease. Neuroimage Clin 2019; 24:101965Crossref, Medline, Google Scholar
68 : Assessing mental health difficulties of persons with Huntington’s disease: does informant presence make a difference? J Neuropsychiatry Clin Neurosci 2020; 32:244–251Link, Google Scholar
69 : Apathy predicts rate of cognitive decline over 24 months in premanifest Huntington’s disease. Psychol Med 2020; 51:1338 –1344Crossref, Medline, Google Scholar
70 : Maladie de Huntington. Swiss Med Forum 2018; 15:1022–1026Google Scholar
71 : Huntington’s disease: a clinical review. Eur J Neurol 2018; 25:24–34Crossref, Medline, Google Scholar
72 : The prevalence, clinical correlates and treatment of apathy in Alzheimer’s disease. Eur J Psychiatry 2006; 20:96–106Crossref, Google Scholar
73 : Prevalence of apathy following head injury. Brain Inj 1998; 12:87–92Crossref, Medline, Google Scholar
74 : Apathy and depression: separate factors in Parkinson’s disease. J Int Neuropsychol Soc 2011; 17:1058–1066Crossref, Medline, Google Scholar
75 : Apathy is not depression. J Neuropsychiatry Clin Neurosci 1998; 10:314–319Link, Google Scholar
76 : Categories versus dimensions in personality and psychopathology: a quantitative review of taxometric research. Psychol Med 2012; 42:903–920Crossref, Medline, Google Scholar
77 :
78 : Network analysis: an integrative approach to the structure of psychopathology. Annu Rev Clin Psychol 2013; 9:91–121Crossref, Medline, Google Scholar
79 : The pathoplasticity of dysphoric episodes: differential impact of stressful life events on the pattern of depressive symptom inter-correlations. Psychol Med 2012; 42:957–965Crossref, Medline, Google Scholar
80 : Deconstructing the construct: a network perspective on psychological phenomena. New Ideas Psychol 2013; 31:43–53Crossref, Google Scholar
81 : Transdiagnostic networks: commentary on Nolen-Hoeksema and Watkins (2011). Perspect Psychol Sci 2011; 6:610–614Crossref, Medline, Google Scholar
82 : A psychological model of mental disorders. Harv Rev Psychiatry 2014; 13:206–217Crossref, Google Scholar
83 : Imagine there’s no diagnosis, it’s easy if you try. Psychopathol Rev 2015; 2:154–161Crossref, Google Scholar
84 : Deconstructing negative symptoms of schizophrenia: avolition-apathy and diminished expression clusters predict clinical presentation and functional outcome. J Psychiatr Res 2013; 47:783–790Crossref, Medline, Google Scholar



